Abstract
Background
The purpose of the study was to examine the effects of continuous positive airway pressure (CPAP) treatment on inflammation parameters in patients with obstructive sleep apnea syndrome (OSAS).
Methods
Patients aged 18 to 65 years who underwent polysomnography (PSG) in the sleep clinic between January 1, 2019, and December 31, 2019, were included in the study. Patients with severe OSAS initiated treatment with CPAP. Patients and control subjects were assessed for levels of periostin, TNF-alpha, TGF-beta, and IL-6. Patients were re-evaluated 3 months later. Comparisons for the serum markers were made between controls and patients of different severity of OSAS. Comparisons of serum markers were also made between baseline and 3 month follow-up.
Results
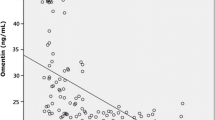
A total of 92 patients were enrolled in the study, including 25 controls (apnea-hypopnea index or AHI < 5/h), 39 patents with mild to moderate OSAS who did not receive CPAP, and 28 patients with severe OSAS receiving CPAP treatment. When all three groups were compared, levels of periostin, TNF-alpha, TGF-beta, and IL-6, as inflammatory markers, were higher in the OSAS group, though not at a statistically significant level. In patients with severe OSAS, there were statistically significant decreases in the TGF-beta 1, TNF-alpha, and IL-6 values between baseline values and the same measures taken after 3 months of CPAP treatment. Periostin values also decreased after treatment, but this decrease was not at a significant level.
Conclusion
Inflammatory parameters of patients with OSAS were significantly higher compared with healthy participants. Regression of inflammation was detected after CPAP treatment.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Gaines J, Vgontzas AN, Fernandez-Mendoza J et al (2018) Obstructive sleep apnea and the metabolic syndrome: the road to clinically-meaningful phenotyping, improved prognosis, and personalized treatment. Sleep Med Rev 42:211–219. https://doi.org/10.1016/j.smrv.2018.08.009
Dursunoğlu N, Dursunoğlu D (2005) Obstrüktif uyku apne hipopne sendromunun kardiyovasküler sistem üzerine etkileri [The effects of obstructive sleep apnea hypopnea syndrome on cardiovascular system]. Anadolu Kardiyol Derg. 5(1):41–45 (Turkish)
Wolk R, Somers VK (2006) Obesity-related cardiovascular disease: implications of obstructive sleep apnea. Diabetes Obes Metab 8(3):250–260. https://doi.org/10.1111/j.1463-1326.2005.00508.x
Meier-Ewert HK, Ridker PM, Rifai N et al (2004) Effect of sleep loss on C-reactive protein, an inflammatory marker of cardiovascular risk. J Am Coll Cardiol 43(4):678–683. https://doi.org/10.1016/j.jacc.2003.07.050
Masuoka M, Shiraishi H, Ohta S et al (2012) Periostin promotes chronic allergic inflammation in response to Th2 cytokines. J Clin Invest 122(7):2590–2600. https://doi.org/10.1172/JCI58978
Izuhara K, Nunomura S, Nanri Y, Ono J, Takai M, Kawaguchi A (2019) Periostin: an emerging biomarker for allergic diseases. Allergy 74:2116–2128
Kimura H, Konno S, Makita H, Taniguchi N, Kimura H, Goudarzi H, Shimizu K, Suzuki M, Shijubo N, Shigehara K, Ono J, Izuhara K, Ito YM, Nishimura M (2018) Serum periostin is associated with body mass index and allergic rhinitis in healthy and asthmatic subjects. Allergol Int 67(3):357–363. https://doi.org/10.1016/j.alit.2017.11.006
Shirai T, Hirai K, Gon Y, Maruoka S, Mizumura K, Hikichi M, Holweg C, Itoh K, Inoue H, Hashimoto S (2019) Combined assessment of serum periostin and YKL-40 may identify asthma-COPD overlap. J Allergy Clin Immunol Pract 7(1):134-145.e1. https://doi.org/10.1016/j.jaip.2018.06.015
