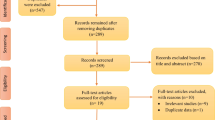
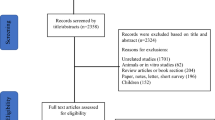
Abstract
Purpose
The pathogenesis of cardiovascular disease (CVD) in patients with obstructive sleep apnea (OSA) is unclear. Several studies have suggested that CVD may be caused by oxidative stress from chronic intermittent hypoxia and associated vascular endothelial dysfunction. Oxidative stress in patients with OSA can induce endothelial cell apoptosis, aggravate vascular endothelial damage, and promote the expression of redox-sensitive genes and adhesion molecules. No meta-analysis has explored whether or not OSA is related to nitric oxide (NO).
Method
To assess the association between serum/plasma NO levels and OSA, we performed a meta-analysis of the literature on the subject to grade the strength of evidence.
Results
OSA was significantly related to decreased serum or plasma NO levels (WMD = − 11.66, 95% CI − 17.21 to − 6.11; P < 0.01). Among the studies analyzed, there was high degree of heterogeneity (I2 = 79%, P < 0.01). Sensitivity analysis showed that after omitting any single study or converting a random effects model (REM) to a fixed effects model (FEM), the main results still held.
Conclusions
This meta-analysis suggests a strong correlation between OSA and serum or plasma NO levels which may explain the link between intermittent hypoxia of OSA and risk of CVD. The strength of this finding may spur further basic and clinical research into vascular endothelial dysfunction in patients with OSA.









Similar content being viewed by others
References
Wiggins C (1993) The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med 328(17):1230–1235. https://doi.org/10.1056/NEJM199304293281704
Lam CM, Lui MS, Ip SM (2010) Diabetes and metabolic aspects of OSA. Sleep Apnoea:189–215. https://doi.org/10.1183/1025448x.00024809
McNicholas WT, Bonsignore MR, Management Committee of EU COST ACTION B26 (2007) Sleep apnoea as an independent risk factor for cardiovascular disease: current evidence, basic mechanisms and research priorities. Eur Respir J 29(1):158–178. https://doi.org/10.1183/09031936.00027406
Young T, Peppard EP, Gottieb DJ (2002) Epidemiology of obstructive sleep apnea. A population health perspective. Am J Respir Crit Care Med 165:1217–1239. https://doi.org/10.1164/rccm.2109080
Tanigawa T, Tachibana N, Yamagishi K (2004) Relationship between sleep-disordered breathing and blood pressure levels in community-based samples of Japanese men. Hypertens Res 27:479–484. https://doi.org/10.1291/hypres.27.479
Wu ZH, Yang XP, Niu X (2018) The relationship between obstructive sleep apnea hypopnea syndrome and gastroesophageal reflux disease: a meta-analysis. Sleep Breath 23:389–397. https://doi.org/10.1007/s11325-018-1691-x
Hoover DB, Chang Y, Hancock JC (2000) Actions of tachykinins within the heart and their relevance to cardiovascular disease. Jpn J Pharmacol 84:367–373. https://doi.org/10.1254/jjp.84.367
Kapusuz Gencer Z, Intepe YS, Akin I (2014) The correlation of serum levels of leptin, leptin receptor and NO x (NO 2 (−) and NO 3 (−)) in patients with obstructive sleep apnea syndrome. Eur Arch Otorhinolaryngol 271(11):2943–2948. https://doi.org/10.1007/s00405-014-3047-x
Moshage H, Kok B, Huizenga JR, Jansen PL (1995) Nitrite andnitrate determinations in plasma: a critical evaluation. Clin Chem 41:892–896. https://doi.org/10.1016/0009-9120(95)00014-Z
Bredt DS, Snyder SH (1994) Nitric oxide: a physiologic messenger molecule. Annu Rev Biochem 63:175e195–175e120. https://doi.org/10.1016/0190-9622(95)90200-7
Ghasemi A, Zahedi Asl S, Mehrabi Y, Saadat N, Azizi F (2008) Serum nitric oxide metabolite levels in a general healthy population:relation to sex and age. Life Sci 83:326–331. https://doi.org/10.1016/j.lfs.2008.06.010
Bounhoure JP, Galinier M, Didier A (2005) Sleep apnea syndromes and cardiovascular disease. Bull Acad Natl Med 189(3):445–492. https://doi.org/10.1370/afm.278
Lavie L (2009) Oxidative stress--a unifying paradigm in obstructive sleep apnea and comorbidities. Prog Cardiovasc Dis 51(4):303–312. https://doi.org/10.1016/j.pcad.2008.08.003
Kato M, Roberts TP (2000) Impairment of endothelium dependent vasodilation of resistance vessels in patients with obstructive sleep apnea. Circulation 102:2607–2610. https://doi.org/10.1161/01.CIR.102.21.2607
