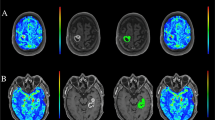
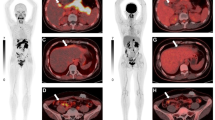
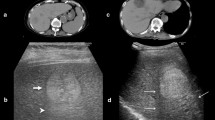
Abstract
Purpose
This study evaluated the use of molecular imaging of fluorescent glucose analog 2-(N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino)-2-deoxyglucose (2-NBDG) as a discriminatory marker for intraoperative tumor border identification in a murine glioma model.
Procedures
2-NBDG was assessed in GL261 and U251 orthotopic tumor-bearing mice. Intraoperative fluorescence of topical and intravenous 2-NBDG in normal and tumor regions was assessed with an operating microscope, handheld confocal laser scanning endomicroscope (CLE), and benchtop confocal laser scanning microscope (LSM). Additionally, 2-NBDG fluorescence in tumors was compared with 5-aminolevulinic acid–induced protoporphyrin IX fluorescence.
Results
Intravenously administered 2-NBDG was detectable in brain tumor and absent in contralateral normal brain parenchyma on wide-field operating microscope imaging. Intraoperative and benchtop CLE showed preferential 2-NBDG accumulation in the cytoplasm of glioma cells (mean [SD] tumor-to-background ratio of 2.76 [0.43]). Topically administered 2-NBDG did not create sufficient tumor-background contrast for wide-field operating microscope imaging or under benchtop LSM (mean [SD] tumor-to-background ratio 1.42 [0.72]). However, topical 2-NBDG did create sufficient contrast to evaluate cellular tissue architecture and differentiate tumor cells from normal brain parenchyma. Protoporphyrin IX imaging resulted in a more specific delineation of gross tumor margins than intravenous or topical 2-NBDG and a significantly higher tumor-to-normal-brain fluorescence intensity ratio.
Conclusion
After intravenous administration, 2-NBDG selectively accumulated in the experimental brain tumors and provided bright contrast under wide-field fluorescence imaging with a clinical-grade operating microscope. Topical 2-NBDG was able to create a sufficient contrast to differentiate tumor from normal brain cells on the basis of visualization of cellular architecture with CLE. 5-Aminolevulinic acid demonstrated superior specificity in outlining tumor margins and significantly higher tumor background contrast. Given the nontoxicity of 2-NBDG, its use as a topical molecular marker for noninvasive in vivo intraoperative microscopy is encouraging and warrants further clinical evaluation.






Similar content being viewed by others
Abbreviations
- 2-NBDG:
-
2-(N-(7-nitrobenz-2-oxa-1, 3-diazol-4-yl) amino)-2-deoxyglucose
- 5 ALA:
-
5-Aminolevulinic Acid
- FDG:
-
18F labeled 2-fluoro-2-deoxy-D-glucose
- CLE:
-
Confocal laser endomicroscope
- IV:
-
Intravenous
- PET:
-
Positron emission tomography
- PpIX:
-
Protoporphyrin IX
- RFP:
-
Red fluorescent protein
References
Carlsson SK, Brothers SP, Wahlestedt C (2014) Emerging treatment strategies for glioblastoma multiforme. EMBO Molecular Medicine 6:1359–1370
Lacroix M, Abi-Said D, Fourney DR, Gokaslan ZL, Shi W, DeMonte F, Lang FF, McCutcheon IE, Hassenbusch SJ, Holland E, Hess K, Michael C, Miller D, Sawaya R (2001) A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 95:190–198
Belykh E, Martirosyan NL, Yagmurlu K, et al. (2016) Intraoperative Fluorescence Imaging for Personalized Brain Tumor Resection: Current State and Future Directions Frontiers in Surgery 3
Lakomkin N, Hadjipanayis CG (2019) The use of spectroscopy handheld tools in brain tumor surgery: current evidence and techniques. Front Surg 6:30
Lakomkin N, Hadjipanayis CG (2018) Fluorescence-guided surgery for high-grade gliomas. J Surg Oncol 118:356–361
Cheng Z, Levi J, Xiong Z, Gheysens O, Keren S, Chen X, Gambhir SS (2006) Near-infrared fluorescent deoxyglucose analogue for tumor optical imaging in cell culture and living mice. Bioconjug Chem 17:662–669
Warburg O, Wind F, Negelein E (1927) The metabolism of tumors in the body. J Gen Physiol 8:519–530
Wahl RL, Cody RL, Hutchins GD, Mudgett EE (1991) Primary and metastatic breast carcinoma: initial clinical evaluation with PET with the radiolabeled glucose analogue 2-[F-18]-fluoro-2-deoxy-D-glucose. Radiology 179:765–770
Avril N, Menzel M, Dose J, Schelling M, Weber W, Jänicke F, Nathrath W, Schwaiger M (2001) Glucose metabolism of breast cancer assessed by 18F-FDG PET: histologic and immunohistochemical tissue analysis. J Nucl Med 42:9–16
Cox BL, Mackie TR, Eliceiri KW (2015) The sweet spot: FDG and other 2-carbon glucose analogs for multi-modal metabolic imaging of tumor metabolism. Am J Nucl Med Mol Imaging 5:1–13
O'Neil RG, Wu L, Mullani N (2005) Uptake of a fluorescent deoxyglucose analog (2-NBDG) in tumor cells. Mol Imaging Biol 7:388–392
Millon SR, Ostrander JH, Brown JQ, Raheja A, Seewaldt VL, Ramanujam N (2011) Uptake of 2-NBDG as a method to monitor therapy response in breast cancer cell lines. Breast Cancer Res Treat 126:55–62
