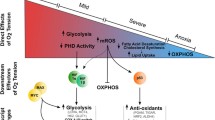
Abstract
Cells sense oxygen availability using not only the absolute value for cellular oxygen in regard to its energetic and metabolic functions, but also the gradient from the cell surface to the lowest levels in the mitochondria. Signals are used for regulatory purposes locally as well as in the generation of cellular, tissue, and humoral remodeling. Lowered oxygen availability (hypoxia) is theoretically important in the consideration of pharmacology because (1) hypoxia can alter cellular function and thereby the therapeutic effectiveness of the agent, (2) therapeutic agents may potentiate or protect against hypoxia-induced pathology, (3) hypoxic conditions may potentiate or mitigate drug-induced toxicity, (4) hypoxia may alter drug metabolism and thereby therapeutic effectiveness, and (5) therapeutic agents might alter the relative coupling of blood flow and energy metabolism in an organ. The prototypic biochemical effect of hypoxia is related to its known role as a cofactor in a number of enzymatic reactions, e.g., oxidases and oxygenases, which are affected independently from the bioenergetic effect of low oxygen on energetic functions. The cytochrome P-450 family of enzymes is another example. Here, there is a direct effect of oxygen availability on the conformation of the enzyme, thereby altering the metabolism of drug substrates. Indirectly, the NADH/NAD+ ratio is increased with 10% inspired oxygen, leading not only to reduced oxidation of ethanol but also to reduction of azo- and nitro-compounds to amines and disulfides to sulfhydryls. With chronic hypoxia, many of these processes are reversed, suggesting that hypoxia induces the drug-metabolizing systems. Support for this comes from observations that hypoxia can induce the hypoxic inducible factors which in turn alters transcription and function of some but not all cytochrome P-450 isoforms. Hypoxia is identified as a cofactor in cancer expression and metastatic potential. Thus, the effects of hypoxia play an important role in pharmacology, and the signaling pathways that are affected by hypoxia could become new targets for novel therapy or avenues for prevention.


Similar content being viewed by others
References
Koch LG, Britton SL (2008) Aerobic metabolism underlies complexity and capacity. J Physiol 586:83–95
Britton SL, Koch LG (2005) Animal models of complex diseases: an initial strategy. IUBMB Life 57:631–638
Budd GE (2008) The earliest fossil record of the animals and its significance. Philos Trans R Soc Lond B Biol Sci 363:1425–1434
Hochachka PW, Land SC, Buck LT (1997) Oxygen sensing and signal transduction in metabolic defense against hypoxia: lessons from vertebrate facultative anaerobes. Comp Biochem Physiol A Physiol 118:23–29
Webster WS, Abela D (2007) The effect of hypoxia in development. Birth Defects Res C Embryo Today 81:215–228
Koch LG, Britton SL (2007) Evolution, atmospheric oxygen, and complex disease. Physiol Genomics 30:205–208
Hochachka PW (1998) Mechanisms and evolution of hypoxia-tolerance in humans. J Exp Biol 201:1243–1254
Baldwin JE, Krebs H (1981) The evolution of metabolic cycles. Nature 291:381–382
Karlin KD (1993) Metalloenzymes, structural motifs, and inorganic models. Science 261:701–708
Dowling DK, Simmons LW (2009) Reactive oxygen species as universal constraints in life-history evolution. Proc Biol Sci 276:1737–1745
