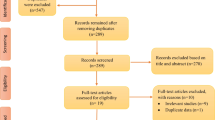
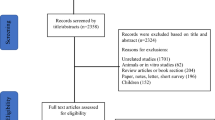
Abstract
Background
Obstructive sleep apnea–hypopnea syndrome (OSAHS) is a disease characterized with intermittent hypoxia and sleep fragmentation. Obesity and gender are major risk factors for the onset of OSAHS. Previous studies on obese men with OSAHS have been performed, while few studies on non-obese men with OSAHS have been carried out. The purpose of this study was to explore the clinical characteristics of polysomnography and blood biochemical indexes in non-obese men with OSAHS and to identify the possible influencing factors.
Methods
This retrospective study included patients with OSAHS who underwent polysomnography in our hospital. General clinical data such as overnight polysomnography and biochemical indicators were recorded. The patients were divided into two groups according to the apnea–hypopnea index (AHI): mild to moderate OSAHS and severe OSAHS. The differences in biochemical parameters such as the levels of γ-glutamine transaminase (GGT), triglyceride (TG), glucose (GLU), and sleep structure parameters such as N1, N2, slow-wave sleep (SWS), and rapid eye movement (REM) sleep were compared and analyzed. Spearman correlation analysis and logistic regression were used to identify the risk factors of non-obese men with OSAHS. ROC curves were used to evaluate the predictive ability of SWS and GGT on disease severity.
Results
Of 94 non-obese men with OSAHS, 49 had mild to moderate OSAHS and 45 had severe OSAHS. Our data suggested that the levels of low oxygen saturation (L-SaO2), mean oxygen saturation (M-SaO2), SWS, and GGT were significantly changed in the mild to moderate OSAHS group compared with the severe group (p < 0.05). For patients with OSAHS, the proportion of SWS in the group with severe OSAHS was higher than that in the mild to moderate group (p < 0.05), and the serum GGT enzyme levels were significantly elevated in the severe group compared to the mild to moderate group (p < 0.05). Using logistic regression analyses, our data revealed that both SWS and GGT enzyme levels were independent risk factors for AHI (p < 0.05). In addition, the results of correlation analysis indicated that SWS was related to triglyceride (TG), total cholesterol (TC), apolipoprotein E (APOE), and triglyceride glucose (TyG) index (p < 0.05); GGT was related to TG, TC, APOE, and TyG index (p < 0.05). Furthermore, SWS was independently associated with GGT (p < 0.05). The area under the ROC curve plotted with the combined coefficient of SWS and serum GGT was 0.728, which was predictive of the disease severity.
Conclusions
These results suggest that SWS and GGT are independent associated factors of the severity of the disease. However, TyG index was not an independent associated factor of the severity of disease in non-obese men with OSAHS. In addition, SWS and GGT were negatively correlated. SWS combined with serum GGT may be predictive of the severity of the disease. This study may have added to our understanding of the pathogenesis of OSAHS in non-obese men.


Similar content being viewed by others
Data availability
Data will be made available on reasonable request.
