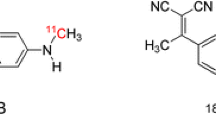
Abstract
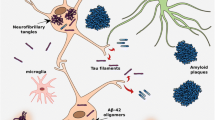
Evolving scientific evidence has begun to point towards hyperphosphorylated tau as a major neurotoxic component in the pathophysiological development of many major neurodegenerative conditions. In response to a need for accurate and reliable diagnosis and disease monitoring in clinical and trial settings, there has been great effort put into the development of tau radiotracers. While first-generation and second-generation radiotracers have provided a basis for assessing tau, concerns of inadequate specificity and selectivity have continued to motivate further study of these radiotracers and the development of novel radiopharmaceuticals. Given the prospective scientific and clinical value of a valid tau radiotracer, the molecular neuroimaging community must be aware of the most recent developments in the realm of tau radiotracer development. This brief review article will critically overview the most established tau radiotracers and, most importantly, concentrate on the progress of more recently developed tau radiotracers.




Similar content being viewed by others
References
Iqbal K, Liu F, Gong C-X (2016) Tau and neurodegenerative disease: the story so far. Nat Rev Neurol 12:15–27. https://doi.org/10.1038/nrneurol.2015.225
Buée L, Bussière T, Buée-Scherrer V, Delacourte A, Hof PR (2000) Tau protein isoforms, phosphorylation and role in neurodegenerative disorders. Brain Res Rev 33:95–130
Agorogiannis EI, Agorogiannis GI, Papadimitriou A, Hadjigeorgiou GM (2004) Protein misfolding in neurodegenerative diseases. Neuropathol Appl Neurobiol 30:215–224
Ayubcha C, Revheim M-E, Newberg A, Moghbel M, Rojulpote C, Werner TJ, Alavi A (2020) A critical review of radiotracers in the positron emission tomography imaging of traumatic brain injury: FDG, tau, and amyloid imaging in mild traumatic brain injury and chronic traumatic encephalopathy. Eur J Nucl Med Mol Imaging 48:623–641. https://doi.org/10.1007/s00259-020-04926-4
Leuzy A, Chiotis K, Lemoine L, Gillberg PG, Almkvist O, Rodriguez-Vieitez E, Nordberg A (2019) Tau PET imaging in neurodegenerative tauopathies-still a challenge. Mol Psychiatry 24:1112–1134. https://doi.org/10.1038/s41380-018-0342-8
Small GW, Kepe V, Ercoli LM, Siddarth P, Bookheimer SY, Miller KJ, Lavretsky H, Burggren AC, Cole GM, Vinters HV, Thompson PM, Huang SC, Satyamurthy N, Phelps ME, Barrio JR (2006) PET of Brain amyloid and tau in mild cognitive impairment. N Engl J Med 355:2652–2663. https://doi.org/10.1056/NEJMoa054625
Okamura N, Harada R, Ishiki A, Kikuchi A, Nakamura T, Kudo Y (2018) The development and validation of tau PET tracers: current status and future directions. Clin Transl Imaging 6:305–316. https://doi.org/10.1007/s40336-018-0290-y
Kepe V, Bordelon Y, Boxer A, Huang SC, Liu J, Thiede FC, Mazziotta JC, Mendez MF, Donoghue N, Small GW, Barrio JR (2013) PET imaging of neuropathology in tauopathies: progressive supranuclear palsy. J Alzheimers Dis 36:145–153. https://doi.org/10.3233/JAD-130032
Barrio JR, Small GW, Wong K-P, Huang SC, Liu J, Merrill DA, Giza CC, Fitzsimmons RP, Omalu B, Bailes J, Kepe V (2015) In vivo characterization of chronic traumatic encephalopathy using [F-18]FDDNP PET brain imaging. Proc Natl Acad Sci U S A 112:E2039–E2047. https://doi.org/10.1073/pnas.1409952112