Beilman GJ, Cerra FB (1996) The future. Monitoring cellular energetics. Crit Care Clin 12:1031–1042
Duke T (1999) Dysoxia and lactate. Arch Dis Child 81:343–350
Caille V, Squara P (2006) Oxygen uptake-to-delivery relationship: a way to assess adequate flow. Crit Care 10(Suppl 3):4
Levy RJ, Deutschman CS (2007) Cytochrome c oxidase dysfunction in sepsis. Crit Care Med 35:S468–475
Kunze K (1969) Significance of oxygen pressure field measurements in human muscle, with special reference on PO2 micro-needle electrodes. Progr Respir Res 3:153–157
Van Den Brenk HA, Jamieson D (1962) Potentiation by anaesthetics of brain damage due to breathing high-pressure oxygen in mammals. Nature 194:777–778
Gnaiger E, Lassnig B, Kuznetsov A, Rieger G, Margreiter R (1998) Mitochondrial oxygen affinity, respiratory flux control and excess capacity of cytochrome c oxidase. J Exp Biol 201:1129–1139
Gnaiger E, Steinlechner-Maran R, Mendez G, Eberl T, Margreiter R (1995) Control of mitochondrial and cellular respiration by oxygen. J Bioenerg Biomembr 27:583–596
Garofalo F, Pellegrino D, Amelio D, Tota B (2009) The Antarctic hemoglobinless icefish, fifty five years later: a unique cardiocirculatory interplay of disaptation and phenotypic plasticity. Comp Biochem Physiol A Mol Integr Physiol 154:10–28
Gonzalez NC, Howlett RA, Henderson KK, Koch LG, Britton SL, Wagner HE, Favret F, Wagner PD (2006) Systemic oxygen transport in rats artificially selected for running endurance. Respir Physiol Neurobiol 151:141–150
Zuurbier CJ, van Iterson M, Ince C (1999) Functional heterogeneity of oxygen supply-consumption ratio in the heart. Cardiovasc Res 44:488–497
Prabhakar NR (2006) O2 sensing at the mammalian carotid body: why multiple O2 sensors and multiple transmitters? Exp Physiol 91:17–23
Liss B, Roeper J (2001) Molecular physiology of neuronal K-ATP channels (review). Mol Membr Biol 18:117–127
Taburet AM, Tollier C, Richard C (1990) The effect of respiratory disorders on clinical pharmacokinetic variables. Clin Pharmacokinet 19:462–490
Richer M, Lam YW (1993) Hypoxia, arterial pH and theophylline disposition. Clin Pharmacokinet 25:283–299
Burke PV, Poyton RO (1998) Structure/function of oxygen-regulated isoforms in cytochrome c oxidase. J Exp Biol 201:1163–1175
Lahiri S, Antosiewicz J, Pokorski M (2007) A common oxygen sensor regulates the sensory discharge and glomus cell HIF-1alpha in the rat carotid body. J Physiol Pharmacol 58(Suppl 5):327–333
Acker T, Acker H (2004) Cellular oxygen sensing need in CNS function: physiological and pathological implications. J Exp Biol 207:3171–3188
Mironov V, Hritz MA, LaManna JC, Hudetz AG, Harik SI (1994) Architectural alterations in rat cerebral microvessels after hypobaric hypoxia. Brain Res 660:73–80
Lahiri S, Roy A, Li J, Baby SM, Mokashi A, Di Giulio C (2004) Role of Fe2+ in oxygen sensing in the carotid body. Adv Exp Med Biol 551:59–64
Jamieson D, van den Brenk HA (1965) Electrode size and tissue pO2 measurement in rats exposed to air or high pressure oxygen. J Appl Physiol 20:514–518
Cramer T, Yamanishi Y, Clausen BE, Forster I, Pawlinski R, Mackman N, Haase VH, Jaenisch R, Corr M, Nizet V, Firestein GS, Gerber HP, Ferrara N, Johnson RS (2003) HIF-1alpha is essential for myeloid cell-mediated inflammation. Cell 112:645–657
Korzeniewski B (1998) Is it possible to predict any properties of oxidative phosphorylation in a theoretical way? Mol Cell Biochem 184:345–358