References
White DP (1995) Sleep-related breathing disorder.2. Pathophysiology of obstructive sleep apnoea. Thorax 50(7):797–804. https://doi.org/10.1136/thx.50.7.797
Peppard PE, Young T, Palta M et al (2000) Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 342(19):1378–1384. https://doi.org/10.1056/NEJM200005113421901
Gottlieb DJ, Yenokyan G, Newman AB et al (2010) Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: the sleep heart health study. Circulation 122(4):352–360. https://doi.org/10.1161/CIRCULATIONAHA.109.901801
Yaggi HK, Concato J, Kernan WN et al (2005) Obstructive sleep apnea as a risk factor for stroke and death. N Engl J Med 353(19):2034–2041. https://doi.org/10.1056/NEJMoa043104
Redline S, Yenokyan G, Gottlieb DJ et al (2010) Obstructive sleep apnea-hypopnea and incident stroke: the sleep heart health study. Am J Respir Crit Care Med 182(2):269–277. https://doi.org/10.1164/rccm.200911-1746OC
Kendzerska T, Gershon AS, Hawker G et al (2014) Obstructive sleep apnea and incident diabetes. A historical cohort study. Am J Respir Crit Care Med 190(2):218–225. https://doi.org/10.1164/rccm.201312-2209OC
Peppard PE, Szklo-Coxe M, Hla KM et al (2006) Longitudinal association of sleep-related breathing disorder and depression. Arch Intern Med 166(16):1709–1715. https://doi.org/10.1001/archinte.166.16.1709
Turner K, Zambrelli E, Lavolpe S et al (2019) Obstructive sleep apnea: neurocognitive and behavioral functions before and after treatment. Funct Neurol 34(2):71–78
Wimms A, Woehrle H, Ketheeswaran S et al (2016) Obstructive sleep apnea in women: specific issues and interventions. Biomed Res Int 2016:1764837. https://doi.org/10.1155/2016/1764837
Young T, Skatrud J, Peppard PE (2004) Risk factors for obstructive sleep apnea in adults. JAMA 291(16):2013–2016. https://doi.org/10.1001/jama.291.16.2013
Frey WC, Pilcher J (2003) Obstructive sleep-related breathing disorders in patients evaluated for bariatric surgery. Obes Surg 13(5):676–683. https://doi.org/10.1381/096089203322509228
Heinzer R, Vat S, Marques-Vidal P et al (2015) Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med 3(4):310–318. https://doi.org/10.1016/S2213-2600(15)00043-0
Akahoshi T, Uematsu A, Akashiba T et al (2010) Obstructive sleep apnoea is associated with risk factors comprising the metabolic syndrome. Respirology (Carlton, Vic.) 15(7):1122–1126. https://doi.org/10.1111/j.1440-1843.2010.01818.x
Gündüz C, Basoglu OK, Hedner J et al (2018) Obstructive sleep apnoea independently predicts lipid levels: data from the European Sleep Apnea Database. Respirology (Carlton, Vic.) 23(12):1180–1189. https://doi.org/10.1111/resp.13372
Ratnavadivel R, Chau N, Stadler D et al (2009) Marked reduction in obstructive sleep apnea severity in slow wave sleep. J Clin Sleep Med 5(6):519–524
Léger D, Debellemaniere E, Rabat A et al (2018) Slow-wave sleep: from the cell to the clinic. Sleep Med Rev 41:113–132. https://doi.org/10.1016/j.smrv.2018.01.008
Raison CL, Rye DB, Woolwine BJ et al (2010) Chronic interferon-alpha administration disrupts sleep continuity and depth in patients with hepatitis C: association with fatigue, motor slowing, and increased evening cortisol. Biol Psychiat 68(10):942–949. https://doi.org/10.1016/j.biopsych.2010.04.019
Diekelmann S, Born J (2010) The memory function of sleep. Nat Rev Neurosci 11(2):114–126. https://doi.org/10.1038/nrn2762
Tasali E, Leproult R, Ehrmann DA et al (2008) Slow-wave sleep and the risk of type 2 diabetes in humans. Proc Natl Acad Sci USA 105(3):1044–1049. https://doi.org/10.1073/pnas.0706446105
Somers VK, Dyken ME, Mark AL et al (1993) Sympathetic-nerve activity during sleep in normal subjects. N Engl J Med 328(5):303–307. https://doi.org/10.1056/NEJM199302043280502
Sánchez-García A, Rodríguez-Gutiérrez R, Mancillas-Adame L et al (2020) Diagnostic accuracy of the triglyceride and glucose index for insulin resistance: a systematic review. Int J Endocrinol 2020:4678526. https://doi.org/10.1155/2020/4678526
Simental-Mendía LE, Rodríguez-Morán M, Guerrero-Romero F (2008) The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord 6(4):299–304. https://doi.org/10.1089/met.2008.0034