Ossenkoppele R, Tolboom N, Foster-Dingley JC, Adriaanse SF, Boellaard R, Yaqub M, Windhorst AD, Barkhof F, Lammertsma AA, Scheltens P, van der Flier WM, van Berckel BNM (2012) Longitudinal imaging of Alzheimer pathology using [11C]PIB, [18F]FDDNP and [18F]FDG PET. Eur J Nucl Med Mol Imaging 39:990–1000. https://doi.org/10.1007/s00259-012-2102-3
Jonasson M, Wall A, Chiotis K, Saint-Aubert L, Wilking H, Sprycha M, Borg B, Thibblin A, Eriksson J, Sorensen J, Antoni G, Nordberg A, Lubberink M (2016) Tracer kinetic analysis of (S)-18F-THK5117 as a PET tracer for assessing tau pathology. J Nucl Med 57:574–581. https://doi.org/10.2967/jnumed.115.158519
Harada R, Okamura N, Furumoto S, Furukawa K, Ishiki A, Tomita N, Hiraoka K, Watanuki S, Shidahara M, Miyake M, Ishikawa Y, Matsuda R, Inami A, Yoshikawa T, Tago T, Funaki Y, Iwata R, Tashiro M, Yanai K, Arai H, Kudo Y (2015) [18 F] THK-5117 PET for assessing neurofibrillary pathology in Alzheimer’s disease. Eur J Nucl Med Mol Imaging 42:1052–1061
Chiotis K, Saint-Aubert L, Savitcheva I, Jelic V, Andersen P, Jonasson M, Eriksson J, Lubberink M, Almkvist O, Wall A, Antoni G, Nordberg A (2016) Imaging in-vivo tau pathology in Alzheimer’s disease with THK5317 PET in a multimodal paradigm. Eur J Nucl Med Mol Imaging 43:1686–1699
Mairinger S, Filip T, Sauberer M et al (2020) Plasma pharmacokinetic and metabolism of [18F] THK-5317 are dependent on sex. Nucl Med Biol 84:28–32
Harada R, Okamura N, Furumoto S, Furukawa K, Ishiki A, Tomita N, Tago T, Hiraoka K, Watanuki S, Shidahara M, Miyake M, Ishikawa Y, Matsuda R, Inami A, Yoshikawa T, Funaki Y, Iwata R, Tashiro M, Yanai K, Arai H, Kudo Y (2016) 18F-THK5351: A novel PET radiotracer for imaging neurofibrillary pathology in Alzheimer disease. J Nucl Med 57:208–214. https://doi.org/10.2967/jnumed.115.164848
Harada R, Okamura N, Furumoto S, Tago T, Yanai K, Arai H, Kudo Y (2016) Characteristics of tau and its ligands in PET imaging. Biomolecules 6:7
Harada R, Furumoto S, Tago T, Katsutoshi F, Ishiki A, Tomita N, Iwata R, Tashiro M, Arai H, Yanai K, Kudo Y, Okamura N (2016) Characterization of the radiolabeled metabolite of tau PET tracer 18 F-THK5351. Eur J Nucl Med Mol Imaging 43:2211–2218
Ishiki A, Harada R, Kai H, Sato N, Totsune T, Tomita N, Watanuki S, Hiraoka K, Ishikawa Y, Funaki Y, Iwata R, Furumoto S, Tashiro M, Sasano H, Kitamoto T, Kudo Y, Yanai K, Furukawa K, Okamura N, Arai H (2018) Neuroimaging-pathological correlations of [18 F] THK5351 PET in progressive supranuclear palsy. Acta Neuropathol Commun 6:53
Brendel M, Schönecker S, Höglinger G et al (2018) [18F]-THK5351 PET correlates with topology and symptom severity in progressive supranuclear palsy. Frontiers in Aging. Neuroscience 9:440
Hsu J-L, Chen S-H, Hsiao I-T, Lu CS, Yen TC, Okamura N, Lin KJ, Weng YH (2020) 18 F-THK5351 PET imaging in patients with progressive supranuclear palsy: associations with core domains and diagnostic certainty. Sci Rep 10:19410. https://doi.org/10.1038/s41598-020-76339-0
