Abstract
Background
Continuous positive airway pressure (CPAP) in the treatment of obstructive sleep apnea can produce troublesome nasal symptoms, especially congestion, which may affect the continuity of using CPAP. Intranasal steroids are often prescribed to reduce these side effects, although few recent studies exist supporting the benefits of this treatment for CPAP-induced nasal side effects.
Methods
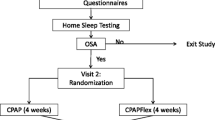
Eighty-three patients with OSA were enrolled in a prospective, randomized controlled study. All patients received CPAP treatment and were divided in two groups. The study group was prescribed fluticasone furoate nasal spray 55 μg, and the compliance to CPAP for patients in both groups was recorded by device memory card. Total nasal symptom score was assessed using a questionnaire by direct interview, with follow-up performed at 30 and 90 days after treatment.
Result
Compliance to CPAP increased in both groups with significantly greater compliance in the intranasal steroid group compared to the control group without intranasal steroid (P value = 0.002, 0.001, and 0.020, respectively) after 90 days of treatment. No difference in nasal symptoms was found between the groups after 30 days of treatment. However, adding an intranasal steroid resulted in decreased rhinorrhea and congestion symptoms (P value < 0.001 and < 0.001) after 90 days of treatment.
Conclusion
The addition of an intranasal steroid decreased the frequency of nasal symptoms, especially rhinorrhea and congestion, among patients with OSA initiating CPAP therapy and increased compliance to CPAP after 90 days of treatment.
Trial registration
IRB approval ID: R179h
Clinical trial ID: TCTR20200715001

Similar content being viewed by others
References
Schwab RJ, Gupta KB, Gefter WB, Metzger LJ (1995) Upper airway and soft tissue anatomy in normal subjects and patient with sleep-disordered breathing. Significant of the lateral pharyngeal walls. Am J Respir Crit Care Med 152:1673–1689
Fogel RB, White DP (2004) Sleep. 2: pathophysiology of obstructive sleep apnoea/hypopnoea syndrome. Thorax 59(2):159–163
Cordero-Guevara J. ea. The association between sleep apnea and the risk of traffic accidents. N Engl J Med 1999; 340:847–851
Kapa S, Sert Kuniyoshi FH, Somer VK (2008) Sleep apnea and hypertension: interaction and implications for management. Hypertension. 51(3):605–608
Jean-Louis G, Zizi F, Clark LT, Brown CD, McFarlane SI (2008) Obstructive sleep apnea and cardiovascular disease: role of the metabolic syndrome and its components. J Clin Sleep Med 4(3):261–272
Sharafkhaneh A, Giray N, Richardson P, Younf T, Hirshkowitz M (2005) Association of psychiatric disorder and sleep apnea in large cohort. Sleeo. 28(11):1405–1411
Pavwoski P, Shelgikar AV (2017) Treatment options for obstructive sleep apnea. Neurol Clin Pract 7(1):77–85. https://doi.org/10.1212/CPJ.0000000000000320
Antonescu-Turcu A, Parthasarathy S (2010) CPAP and bi-level PAP therapy: new and established roles. Respir Care 55(9):1216–1229
Sawyer AM, Gooneratne NS, Marcus CL, Ofer D, Richards KC, Weaver TE (2011) A systematic review of CPAP adherence across age groups: clinical and empiric insights for developing CPAP adherence interventions. Sleep Med Rev 15(6):343–356. https://doi.org/10.1016/j.smrv.2011.01.003
Pepin JL, Leger P, Veale D, Langevin B, Robert D, Levy P (1995) Side effects of nasal continuous positive airway pressure in sleep apnea syndrome. Study of 193 pateints in two French sleep centers. Chest. 107(2):375–381
Nilius G, Franke KJ, Domanski U, Rühle KH (2007) Beschwerden an den oberen Atemwegen bei obstruktiver Schlafapnoe - Auswirkungen der CPAP-Therapie [Upper airway complaints of patients with obstructive sleep apnea - effect of CPAP]. Pneumologie 61(1):15–19. German. https://doi.org/10.1055/s-2006-954966
Balsalobre L, Figueiredo AB, Pezato R, Fujita RR (2019) Effect of topical corticosteroids on nasal patency after acute positive airway pressure exposure. Braz J Otorhinolaryngol S1808–8694(19):30123–30125. https://doi.org/10.1016/j.bjorl.2019.09.011 Epub ahead of print
Efird J (2011) Blocked randomization with randomly selected block sizes. Int J Environ Res Public Health 8(1):15–20