Vasques AC, Novaes FS, de Oliveira MS et al (2011) TyG index performs better than HOMA in a Brazilian population: a hyperglycemic clamp validated study. Diabetes Res Clin Pract 93(3):e98–e100. https://doi.org/10.1016/j.diabres.2011.05.030
Guerrero-Romero F, Villalobos-Molina R, Jim’enez-Flores JR et al (2016) Fasting triglycerides and glucose index as a diagnostic test for insulin resistance in young adults. Arch Med Res 47(5):382–387. https://doi.org/10.1016/j.arcmed.2016.08.012
Kang HH, Kim SW, Lee SH (2020) Association between triglyceride glucose index and obstructive sleep apnea risk in Korean adults: a cross-sectional cohort study. Lipids Health Dis 19(1):182. https://doi.org/10.1186/s12944-020-01358-9
Sánchez-Armengol A, Villalobos-López P, Caballero-Eraso C et al (2015) Gamma glutamyl transferase and oxidative stress in obstructive sleep apnea: a study in 1744 patients. Sleep Breath 19(3):883–890. https://doi.org/10.1007/s11325-014-1115-5
Ruehland WR, Rochford PD, O’Donoghue FJ et al (2009) The new AASM criteria for scoring hypopneas: impact on the apnea hypopnea index. Sleep 32(2):150–7. https://doi.org/10.1093/sleep/32.2.150
Berry RB, Budhiraja R, Gottlieb DJ et al (2012) American Academy of Sleep Medicine. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med 8(5):597–619. https://doi.org/10.5664/jcsm.2172
Dresner A, Laurent D, Marcucci M et al (1999) Effects of free fatty acids on glucose transport and IRS-1-associated phosphatidylinositol 3-kinase activity. J Clin Investig 103(2):253–259. https://doi.org/10.1172/JCI5001
Xu H, Barnes GT, Yang Q et al (2003) Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Investig 112(12):1821–1830. https://doi.org/10.1172/JCI19451
Huang W, Liu Y, Wang X et al (2021) Effect of interaction between slow wave sleep and obstructive sleep apnea on insulin resistance: a large-scale study. Nat Sci Sleep 13:739–749. https://doi.org/10.2147/NSS.S311130
Qi JC, Huang JC, Lin QC et al (2016) Relationship between obstructive sleep apnea and nonalcoholic fatty liver disease in nonobese adults. Sleep Breath 20(2):529–35. https://doi.org/10.1007/s11325-015-1232-9
Feng N, Yang J, Xu H et al (2021) The associations between sleep architecture and metabolic parameters in patients with obstructive sleep apnea: a hospital-based cohort study. Front Neurol 12:606031. https://doi.org/10.3389/fneur.2021.606031
Xu H, Xia Y, Li X et al (2020) Association between obstructive sleep apnea and lipid metabolism during REM and NREM sleep. J Clin Sleep Med 16(4):475–482. https://doi.org/10.5664/jcsm.8242
Bikov A, Lazar Z, Horvath P et al (2019) Association between serum lipid profile and obstructive respiratory events during REM and Non-REM sleep. Lung 197(4):443–450. https://doi.org/10.1007/s00408-019-00195-7
Jun JC, Shin MK, Yao Q et al (2012) Acute hypoxia induces hypertriglyceridemia by decreasing plasma triglyceride clearance in mice. Am J Physiol Endocrinol Metab 303(3):E377-88. https://doi.org/10.1152/ajpendo.00641.2011
Martínez-Cerón E, Casitas R, Galera R et al (2021) Contribution of sleep characteristics to the association between obstructive sleep apnea and dyslipidemia. Sleep Med 84:63–72. https://doi.org/10.1016/j.sleep.2021.05.012
Van Cauter E, Latta F, Nedeltcheva A et al (2004) Reciprocal interactions between the GH axis and sleep. Growth Horm IGF Res 14(Suppl A):S10–S17. https://doi.org/10.1016/j.ghir.2004.03.006
Adedayo AM, Olafiranye O, Smith D et al (2014) Obstructive sleep apnea and dyslipidemia: evidence and underlying mechanism. Sleep Breath 18(1):13–18. https://doi.org/10.1007/s11325-012-0760-9
Liu X, Lin Q, Fan K et al (2021) The effects of genetic polymorphisms of APOE on circulating lipid levels in middle-aged and elderly chinese Fujian Han population: toward age- and sex-personalized management. Lipids Health Dis 20(1):158. https://doi.org/10.1186/s12944-021-01587-6
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Ethics Committee of the Second Affiliated Hospital of Chongqing Medical University (2022 Colum Review No. (144)).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, Q., Li, H., Gan, X. et al. Relationship between slow-wave sleep and serum γ-glutamine transaminase in non-obese men with obstructive sleep apnea–hypopnea syndrome. Sleep Breath 27, 1717–1724 (2023). https://doi.org/10.1007/s11325-022-02775-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-022-02775-z