Betthauser TJ, Lao PJ, Murali D, Barnhart TE, Furumoto S, Okamura N, Stone CK, Johnson SC, Christian BT (2017) In vivo comparison of tau radioligands 18F-THK-5351 and 18F-THK-5317. J Nucl Med 58:996–1002
van Eimeren T, Bischof GN, Drzezga A (2017) Is tau imaging more than just upside-down 18F-FDG imaging? J Nucl Med 58:1357–1359. https://doi.org/10.2967/jnumed.117.190082
Shcherbinin S, Schwarz AJ, Joshi A, Navitsky M, Flitter M, Shankle WR, Devous MD, Mintun MA (2016) Kinetics of the tau PET Tracer 18F-AV-1451 (T807) in subjects with normal cognitive function, mild cognitive impairment, and Alzheimer disease. J Nucl Med 57:1535–1542. https://doi.org/10.2967/jnumed.115.170027
Smith R, Schöll M, Widner H, van Westen D, Svenningsson P, Hägerström D, Ohlsson T, Jögi J, Nilsson C, Hansson O (2017) In vivo retention of 18F-AV-1451 in corticobasal syndrome. Neurology 89:845–853
Lowe VJ, Curran G, Fang P, Liesinger AM, Josephs KA, Parisi JE, Kantarci K, Boeve BF, Pandey MK, Bruinsma T, Knopman DS, Jones DT, Petrucelli L, Cook CN, Graff-Radford NR, Dickson DW, Petersen RC, Jack CR Jr, Murray ME (2016) An autoradiographic evaluation of AV-1451 Tau PET in dementia. Acta Neuropathol Commun 4:58
Marquié M, Normandin MD, Vanderburg CR, Costantino IM, Bien EA, Rycyna LG, Klunk WE, Mathis CA, Ikonomovic MD, Debnath ML, Vasdev N, Dickerson BC, Gomperts SN, Growdon JH, Johnson KA, Frosch MP, Hyman BT, Gómez-Isla T (2015) Validating novel tau positron emission tomography tracer [F-18]-AV-1451 (T807) on postmortem brain tissue. Ann Neurol 78:787–800
Drake LR, Pham JM, Desmond TJ, Mossine AV, Lee SJ, Kilbourn MR, Koeppe RA, Brooks AF, Scott PJH (2019) Identification of AV-1451 as a weak, nonselective inhibitor of monoamine oxidase. ACS Chem Neurosci 10:3839–3846. https://doi.org/10.1021/acschemneuro.9b00326
Ono M, Sahara N, Kumata K, Ji B, Ni R, Koga S, Dickson DW, Trojanowski JQ, Lee VM, Yoshida M, Hozumi I, Yoshiyama Y, van Swieten J, Nordberg A, Suhara T, Zhang MR, Higuchi M (2017) Distinct binding of PET ligands PBB3 and AV-1451 to tau fibril strains in neurodegenerative tauopathies. Brain 140:764–780
Suhara T, Shimada H, Shinotoh H et al (2014) In vivo tau PET imaging using [11C] PBB3 in Alzheimer’s disease and non-Alzheimer’s disease tauopathies. J Nucl Med 55:1824–1824
Lemoine L, Gillberg P-G, Svedberg M et al (2017) Comparative binding properties of the tau PET tracers THK5117, THK5351, PBB3, and T807 in postmortem Alzheimer brains. Alzheimers Res Ther 9:1–13
Walji AM, Hostetler ED, Selnick H, Zeng Z, Miller P, Bennacef I, Salinas C, Connolly B, Gantert L, Holahan M, O’Malley S, Purcell M, Riffel K, Li J, Balsells J, OBrien JA, Melquist S, Soriano A, Zhang X, Ogawa A, Xu S, Joshi E, Della Rocca J, Hess FJ, Schachter J, Hesk D, Schenk D, Struyk A, Babaoglu K, Lohith TG, Wang Y, Yang K, Fu J, Evelhoch JL, Coleman PJ (2016) Discovery of 6-(Fluoro-18 F)-3-(1 H-pyrrolo [2, 3-c] pyridin-1-yl) isoquinolin-5-amine ([18F]-MK-6240): a positron emission tomography (PET) imaging agent for quantification of neurofibrillary tangles (NFTs). J Med Chem 59:4778–4789
Betthauser TJ, Cody KA, Zammit MD, Murali D, Converse AK, Barnhart TE, Stone CK, Rowley HA, Johnson SC, Christian BT (2019) In vivo characterization and quantification of neurofibrillary tau PET radioligand 18F-MK-6240 in humans from Alzheimer disease dementia to young controls. J Nucl Med 60:93–99