Flavahan WA, Wu Q, Hitomi M, Rahim N, Kim Y, Sloan AE, Weil RJ, Nakano I, Sarkaria JN, Stringer BW, Day BW, Li M, Lathia JD, Rich JN, Hjelmeland AB (2013) Brain tumor initiating cells adapt to restricted nutrition through preferential glucose uptake. Nat Neurosci 16:1373–1382
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682
Yoshioka K, Takahashi H, Homma T, Saito M, Oh KB, Nemoto Y, Matsuoka H (1996) A novel fluorescent derivative of glucose applicable to the assessment of glucose uptake activity of Escherichia coli. Biochim Biophys Acta 1289:5–9
Barros LF, Courjaret R, Jakoby P, Loaiza A, Lohr C, Deitmer JW (2009) Preferential transport and metabolism of glucose in Bergmann glia over Purkinje cells: a multiphoton study of cerebellar slices. Glia 57:962–970
Valdes-Sosa PA, Zhou H, Luby-Phelps K, et al. (2009) Dynamic near-infrared optical imaging of 2-deoxyglucose uptake by intracranial glioma of Athymic mice. PLoS ONE 4
Yamada K, Saito M, Matsuoka H, Inagaki N (2007) A real-time method of imaging glucose uptake in single, living mammalian cells. Nat Protoc 2:753–762
Sheth RA, Josephson L, Mahmood U (2009) Evaluation and clinically relevant applications of a fluorescent imaging analog to fluorodeoxyglucose positron emission tomography. J Biomed Opt 14:064014
La Rocca G, Sabatino G, Menna G et al (2020) 5-Aminolevulinic acid false positives in cerebral Neuro-oncology: not all that is fluorescent is tumor. A case-based update and literature review. World Neurosurgery 137:187–193
Müther M, Stummer W (2019) Ependymal fluorescence in fluorescence-guided resection of malignant glioma: a systematic review. Acta Neurochir 162:365–372
Hebeda KM, Saarnak AE, Olivo M, Sterenborg HJCM, Wolbers JG (1998) 5-Aminolevulinic acid induced endogenous porphyrin fluorescence in 9L and C6 brain tumours and in the normal rat brain. Acta Neurochir 140:503–513
Belykh E, Miller EJ, Hu D, Martirosyan NL, Woolf EC, Scheck AC, Byvaltsev VA, Nakaji P, Nelson LY, Seibel EJ, Preul MC (2018) Scanning fiber endoscope improves detection of 5-aminolevulinic acid-induced protoporphyrin IX fluorescence at the boundary of infiltrative Glioma. World Neurosurg 113:e51–e69
Folaron M, Strawbridge R, Samkoe KS, Filan C, Roberts DW, Davis SC (2018) Elucidating the kinetics of sodium fluorescein for fluorescence-guided surgery of glioma. J Neurosurg 131:724–734
Belykh E, Miller EJ, Patel AA, Yazdanabadi MI, Martirosyan NL, Yağmurlu K, Bozkurt B, Byvaltsev VA, Eschbacher JM, Nakaji P, Preul MC (2018) Diagnostic accuracy of a confocal laser endomicroscope for in vivo differentiation between normal injured and tumor tissue during fluorescein-guided glioma resection: laboratory investigation. World Neurosurg 115:e337–e348
Martirosyan NL, Georges J, Eschbacher JM, Cavalcanti DD, Elhadi AM, Abdelwahab MG, Scheck AC, Nakaji P, Spetzler RF, Preul MC (2014) Potential application of a handheld confocal endomicroscope imaging system using a variety of fluorophores in experimental gliomas and normal brain. Neurosurg Focus 36:E16
Martirosyan NL, Eschbacher JM, Kalani MY et al (2016) Prospective evaluation of the utility of intraoperative confocal laser endomicroscopy in patients with brain neoplasms using fluorescein sodium: experience with 74 cases. Neurosurg Focus 40:E11
Nitin N, Carlson AL, Muldoon T, El-Naggar AK, Gillenwater A, Richards-Kortum R (2009) Molecular imaging of glucose uptake in oral neoplasia following topical application of fluorescently labeled deoxy-glucose. Int J Cancer 124:2634–2642
Thekkek N, Maru DM, Polydorides AD, Bhutani MS, Anandasabapathy S, Richards-Kortum R (2011) Pre-clinical evaluation of fluorescent deoxyglucose as a topical contrast agent for the detection of Barrett’s-associated neoplasia during confocal imaging. Technol Cancer Res Treat 10:431–441
Yokoyama H, Sasaki A, Yoshizawa T, Kijima H, Hakamada K, Yamada K (2016) Imaging hamster model of bile duct cancer in vivo using fluorescent L-glucose derivatives. Hum Cell 29:111–121
Pal R, Villarreal P, Qiu S, Vargas G (2018) In-vivo topical mucosal delivery of a fluorescent deoxy-glucose delineates neoplasia from normal in a preclinical model of oral epithelial neoplasia. Sci Rep 8:9760
Acknowledgments
The authors thank the staff of Neuroscience Publications at Barrow Neurological Institute for assistance with manuscript preparation. We thank Carl Zeiss, AG, Oberkochen, Germany, for providing the LSM 710 confocal laser scanning microscope.
Funding
The Barrow Neurological Foundation and the Newsome Chair in Neurosurgery Research (to MCP). The confocal laser endomicroscope was provided by Zeiss. Zeiss did not have any contribution or effect on the idea, study design, data collection, analysis, or paper preparation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Belykh, E., Jubran, J.H., George, L.L. et al. Molecular Imaging of Glucose Metabolism for Intraoperative Fluorescence Guidance During Glioma Surgery. Mol Imaging Biol 23, 586–596 (2021). https://doi.org/10.1007/s11307-021-01579-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-021-01579-z