Keevil T, Mason HS (1978) Molecular oxygen in biological oxidations—an overview. Meth Enzymol 52:3–40
Jones DP, Aw TY, Shan X (1989) Drug metabolism and toxicity during hypoxia. Drug Metab Rev 2–4:247–260
Feiters MC (2001) Mimicking biological electron transfer and oxygen activation involving iron and copper proteins: a bio(in)organic supramolecular approach. Met Ions Biol Syst 38:461–655
Sun H, Scott DO (2010) Structure-based drug metabolism predictions for drug design. Chem Biol Drug Des 75:3–17
Schumacker PT, Chandel N, Agusti AG (1993) Oxygen conformance of cellular respiration in hepatocytes. Am J Physiol 265:L395–402
Subramanian RM, Chandel N, Budinger GR, Schumacker PT (2007) Hypoxic conformance of metabolism in primary rat hepatocytes: a model of hepatic hibernation. Hepatology 45:455–464
Heerlein K, Schulze A, Hotz L, Bartsch P, Mairbaurl H (2005) Hypoxia decreases cellular ATP demand and inhibits mitochondrial respiration of a549 cells. Am J Respir Cell Mol Biol 32:44–51
Hsu T, Adereth Y, Kose N, Dammai V (2006) Endocytic function of von Hippel-Lindau tumor suppressor protein regulates surface localization of fibroblast growth factor receptor 1 and cell motility. J Biol Chem 281:12069–12080
Wolin MS, Ahmad M, Gao Q, Gupte SA (2007) Cytosolic NAD(P)H regulation of redox signaling and vascular oxygen sensing. Antioxid Redox Signal 9:671–678
Fradette C, Du Souich P (2004) Effect of hypoxia on cytochrome P450 activity and expression. Curr Drug Metab 5:257–271
Savransky V, Reinke C, Jun J, Bevans-Fonti S, Nanayakkara A, Li J, Myers AC, Torbenson MS, Polotsky VY (2009) Chronic intermittent hypoxia and acetaminophen induce synergistic liver injury in mice. Exp Physiol 94:228–239
Smith LL (1987) Mechanism of paraquat toxicity in lung and its relevance to treatment. Hum Toxicol 6:31–36
Demeere JL (1984) Paraquat toxicity. The use of hypoxic ventilation. Acta Anaesthesiol Belg 35:219–230
Riepe MW, Esclaire F, Kasischke K, Schreiber S, Nakase H, Kempski O, Ludolph AC, Dirnagl U, Hugon J (1997) Increased hypoxic tolerance by chemical inhibition of oxidative phosphorylation: “chemical preconditioning”. J Cereb Blood Flow Metab 17:257–264
Huber R, Kasischke K, Ludolph AC, Riepe MW (1999) Increase of cellular hypoxic tolerance by erythromycin and other antibiotics. NeuroReport 10:1543–1546
Riepe MW, Kasischke K, Raupach A (1997) Acetylsalicylic acid increases tolerance against hypoxic and chemical hypoxia. Stroke 28:2006–2011
Burtscher M, Likar R, Nachbauer W, Philadelphy M (1998) Aspirin for prophylaxis against headache at high altitudes: randomised, double blind, placebo controlled trial. Bmj 316:1057–1058
LaManna JC, Chavez JC, Pichiule P (2004) Structural and functional adaptation to hypoxia in the rat brain. J Exp Biol 207:3163–3169
Garnock-Jones KP, Dhillon S, Scott LJ (2009) Armodafinil. CNS Drugs 23:793–803
Fradette C, du Souich P (2003) Hypoxia-inducible factor-1 and activator protein-1 modulate the upregulation of CYP3A6 induced by hypoxia. Br J Pharmacol 140:1146–1154
Semenza GL (2007) Evaluation of HIF-1 inhibitors as anticancer agents. Drug Discov Today 12:853–859
Semenza GL (2007) Oxygen-dependent regulation of mitochondrial respiration by hypoxia-inducible factor 1. Biochem J 405:1–9
Beauvoit B, Rigoulet M (2001) Regulation of cytochrome c oxidase by adenylic nucleotides. Is oxidative phosphorylation feedback regulated by its end-products? IUBMB Life 52:143–152