Hostetler ED, Walji AM, Zeng Z, Miller P, Bennacef I, Salinas C, Connolly B, Gantert L, Haley H, Holahan M, Purcell M, Riffel K, Lohith TG, Coleman P, Soriano A, Ogawa A, Xu S, Zhang X, Joshi E, Della Rocca J, Hesk D, Schenk DJ, Evelhoch JL (2016) Preclinical characterization of 18F-MK-6240, a promising pet tracer for in vivo quantification of human neurofibrillary tangles. J Nucl Med 57:1599–1606. https://doi.org/10.2967/jnumed.115.171678
Aguero C, Dhaynaut M, Normandin MD, Amaral AC, Guehl NJ, Neelamegam R, Marquie M, Johnson KA, el Fakhri G, Frosch MP, Gomez-Isla T (2019) Autoradiography validation of novel tau PET tracer [F-18]-MK-6240 on human postmortem brain tissue. Acta Neuropathol Commun 7:37
Pascoal TA, Therriault J, Benedet AL, Savard M, Lussier FZ, Chamoun M, Tissot C, Qureshi MNI, Kang MS, Mathotaarachchi S, Stevenson J, Hopewell R, Massarweh G, Soucy JP, Gauthier S, Rosa-Neto P (2020) 18F-MK-6240 PET for early and late detection of neurofibrillary tangles. Brain 143:2818–2830
Honer M, Gobbi L, Knust H, Kuwabara H, Muri D, Koerner M, Valentine H, Dannals RF, Wong DF, Borroni E (2018) Preclinical evaluation of 18F-RO6958948, 11C-RO6931643, and 11C-RO6924963 as novel PET radiotracers for imaging tau aggregates in Alzheimer disease. J Nucl Med 59:675–681
Wong DF, Comley RA, Kuwabara H, Rosenberg PB, Resnick SM, Ostrowitzki S, Vozzi C, Boess F, Oh E, Lyketsos CG, Honer M, Gobbi L, Klein G, George N, Gapasin L, Kitzmiller K, Roberts J, Sevigny J, Nandi A, Brasic J, Mishra C, Thambisetty M, Moghekar A, Mathur A, Albert M, Dannals RF, Borroni E (2018) Characterization of 3 novel tau radiopharmaceuticals, 11C-RO-963, 11C-RO-643, and 18F-RO-948, in healthy controls and in Alzheimer subjects. J Nucl Med 59:1869–1876
Smith R, Schöll M, Leuzy A, Jögi J, Ohlsson T, Strandberg O, Hansson O (2020) Head-to-head comparison of tau positron emission tomography tracers [18 F] flortaucipir and [18 F] RO948. Eur J Nucl Med Mol Imaging 47:342–354
Kuwabara H, Comley RA, Borroni E, Honer M, Kitmiller K, Roberts J, Gapasin L, Mathur A, Klein G, Wong DF (2018) Evaluation of 18F-RO-948 PET for quantitative assessment of tau accumulation in the human brain. J Nucl Med 59:1877–1884
Barret O, Seibyl J, Stephens A et al (2017) Initial clinical pet studies with the novel tau agent 18-F Pi-2620 in Alzheimer’s disease and controls. J Nucl Med 58:630–630
Lois C, Gonzalez I, Johnson KA, Price JC (2019) PET imaging of tau protein targets: a methodology perspective. Brain Imaging Behav 13:333–344. https://doi.org/10.1007/s11682-018-9847-7
Mueller A, Bullich S, Barret O, Madonia J, Berndt M, Papin C, Perrotin A, Koglin N, Kroth H, Pfeifer A, Tamagnan G, Seibyl JP, Marek K, de Santi S, Dinkelborg LM, Stephens AW (2020) Tau PET imaging with 18F-PI-2620 in patients with Alzheimer disease and healthy controls: a first-in-humans study. J Nucl Med 61:911–919
Brendel M, Barthel H, van Eimeren T et al (2020) Assessment of 18F-PI-2620 as a biomarker in progressive supranuclear palsy. JAMA Neurol 77(11):1408-1419. https://doi.org/10.1001/jamaneurol.2020.2526