Zhang Z, Nie F, Kang C, Chen B, Qin Z, Ma J, Ma Y, Zhao X (2014) Increased periostin expression affects the proliferation, collagen synthesis, migration and invasion of keloid fibroblasts under hypoxic conditions. Int J Mol Med 34(1):253–261. https://doi.org/10.3892/ijmm.2014.1760
Çayakar A (2018) What is tumor necrosis factor alpha ? Turkiye Klinikleri J Intern Med 3(2):67–76. https://doi.org/10.5336/intermed.2018-61424
Tanaka T, Narazaki M, Kishimoto T (2014) IL-6 in inflammation immunity and disease. Cold Spring Harb Perspect Biol 6(10):a016295. https://doi.org/10.1101/cshperspect.a016295
Nagayoshi M, Yamagishi K, Tanigawa T et al (2011) Risk factors for snoring among Japanese men and women: a community-based cross-sectional study. Sleep Breath 15(1):63–69. https://doi.org/10.1007/s11325-009-0319-6
Young T, Peppard PE, Taheri S (2005) Excess weight and sleep-disordered breathing. J Appl Physiol (1985) 99(4):1592–1599
Kandasamy G, Almaghaslah D, Sivanandy P et al (2019) Effectiveness of nasal continuous airway pressure TREATMENT in patients with obstructive sleep apnea. Int J Health Plann Manage 34(2):e1200–e1207. https://doi.org/10.1002/hpm.2758
Ding X, Yu C, Liu Y et al (2016) Chronic obstructive sleep apnea accelerates pulmonary remodeling via TGF-β/miR-185/CoLA1 signaling in a canine model. Oncotarget 7(36):57545–57555
McNicholas WT (2009) Obstructive sleep apnea and inflammation. Prog Cardiovasc Dis 51(5):392–399. https://doi.org/10.1016/j.pcad.2008.10.005
Walsh JA, Duffin KC, Crim J et al (2012) Lower frequency of obstructive sleep apnea in spondyloarthritis patients taking TNF-inhibitors. J Clin Sleep Med 8(6):643–648. https://doi.org/10.5664/jcsm.2254
Ifergane G, Ovanyan A, Toledano R et al (2016) Obstructive sleep apnea in acute stroke: a role for systemic inflammation. Stroke 47(5):1207–1212. https://doi.org/10.1161/STROKEAHA.115.011749
Thunström E, Glantz H, Fu M et al (2015) Increased inflammatory activity in nonobese patients with coronary artery disease and obstructive sleep apnea. Sleep 38(3):463–471. https://doi.org/10.5665/sleep.4510
Leon-Cabrera S, Arana-Lechuga Y, Esqueda-León E et al (2015) Reduced systemic levels of IL-10 are associated with the severity of obstructive sleep apnea and insulin resistance in morbidly obese humans. Mediators Inflamm 2015:493409. https://doi.org/10.1155/2015/493409
Li Q, Zheng X (2017) Tumor necrosis factor alpha is a promising circulating biomarker for the development of obstructive sleep apnea syndrome a meta analysis. Oncotarget 8(16):27616–27626. https://doi.org/10.18632/oncotarget.15203
Clark DA, Coker R (1998) Transforming growth factor-beta (TGF-beta). Int J Biochem Cell Biol 30(3):293–298. https://doi.org/10.1016/s1357-2725(97)00128-3
Ding WX, Dong YB, Ding N et al (2014) Adiponectin protects rat heart from left ventricular remodeling induced by chronic intermittent hypoxia via inhibition of TGF-β/smad2/3 pathway. J Thorac Dis 6(9):1278–1284. https://doi.org/10.3978/j.issn.2072-1439.2014.07.44
Steffanina A, Proietti L, Antonaglia C et al (2015) The plasminogen system and transforming growth factor-β in subjects with obstructive sleep apnea syndrome: effects of CPAP treatment. Respir Care 60(11):1643–1651. https://doi.org/10.4187/respcare.03571
Alberti A, Sarchielli P, Gallinella E et al (2003) Plasma cytokine levels in patients with obstructive sleep apnea syndrome: a preliminary study. J Sleep Res 12(4):305–311. https://doi.org/10.1111/j.1365-2869.2003.00361.x
Tanaka T, Narazaki M, Kishimoto T (2014) IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol 6(10):a016295. https://doi.org/10.1101/cshperspect.a016295
Li Y, Vgontzas AN, Fernandez-Mendoza J, et al (2017) Objective, but not subjective, sleepiness is associated with inflammation in sleep apnea. Sleep 40 https://doi.org/10.1093/sleep/zsw033.