Maxwell PH (2005) The HIF pathway in cancer. Semin Cell Dev Biol 16:523–530
Tavani A, La Vecchia C (2004) Coffee, decaffeinated coffee, tea and cancer of the colon and rectum: a review of epidemiological studies, 1990-2003. Cancer Causes Control 15:743–757
LaCroix AZ, Mead LA, Liang KY, Thomas CB, Pearson TA (1986) Coffee consumption and the incidence of coronary heart disease. N Engl J Med 315:977–982
Merighi S, Benini A, Mirandola P, Gessi S, Varani K, Simioni C, Leung E, Maclennan S, Baraldi PG, Borea PA (2007) Caffeine inhibits adenosine-induced accumulation of hypoxia-inducible factor-1alpha, vascular endothelial growth factor, and interleukin-8 expression in hypoxic human colon cancer cells. Mol Pharmacol 72:395–406
Purushothaman J, Suryakumar G, Shukla D, Jayamurthy H, Kasiganesan H, Kumar R, Sawhney RC (2010) Modulation of hypoxia-induced pulmonary vascular leakage in rats by seabuckthorn (Hippophae rhamnoides L.). Evid Based Complement Alternat Med (in press)
Brown JM, Wilson WR (2004) Exploiting tumour hypoxia in cancer treatment. Nat Rev Cancer 4:437–447
Reynolds TY, Rockwell S, Glazer PM (1996) Genetic instability induced by the tumor microenvironment. Cancer Res 56:5754–5757
Chen Y, Cairns R, Papandreou I, Koong A, Denko NC (2009) Oxygen consumption can regulate the growth of tumors, a new perspective on the Warburg effect. PLoS ONE 4:e7033
Kimura K, Iwano M, Higgins DF, Yamaguchi Y, Nakatani K, Harada K, Kubo A, Akai Y, Rankin EB, Neilson EG, Haase VH, Saito Y (2008) Stable expression of HIF-1alpha in tubular epithelial cells promotes interstitial fibrosis. Am J Physiol Ren Physiol 295:F1023–1029
Otrock ZK, Hatoum HA, Awada AH, Ishak RS, Shamseddine AI (2009) Hypoxia-inducible factor in cancer angiogenesis: structure, regulation and clinical perspectives. Crit Rev Oncol Hematol 70:93–102
Maxwell PH (2002) Oxygen homeostasis and cancer: insights from a rare disease. Clin Med 2:356–362
Loges S, Roncal C, Carmeliet P (2009) Development of targeted angiogenic medicine. J Thromb Haemost 7:21–33
Sasabe E, Zhou X, Li D, Oku N, Yamamoto T, Osaki T (2007) The involvement of hypoxia-inducible factor-1alpha in the susceptibility to gamma-rays and chemotherapeutic drugs of oral squamous cell carcinoma cells. Int J Cancer 120:277
Bedogni B, Welford SM, Cassarino DS, Nickoloff BJ, Giaccia AJ, Powell MB (2005) The hypoxic microenvironment of the skin contributes to Akt-mediated melanocyte transformation. Cancer Cell 8:443–454
Bedogni B, O’Neill MS, Welford SM, Bouley DM, Giaccia AJ, Denko NC, Powell MB (2004) Topical treatment with inhibitors of the phosphatidylinositol 3′-kinase/Akt and Raf/mitogen-activated protein kinase kinase/extracellular signal-regulated kinase pathways reduces melanoma development in severe combined immunodeficient mice. Cancer Res 64:2552–2560
McCarty MF, Barroso-Aranda J, Contreras F (2010) Practical strategies for suppressing hypoxia-inducible factor activity in cancer therapy. Med Hypotheses 74:789–797
Henk JM, Bishop K, Shepherd SF (2003) Treatment of head and neck cancer with CHART and nimorazole: phase II study. Radiother Oncol 66:65–70
Reddy SB, Williamson SK (2009) Tirapazamine: a novel agent targeting hypoxic tumor cells. Expert Opin Investig Drugs 18:77–87
