Abstract
Purpose
Loss of neuronal synapse function is associated with a number of brain disorders. The [11C]UCB-J positron emission tomography (PET) tracer allows for in vivo examination of synaptic density, as it binds to synaptic vesicle glycoprotein 2A (SV2A) expressed in presynaptic terminals. Here, we characterise [11C]UCB-J imaging in Göttingen minipigs.
Procedures
Using PET imaging, we examined tracer specificity and compared kinetic models. We explored the use of a standard blood curve and centrum semiovale white matter as a reference region. We compared in vivo [11C]UCB-J PET imaging to in vitro autoradiography, Western blotting and real-time quantitative polymerase chain reaction.
Results
The uptake kinetics of [11C]UCB-J could be described using a 1-tissue compartment model and blocking of SV2A availability with levetiracetam showed dose-dependent specific binding. Population-based blood curves resulted in reliable [11C]UCB-J binding estimates, while it was not possible to use centrum semiovale white matter as a non-specific reference region. Brain [11C]UCB-J PET signals correlated well with [3H]UCB-J autoradiography and SV2A protein levels.
Conclusions
[11C]UCB-J PET is a valid in vivo marker of synaptic density in the minipig brain, with binding values close to those reported for humans. Minipig models of disease could be valuable for investigating the efficacy of putative neuroprotective agents for preserving synaptic function in future non-invasive, longitudinal studies.





Similar content being viewed by others
References
Feng G, Xiao F, Lu Y, Huang Z, Yuan J, Xiao Z, Xi Z, Wang X (2009) Down-regulation synaptic vesicle protein 2A in the anterior temporal neocortex of patients with intractable epilepsy. J Mol Neurosci 39(3):354–359
Robinson JL, Molina-Porcel L, Corrada MM, Raible K, Lee EB, Lee VMY, Kawas CH, Trojanowski JQ (2014) Perforant path synaptic loss correlates with cognitive impairment and Alzheimer’s disease in the oldest-old. Brain 137(Pt 9):2578–2587
Bellucci A, Zaltieri M, Navarria L, Grigoletto J, Missale C, Spano PF (2012) From alpha-synuclein to synaptic dysfunctions: new insights into the pathophysiology of Parkinson’s disease. Brain Res 1476:183–202
Chen F, Ardalan M, Elfving B, Wegener G, Madsen TM, Nyengaard JR (2018) Mitochondria are critical for BDNF-mediated synaptic and vascular plasticity of hippocampus following repeated electroconvulsive seizures. Int J Neuropsychopharmacol 21(3):291–304
Nabulsi NB, Mercier J, Holden D, Carre S, Najafzadeh S, Vandergeten MC, Lin SF, Deo A, Price N, Wood M, Lara-Jaime T, Montel F, Laruelle M, Carson RE, Hannestad J, Huang Y (2016) Synthesis and preclinical evaluation of 11C-UCB-J as a PET tracer for imaging the synaptic vesicle glycoprotein 2A in the brain. J Nucl Med 57(5):777–784
Finnema SJ et al (2016) Imaging synaptic density in the living human brain. Sci Transl Med 8(348):348ra96
Finnema SJ, Nabulsi NB, Mercier J, Lin SF, Chen MK, Matuskey D, Gallezot JD, Henry S, Hannestad J, Huang Y, Carson RE (2017) Kinetic evaluation and test-retest reproducibility of [(11)C]UCB-J, a novel radioligand for positron emission tomography imaging of synaptic vesicle glycoprotein 2A in humans. J Cereb Blood Flow Metab 38(11):2041–2052
Chen MK, Mecca AP, Naganawa M, Finnema SJ, Toyonaga T, Lin SF, Najafzadeh S, Ropchan J, Lu Y, McDonald JW, Michalak HR, Nabulsi NB, Arnsten AFT, Huang Y, Carson RE, van Dyck CH (2018) Assessing synaptic density in Alzheimer disease with synaptic vesicle glycoprotein 2A positron emission tomographic imaging. JAMA Neurol 75(10):1215–1224
Bertoglio D, et al (2019) Validation and noninvasive kinetic modeling of [(11)C]UCB-J PET imaging in mice. J Cereb Blood Flow Metab 271678X19864081
Toyonaga T, Smith LM, Finnema SJ, Gallezot JD, Naganawa M, Bini J, Mulnix T, Cai Z, Ropchan J, Huang Y, Strittmatter SM, Carson RE (2019) In vivo synaptic density imaging with (11)C-UCB-J detects treatment effects of saracatinib (AZD0530) in a mouse model of Alzheimer’s disease. J Nucl Med 60:1780–1786