Bohorquez SS, Marik J, Ogasawara A et al (2019) [18 F] GTP1 (Genentech Tau Probe 1), a radioligand for detecting neurofibrillary tangle tau pathology in Alzheimer’s disease. Eur J Nucl Med Mol Imaging 46:2077–2089
Teng E, Ward M, Manser PT, Sanabria-Bohorquez S, Ray RD, Wildsmith KR, Baker S, Kerchner GA, Weimer RM (2019) Cross-sectional associations between [18F] GTP1 tau PET and cognition in Alzheimer’s disease. Neurobiol Aging 81:138–145
Teng E, Manser PT, Ward M, Sanabria-Bohorquez S, Ray R, Baker SL, Kerchner GA, Weimer R (2018) P4-353: Baseline tau burden measured by [18F] GTP1 imaging is associated with subsequent cognitive decline in prodromal to mild Alzheimer’s disease. Alzheimers Dement 14:P1604–P1605
Shimada H, Ono M, Tagai K, Kubota M, Kitamura S, Takuwa H, Seki C, Kimura Y, Ichise M, Shinotoh H, Takahata K, Yamamoto N, Sano Y, Takado Y, Tempest P, Jang MK, Seibyl J, Barret O, Alagille D, Marek K, Sahara N, Kawamura K, Zhang MR, Suhara T, Higuchi M (2018) P1-125: preclinical and clinical characterization of 18f-Pm-Pbb3, a PET ligand for diverse tau pathologies. Alzheimers Dement 14:P318–P319. https://doi.org/10.1016/j.jalz.2018.06.128
Alberts I, Lu J, Guan Y et al (2020) [18F]-PM-PBB3-PET tau imaging in Alzheimer’s dementia and PSP. Nuklearmedizin 59:V32
Shimada H, Tagai K, Kubota M, Takahata K, Takado Y, Shinotoh H, Yamamoto Y, Sano Y, Seki C, Hirano S, Kimura Y, Ichise M, Ono M, Tempest P, Jang MK, Sahara N, Kawamura K, Zhang MR, Kuwabara S, Hattori N, Suhara T, Higuchi M (2019) P2-382: in vivo tracking of tau pathologies with 18f-Pm-Pbb3 (18f-Apn-1607) PET in ad and diverse non-ad tauopathies. Alzheimers Dement 15:P749–P749. https://doi.org/10.1016/j.jalz.2019.06.2789
Declercq L, Rombouts F, Koole M, Fierens K, Mariën J, Langlois X, Andrés JI, Schmidt M, Macdonald G, Moechars D, Vanduffel W, Tousseyn T, Vandenberghe R, van Laere K, Verbruggen A, Bormans G (2017) Preclinical evaluation of 18F-JNJ64349311, a novel PET tracer for tau imaging. J Nucl Med 58:975–981
Kolb HC, Bottelbergs A, Moechars D, Declercq L, Bormans G, Rombouts F (2017) [P3–316]: pre-clinical characterization of the novel tau PET ligand [18F]-JNJ’067. Alzheimers Dement 13:P1069–P1069
Kolb HC, Moechars D, Rombouts F, Schmidt ME, Szardenings AK, Timmers M, Barret O, Madonia J, Marek K, Sandiego C (2018) P2-372: Imaging Alzheimer’s tau pathology in humans: [18F]-JNJ-067 - a phase 0 exploratory study in healthy volunteers and subjects with probable Alzheimer’s disease. Alzheimers Dement 14:P838–P838
Baker SL, Provost K, Thomas WP et al (2020) Evaluation of F18-JNJ-067 as a tau tracer. ALZ
Blennow K, Chen C, Cicognola C, Wildsmith KR, Manser PT, Bohorquez SMS, Zhang Z, Xie B, Peng J, Hansson O, Kvartsberg H, Portelius E, Zetterberg H, Lashley T, Brinkmalm G, Kerchner GA, Weimer RM, Ye K, Höglund K (2020) Cerebrospinal fluid tau fragment correlates with tau PET: a candidate biomarker for tangle pathology. Brain 143:650–660. https://doi.org/10.1093/brain/awz346
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rigney, G., Ayubcha, C., Werner, T.J. et al. An Update on the State of Tau Radiotracer Development: a Brief Review. Mol Imaging Biol 23, 797–808 (2021). https://doi.org/10.1007/s11307-021-01612-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-021-01612-1