Kapur VK, Auckley DH, Chowdhuri S, Kuhlmann DC, Mehra R, Ramar K, Harrod CG (2017) Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med 13(3):479–504
Kushida CA, Chediak A, Berry RB, Brown LK, Gozal D, Iber C, Parthasarathy S, Quan SF, Rowley JA, Positive Airway Pressure Titration Task Force, American Academy of Sleep Medicine (2008) Clinical guidelines for the manual titration of positive airway pressure in patients with obstructive sleep apnea. J Clin Sleep Med 4(2):157–171
Lopez-Campos JL, Garcia Polo C, Leon Jimenez A, Gonzalez-Moya E, Arnedillo A, Fernandez Berni JJ (2007) CPAP titration: different methods for similar clinical results. Eur J Intern Med 18(3):230–234
Berry RB, Sriram P (2014) Auto-adjusting positive airway pressure treatment for sleep apnea diagnosed by home sleep testing. J Clin Sleep Med 10(12):1269–1275. Published 2014 Dec 15. https://doi.org/10.5664/jcsm.4272
Banhiran W, Assanasen P, Nopmaneejumruslers C, Metheetrairut C (2011) Epworth sleepiness scale in obstructive sleep disordered breathing: the reliability and validity of the Thai version. Sleep Breath 15(3):571–577
Banhiran W, Durongphan A, Saleesing C, Chongkolwatana C (2014) Diagnostic properties of the STOP-Bang and its modified version in screening for obstructive sleep apnea in Thai patients. J Med Assoc Thail 97(6):644–654
Baltzan MA, Elkholi O, Wolkove N (2009) Evidence of interrelated side effects with reduced compliance in patients treated with nasal continuous positive airway pressure. Sleep Med 10(2):198–205. https://doi.org/10.1016/j.sleep.2007.12.005 Epub 2008 Mar 7
Kumar R, Kumar D, Paeakh A (2012) Fluticasone furoate: a new intranasal corticosteroid. J Postgrad Med 58(1):79–83
Charakorn N, Hirunwiwatkul P, Chirakalwasan N, Chaitusaney B, Prakassajjatham M (2017) The effects of topical nasal steroids on continuous positive airway pressure compliance in patients with obstructive sleep apnea: a systematic review and meta-analysis. Sleep Breath 21(1):3–8. https://doi.org/10.1007/s11325-016-1375-3
Strobel W, Schlageter M, Andersson M et al (2001) Intranasal steroid treatment does not improve CPAP compliance in unselected patients with OSAS. Respir Med 105:310–315
Zheng M, Wang X, Ge S, Gu Y, Ding X, Zhang Y, Ye J, Zhang L (2017) Allergic and non-allergic rhinitis are common in obstructive sleep apnea but not associated with disease severity. J Clin Sleep Med 13:959–966. https://doi.org/10.5664/jcsm.6694
Thompson A, Sardana N, Craig TJ (2013) Sleep impairment and daytime sleepiness in patients with allergic rhinitis: the role of congestion and inflammation. Ann Allergy Asthma Immunol 111:446–451. https://doi.org/10.1016/j.anai.2013.05.020
Gelardi M, Carbonara G, Maffezzoni E, Marvisi M, Quaranta N, Ferri R (2012) Regular CPAP utilization reduces nasal inflammation assessed by nasal cytology in obstructive sleep apnea syndrome. Sleep Med 13:859–863. https://doi.org/10.1016/j.sleep.2012.04.004
Shadan FF, Jalowayski AA, Fahrenholz J (2005) Nasal cytology: a marker of clinically silent inflammation in patients with obstructive sleep apnea and a predictor of noncompliance with nasal CPAP therapy. J Clin Sleep Med 1:266–270
Cruz AA, Naclerio RM, Proud D, Togias A (2006) Epithelial shedding is associated with nasal reactions to cold, dry air. J Allergy Clin Immunol 117(6):1351–1358. https://doi.org/10.1016/j.jaci.2006.01.054 Epub 2006 May 2
Kiely JL, Nolan P, McNicholas WT (2004) Intranasal corticosteroid therapy for obstructive sleep apnoea in patients with co-existing rhinitis. Thorax. 59(1):50–55
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Human and animal rights
The questionnaire and methodology used in this study were approved by the Human Research Ethics Committee of the Institutional Review Board Royal Thai Army Medical Department, Thailand (R179h/60), and registered at Clinicaltrials.in.th (TCTR20200715001).
Informed consent
Informed consent was obtained from all individuals in this study prior to participation.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Segsarnviriya, C., Chumthong, R. & Mahakit, P. Effects of intranasal steroids on continuous positive airway pressure compliance among patients with obstructive sleep apnea. Sleep Breath 25, 1293–1299 (2021). https://doi.org/10.1007/s11325-020-02236-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-020-02236-5