Onwordi EC, Halff EF, Whitehurst T, Mansur A, Cotel MC, Wells L, Creeney H, Bonsall D, Rogdaki M, Shatalina E, Reis Marques T, Rabiner EA, Gunn RN, Natesan S, Vernon AC, Howes OD (2020) Synaptic density marker SV2A is reduced in schizophrenia patients and unaffected by antipsychotics in rats. Nat Commun 11(1):246
Cai Z, Li S, Matuskey D, Nabulsi N, Huang Y (2019) PET imaging of synaptic density: a new tool for investigation of neuropsychiatric diseases. Neurosci Lett 691:44–50
Matuskey D, Tinaz S, Wilcox KC, Naganawa M, Toyonaga T, Dias M, Henry S, Pittman B, Ropchan J, Nabulsi N, Suridjan I, Comley RA, Huang Y, Finnema SJ, Carson RE (2020) Synaptic changes in Parkinson disease assessed with in vivo imaging. Ann Neurol 87(3):329–338
Holmes SE, Scheinost D, Finnema SJ, Naganawa M, Davis MT, DellaGioia N, Nabulsi N, Matuskey D, Angarita GA, Pietrzak RH, Duman RS, Sanacora G, Krystal JH, Carson RE, Esterlis I (2019) Lower synaptic density is associated with depression severity and network alterations. Nat Commun 10(1):1529
Buckley K, Kelly RB (1985) Identification of a transmembrane glycoprotein specific for secretory vesicles of neural and endocrine cells. J Cell Biol 100(4):1284–1294
Orlowski D, Glud AN, Palomero-Gallagher N, Sørensen JCH, Bjarkam CR (2019) Online histological atlas of the Gottingen minipig brain. Heliyon 5(3):e01363
Bjarkam CR, Glud AN, Orlowski D, Sørensen JCH, Palomero-Gallagher N (2017) The telencephalon of the Gottingen minipig, cytoarchitecture and cortical surface anatomy. Brain Struct Funct 222(5):2093–2114
Sorensen JC et al (2011) Development of neuromodulation treatments in a large animal model--do neurosurgeons dream of electric pigs? Prog Brain Res 194:97–103
Lillethorup TP, Glud AN, Alstrup AKO, Noer O, Nielsen EHT, Schacht AC, Landeck N, Kirik D, Orlowski D, Sørensen JCH, Doudet DJ, Landau AM (2018) Longitudinal monoaminergic PET imaging of chronic proteasome inhibition in minipigs. Sci Rep 8(1):15715
Ettrup KS et al (2011) Basic surgical techniques in the Gottingen minipig: intubation, bladder catheterization, femoral vessel catheterization, and transcardial perfusion. J Vis Exp 52
Watanabe H, Andersen F, Simonsen CZ, Evans SM, Gjedde A, Cumming P (2001) MR-based statistical atlas of the Gottingen minipig brain. Neuroimage 14(5):1089–1096
Shiozaki T, Sadato N, Senda M, Ishii K, Tsuchida T, Yonekura Y, Fukuda H, Konishi J (2000) Noninvasive estimation of FDG input function for quantification of cerebral metabolic rate of glucose: optimization and multicenter evaluation. J Nucl Med 41(10):1612–1618
Lillethorup TP, Glud AN, Landeck N, Alstrup AKO, Jakobsen S, Vang K, Doudet DJ, Brooks DJ, Kirik D, Hinz R, Sørensen JC, Landau AM (2018) In vivo quantification of glial activation in minipigs overexpressing human alpha-synuclein. Synapse 72(12):e22060
Thomsen MB, Lillethorup TP, Jakobsen S, Nielsen EH, Simonsen M, Wegener G, Landau AM, Tasker RA (2016) Neonatal domoic acid alters in vivo binding of [11C]yohimbine to alpha2-adrenoceptors in adult rat brain. Psychopharmacology 233:3779–3785
Phan JA, Landau AM, Wong DF, Jakobsen S, Nahimi A, Doudet DJ, Gjedde A (2015) Quantification of [(11)C]yohimbine binding to alpha2 adrenoceptors in rat brain in vivo. J Cereb Blood Flow Metab 35(3):501–511
Gunn RN, Gunn SR, Cunningham VJ (2001) Positron emission tomography compartmental models. J Cereb Blood Flow Metab 21(6):635–652
Cunningham VJ, Rabiner EA, Slifstein M, Laruelle M, Gunn RN (2010) Measuring drug occupancy in the absence of a reference region: the Lassen plot re-visited. J Cereb Blood Flow Metab 30(1):46–50
Lassen NA, Bartenstein PA, Lammertsma AA, Prevett MC, Turton DR, Luthra SK, Osman S, Bloomfield PM, Jones T, Patsalos PN, O'Connell MT, Duncan JS, Andersen JV (1995) Benzodiazepine receptor quantification in vivo in humans using [11C]flumazenil and PET: application of the steady-state principle. J Cereb Blood Flow Metab 15(1):152–165
Bjarkam CR et al (2017) Exposure of the pig CNS for histological analysis: a manual for decapitation, skull opening, and brain removal. J Vis Exp 122
Müller HK, Wegener G, Popoli M, Elfving B (2011) Differential expression of synaptic proteins after chronic restraint stress in rat prefrontal cortex and hippocampus. Brain Res 1385:26–37