Huang YS, Guilleminault C, Hwang FM et al (2016) Inflammatory cytokines in pediatric obstructive sleep apnea. Medicine (Baltimore) 95:e4944. https://doi.org/10.1097/MD.0000000000004944
Kaditis AG, Gozal D, Khalyfa A et al (2014) Variants in C-reactive protein and IL-6 genes and susceptibility to obstructive sleep apnea in children: a candidate-gene association study in European American and Southeast European populations. Sleep Med 15:228–235. https://doi.org/10.1016/j.sleep.2013.08.795
Sunadome H, Matsumoto H, Tachikawa R et al (2020) Role of serum periostin in severe obstructive sleep apnea with albuminuria: an observational study. Respir Res 21(1):143. https://doi.org/10.1186/s12931-020-01413-0
Ji L, Liu Y, Liu P, Ji G, He J, Gan Y, Zhu S, Chen B, Zhang W (2021) Serum periostin and TNF-α levels in patients with obstructive sleep apnea-hypopnea syndrome. Sleep and Breathing 25:331–337. https://doi.org/10.1007/s11325-020-02124-y
Izuhara K, Nunomura S, Nanri Y et al (2017) Periostin in inflammation and allergy. Cell Mol Life Sci 74(23):4293–4303. https://doi.org/10.1007/s00018-017-2648-0
Kent BD, Ryan S, McNicholas WT (2011) Obstructive sleep apnea and inflammation: relationship to cardiovascular co-morbidity. Respir Physiol Neurobiol 178(3):475–481. https://doi.org/10.1016/j.resp.2011.03.015
Karamanli H, Özol D, Ugur KS et al (2014) Influence of CPAP treatment on airway and systemic inflammation in OSAS patients. Sleep Breath 18(2):251–256. https://doi.org/10.1007/s11325-012-0761-8
Steiropoulos P, Kotsianidis I, Nena E et al (2009) Long-term effect of continuous positive airway pressure TREATMENT on inflammation markers of patients with obstructive sleep apnea syndrome. Sleep 32(4):537–543. https://doi.org/10.1093/sleep/32.4.537
Yokoe T, Minoguchi K, Matsuo H et al (2003) Elevated levels of C-reactive protein and interleukin-6 in patients with obstructive sleep apnea syndrome are decreased by nasal continuous positive airway pressure. Circulation 107(8):1129–1134. https://doi.org/10.1161/01.cir.0000052627.99976.18
Guasti L, Marino F, Cosentino M et al (2011) Cytokine production from peripheral blood mononuclear cells and polymorphonuclear leukocytes in patients studied for suspected obstructive sleep apnea. Sleep Breath 15(1):3–11. https://doi.org/10.1007/s11325-009-0315-x
Funding
Our study was supported by the Hatay Mustafa Kemal University Scientific Research Projects Coordinator (Project Number: 19.U.013).
Author information
Authors and Affiliations
Contributions
The manuscript has been read and approved by all the authors. The requirements for authorship as stated earlier in this document have been met and each author believes that the manuscript represents honest work.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Confirmation of the Local Ethics Committee was obtained. Our study was approved by the Ethics Committee of Mustafa Kemal University Faculty of Medicine (Date and Number: 2019/54). The purpose of our study was explained to the participants in detail, and informed by Helsinki rules, and consent was obtained.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tosun, F., Babayiğit, C., Dikmen, N. et al. The effect of continuous positive airway pressure treatment on inflammatory parameters and periostin levels in patients with obstructive sleep apnea syndrome. Sleep Breath 27, 275–282 (2023). https://doi.org/10.1007/s11325-022-02616-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-022-02616-z