Albertella MR, Loadman PM, Jones PH, Phillips RM, Rampling R, Burnet N, Alcock C, Anthoney A, Vjaters E, Dunk CR, Harris PA, Wong A, Lalani AS, Twelves CJ (2008) Hypoxia-selective targeting by the bioreductive prodrug AQ4N in patients with solid tumors: results of a phase I study. Clin Cancer Res 14:1096–1104
Sitkovsky MV (2009) T regulatory cells: hypoxia-adenosinergic suppression and re-direction of the immune response. Trends Immunol 30:102–108
Sitkovsky MV, Kjaergaard J, Lukashev D, Ohta A (2008) Hypoxia-adenosinergic immunosuppression: tumor protection by T regulatory cells and cancerous tissue hypoxia. Clin Cancer Res 14:5947–5952
Chen L, Waxman DJ (2002) Cytochrome P450 gene-directed enzyme prodrug therapy (GDEPT) for cancer. Curr Pharm Des 8:1405–1416
McDonough MA, McNeill LA, Tilliet M, Papamicael CA, Chen QY, Banerji B, Hewitson KS, Schofield CJ (2005) Selective inhibition of factor inhibiting hypoxia-inducible factor. J Am Chem Soc 127:7680–7681
Asikainen TM, Ahmad A, Schneider BK, Ho WB, Arend M, Brenner M, Gunzler V, White CW (2005) Stimulation of HIF-1alpha, HIF-2alpha, and VEGF by prolyl 4-hydroxylase inhibition in human lung endothelial and epithelial cells. Free Radic Biol Med 38:1002–1013
Zaman K, Ryu H, Hall D, O’Donovan K, Lin KI, Miller MP, Marquis JC, Baraban JM, Semenza GL, Ratan RR (1999) Protection from oxidative stress-induced apoptosis in cortical neuronal cultures by iron chelators is associated with enhanced DNA binding of hypoxia-inducible factor-1 and ATF-1/CREB and increased expression of glycolytic enzymes, p21(waf1/cip1), and erythropoietin. J Neurosci 19:9821–9830
Siddiq A, Ayoub IA, Chavez JC, Aminova L, Shah S, LaManna JC, Patton SM, Connor JR, Cherny RA, Volitakis I, Bush AI, Langsetmo I, Seeley T, Gunzler V, Ratan RR (2005) Hypoxia-inducible factor prolyl 4-hydroxylase inhibition. A target for neuroprotection in the central nervous system. J Biol Chem 280:41732–41743
Raff H, Tzankoff SP, Fitzgerald RS (1982) Chemoreceptor involvement in cortisol responses to hypoxia in ventilated dogs. J Appl Physiol 52:1092–1096
Raff H, Shinsako J, Dallman MF (1984) Renin and ACTH responses to hypercapnia and hypoxia after chronic carotid chemodenervation. Am J Physiol 247:R412–417
Zinkernagel AS, Johnson RS, Nizet V (2007) Hypoxia inducible factor (HIF) function in innate immunity and infection. J Mol Med 85:1339–1346
Semenza GL, Prabhakar NR (2007) HIF-1-dependent respiratory, cardiovascular, and redox responses to chronic intermittent hypoxia. Antioxid Redox Signal 9:1391–1396
Fraisl P, Aragones J, Carmeliet P (2009) Inhibition of oxygen sensors as a therapeutic strategy for ischaemic and inflammatory disease. Nat Rev Drug Discov 8:139–152
Myllyharju J (2008) Prolyl 4-hydroxylases, key enzymes in the synthesis of collagens and regulation of the response to hypoxia, and their roles as treatment targets. Ann Med 40:402–417
Acknowledgements
We thank Nikolaus Netzer MD for his suggestion to review this topic for the conference. We thank the conference members and the Division of Pulmonary, Critical Care, and Sleep Medicine at University Hospitals of Cleveland, for comments and suggestions on the topic and the presentation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Donovan, L., Welford, S.M., Haaga, J. et al. Hypoxia—implications for pharmaceutical developments. Sleep Breath 14, 291–298 (2010). https://doi.org/10.1007/s11325-010-0368-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-010-0368-x