Nygard AB, Jørgensen CB, Cirera S, Fredholm M (2007) Selection of reference genes for gene expression studies in pig tissues using SYBR green qPCR. BMC Mol Biol 8:67
du Jardin KG, Müller HK, Sanchez C, Wegener G, Elfving B (2016) A single dose of vortioxetine, but not ketamine or fluoxetine, increases plasticity-related gene expression in the rat frontal cortex. Eur J Pharmacol 786:29–35
Lillethorup TP, Glud AN, Alstrup AKO, Mikkelsen TW, Nielsen EH, Zaer H, Doudet DJ, Brooks DJ, Sørensen JCH, Orlowski D, Landau AM (2018) Nigrostriatal proteasome inhibition impairs dopamine neurotransmission and motor function in minipigs. Exp Neurol 303:142–152
Landau AM, Dyve S, Jakobsen S, Alstrup AKO, Gjedde A, Doudet DJ (2015) Acute vagal nerve stimulation lowers alpha2 adrenoceptor availability: possible mechanism of therapeutic action. Brain Stimul 8(4):702–707
Landau AM, Alstrup AKO, Audrain H, Jakobsen S, Simonsen M, Møller A, Videbech P, Wegener G, Gjedde A, Doudet DJ (2018) Elevated dopamine D1 receptor availability in striatum of Gottingen minipigs after electroconvulsive therapy. J Cereb Blood Flow Metab 38(5):881–887
Logan J, Fowler JS, Volkow ND, Wolf AP, Dewey SL, Schlyer DJ, MacGregor RR, Hitzemann R, Bendriem B, Gatley SJ, Christman DR (1990) Graphical analysis of reversible radioligand binding from time-activity measurements applied to [N-11C-methyl]-(−)-cocaine PET studies in human subjects. J Cereb Blood Flow Metab 10(5):740–747
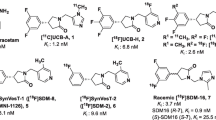
Warnock GI, Aerts J, Bahri MA, Bretin F, Lemaire C, Giacomelli F, Mievis F, Mestdagh N, Buchanan T, Valade A, Mercier J, Wood M, Gillard M, Seret A, Luxen A, Salmon E, Plenevaux A (2014) Evaluation of 18F-UCB-H as a novel PET tracer for synaptic vesicle protein 2A in the brain. J Nucl Med 55(8):1336–1341
Becker G, Warnier C, Serrano ME, Bahri MA, Mercier J, Lemaire C, Salmon E, Luxen A, Plenevaux A (2017) Pharmacokinetic characterization of [(18)F]UCB-H PET radiopharmaceutical in the rat brain. Mol Pharm 14(8):2719–2725
Koole M, van Aalst J, Devrome M, Mertens N, Serdons K, Lacroix B, Mercier J, Sciberras D, Maguire P, van Laere K (2018) Quantifying SV2A density and drug occupancy in the human brain using [(11)C]UCB-J PET imaging and subcortical white matter as reference tissue. Eur J Nucl Med Mol Imaging 46(2):396–406
Diehl KH, Hull R, Morton D, Pfister R, Rabemampianina Y, Smith D, Vidal JM, van de Vorstenbosch C, European Federation of Pharmaceutical Industries Association and European Centre for the Validation of Alternative Methods (2001) A good practice guide to the administration of substances and removal of blood, including routes and volumes. J Appl Toxicol 21(1):15–23
Tian Q, Stepaniants SB, Mao M, Weng L, Feetham MC, Doyle MJ, Yi EC, Dai H, Thorsson V, Eng J, Goodlett D, Berger JP, Gunter B, Linseley PS, Stoughton RB, Aebersold R, Collins SJ, Hanlon WA, Hood LE (2004) Integrated genomic and proteomic analyses of gene expression in mammalian cells. Mol Cell Proteomics 3(10):960–969
Pradet-Balade B, Boulmé F, Beug H, Müllner EW, Garcia-Sanz JA (2001) Translation control: bridging the gap between genomics and proteomics? Trends Biochem Sci 26(4):225–229
Acknowledgements
We are grateful to UCB Pharma for providing precursor for initial studies. We thank the staff at the Aarhus University Hospital PET Centre, the Aarhus University Farm and Birgitte Hviid Mumm from the Translational Neuropsychiatry Unit for assistance.
Funding
This work was supported by Lundbeck Foundation, Parkinsonforeningen, Fonden af 2/7 1984 til Bekæmpelse af Parkinsonsyge and Aarhus University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
All animal experiments were approved by the Danish Animal Experiments Inspectorate, carried out in accordance with the 2010/63/EU directive and reported using the ARRIVE guidelines.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 120 kb)
Rights and permissions
About this article
Cite this article
Thomsen, M.B., Schacht, A.C., Alstrup, A.K.O. et al. Preclinical PET Studies of [11C]UCB-J Binding in Minipig Brain. Mol Imaging Biol 22, 1290–1300 (2020). https://doi.org/10.1007/s11307-020-01506-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-020-01506-8