Canino B, Hopps E, Calandrino V (2015) Nitric oxide metabolites and erythrocyte deformability in a group of subjects with obstructive sleep apnea syndrome. Clin Hemorheol Microcirc 59(1):45–52. https://doi.org/10.3233/CH-141815
Stewart LA, Clarke M, Rovers M (2015) Preferred reporting items for systematic review and meta-analyses of individual participant data: the PRISMA-IPD statement. Jama 313(16):1657–1665. https://doi.org/10.1001/jama.2015.3656
Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25(9):603e605–603e605. https://doi.org/10.1007/s10654-010-9491-z
Schulz R, Schmidt D, Blum A (2000) Decreased plasma levels of nitric oxide derivatives in obstructive sleep apnoea: response to CPAP therapy. Thorax 55(12):1046–1051. https://doi.org/10.1136/thorax.55.12.1046
Ip MS, Lam B, Chan LY (2000) Circulating nitric oxide is suppressed in obstructive sleep apnea and is reversed by nasal continuous positive airway pressure. Am J Respir Crit Care Med 162(6):2166–2171. https://doi.org/10.1164/ajrccm.162.6.2002126
Noda A, Nakata S, Koike Y (2007) Continuous positive airway pressure improves daytime baroreflex sensitivity and nitric oxide production in patients with moderate to severe obstructive sleep apnea syndrome. Hypertens Res Off J Jpn Soc Hypertens 30(8):669–676. https://doi.org/10.1291/hypres.30.669
Ozkan Y, Firat H, Simşek B (2008) Circulating nitric oxide (NO), asymmetric dimethylarginine (ADMA), homocysteine, and oxidative status in obstructive sleep apnea-hypopnea syndrome (OSAHS). Sleep Breath 12(2):149–154. https://doi.org/10.1007/s11325-007-0148-4
Yüksel M, Okur HK, Pelin Z (2014) Arginase activity and nitric oxide levels in patients with obstructive sleep apnea syndrom. Clinics 69(4):247–252. https://doi.org/10.6061/clinics/2014(04)05
Ortaç Ersoy E, Fırat H, Akaydın S (2014) Association of obstructive sleep apnea with homocystein, nitric oxide and total antioxidant capacity levels in patients with or without coronary artery disease. Tuberkuloz Ve Toraks 62(3):207–214. https://doi.org/10.5578/tt.7845
Haight JSJ, Djupesland PG (2003) Nitric oxide (NO) and obstructive sleep apnea (OSA). Sleep Breath 7(2):53–62. https://doi.org/10.1055/s-2003-40663
Ben MD, Fabiani M, Loffredo L (2012) Oxidative stress mediated arterial dysfunction in patients with obstructive sleep apnoea and the effect of continuous positive airway pressure treatment. Bmc Pulm Med 12(1):1–8. https://doi.org/10.1186/1471-2466-12-36
Teramoto S, Kume H, Matsuse T (2003) Oxygen administration improves the serum level of nitric oxide metabolites in patients with obstructive sleep apnea syndrome. Sleep Med 4(5):403–407. https://doi.org/10.1016/S1389-9457(03)00102-3
Wei C, Dong-Yu L, Jing-Lin C (2011) The study of nocturnal hypoxia and serum VEGF and NO levels in patients with obstructive sleep apnea hypopnea syndrome. J Clin Pulm Med 16(02):203–204
Franco CM, Lima AM, Jr AL (2012) Obstructive sleep apnea severity correlates with cellular and plasma oxidative stress parameters and affective symptoms. J Mol Neurosci Mn 47(2):300–310. https://doi.org/10.1007/s12031-012-9738-0
Arcaro G, Zamboni M, Rossi L, Turcato E, Covi G, Armellini F, Bosello O, Lechi A (1999) Body fat distribution predicts the degree of endothelial dysfunction in uncomplicated obesity. Int J Obes Relat Metab Disord 23(9):936–942. https://doi.org/10.1038/sj.ijo.0801022
Matagreenwood E, Chen DB (2008) Racial differences in nitric oxide-dependent vasorelaxation. Reprod Sci 15(1):9–25. https://doi.org/10.1177/1933719107312160
Acknowledgments
This work is supported by grants from The National Natural Science Foundation of China (81600801).
Author information
Authors and Affiliations
Contributions
W.Z.H., T.Y., and N.X. designed and analyzed the research study; W.Z.H. and S.H.Y. wrote and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, ZH., Tang, Y., Niu, X. et al. The role of nitric oxide (NO) levels in patients with obstructive sleep apnea-hypopnea syndrome: a meta-analysis. Sleep Breath 25, 9–16 (2021). https://doi.org/10.1007/s11325-020-02095-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-020-02095-0