Abstract
Purpose
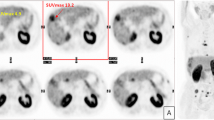
[18F]fluorocholine PET/CT can detect hepatocellular carcinoma (HCC) based on imaging the initial steps of phosphatidylcholine synthesis. To relate the diagnostic performance of [18F]fluorocholine positron emission tomography (PET)/x-ray computed tomography (CT) to the phospholipid composition of liver tumors, radiopathologic correspondence was performed in patients with early-stage liver cancer who had undergone [18F]fluorocholine PET/CT before tumor resection.
Procedures
Tumor and adjacent liver were profiled by liquid chromatography mass spectrometry, quantifying phosphatidylcholine species by mass-to-charge ratio. For clinical-radiopathologic correlation, HCC profiles were reduced to two orthogonal principal component factors (PCF1 and PCF2) accounting for 80 % of total profile variation.
Results
Tissues from 31 HCC patients and 4 intrahepatic cholangiocarcinoma (ICC) patients were analyzed, revealing significantly higher levels of phosphocholine, CDP-choline, and highly saturated phosphatidylcholine species in HCC tumors relative to adjacent liver and ICC tumors. Significant loading values for PCF1 corresponded to phosphatidylcholines containing poly-unsaturated fatty acids while PCF2 corresponded only to highly saturated phosphatidylcholines. Only PCF2 correlated significantly with HCC tumor-to-liver [18F]fluorocholine uptake ratio (ρ = 0.59, p < 0.0005). Sensitivity for all tumors based on an abnormal [18F]fluorocholine uptake ratio was 93 % while sensitivity for HCC based on increased tumor [18F]fluorocholine uptake was 84 %, with lower levels of highly saturated phosphatidylcholines in tumors showing low [18F]fluorocholine uptake.
Conclusion
Most HCC tumors contain high levels of saturated phosphatidylcholines, supporting their dependence on de novo fatty acid metabolism for phospholipid membrane synthesis. While [18F]fluorocholine PET/CT can serve to identify these lipogenic tumors, its imperfect diagnostic sensitivity implies metabolic heterogeneity across HCC and a weaker lipogenic phenotype in some tumors.





Similar content being viewed by others
References
Rahib L, Smith BD, Aizenberg R et al (2014) Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 74:2913–2921
Benson AB 3rd, D'Angelica MI, Abrams TA et al (2014) Hepatobiliary cancers, version 2.2014. J Natl Compr Cancer Netw 12:1152–1182
Lan BY, Kwee SA, Wong LL (2012) Positron emission tomography in hepatobiliary and pancreatic malignancies: a review. Am J Surg 204:232–241
Michel V, Yuan Z, Ramsubir S, Bakovic M (2006) Choline transport for phospholipid synthesis. Exp Biol Med 231:490–504
Kuang Y, Salem N, Tian H et al (2011) Imaging lipid synthesis in hepatocellular carcinoma with [methyl-11C]choline: correlation with in vivo metabolic studies. J Nucl Med 52:98–106
Beyoglu D, Idle JR (2013) The metabolomic window into hepatobiliary disease. J Hepatol 59:842–858
Stubbs CD, Smith AD (1984) The modification of mammalian membrane polyunsaturated fatty acid composition in relation to membrane fluidity and function. Biochim Biophys Acta 779:89–137
Gandola YB, Perez SE, Irene PE et al (2014) Mitogenic effects of phosphatidylcholine nanoparticles on MCF-7 breast cancer cells. Biomed Res Int. doi:10.1155/2014/687037
Guo S, Wang Y, Zhou D, Li Z (2014) Significantly increased monounsaturated lipids relative to polyunsaturated lipids in six types of cancer microenvironment are observed by mass spectrometry imaging. Sci Rpt. doi:10.1038/srep05959
Hilvo M, Denkert C, Lehtinen L et al (2011) Novel theranostic opportunities offered by characterization of altered membrane lipid metabolism in breast cancer progression. Cancer Res 71:3236–3245
Ishikawa S, Tateya I, Hayasaka T et al (2012) Increased expression of phosphatidylcholine (16:0/18:1) and (16:0/18:2) in thyroid papillary cancer. PLoS One 7:e48873
Rysman E, Brusselmans K, Scheys K et al (2010) De novo lipogenesis protects cancer cells from free radicals and chemotherapeutics by promoting membrane lipid saturation. Cancer Res 70:8117–8126
Talbot JN, Fartoux L, Balogova S et al (2010) Detection of hepatocellular carcinoma with PET/CT: a prospective comparison of 18F-fluorocholine and 18F-FDG in patients with cirrhosis or chronic liver disease. J Nucl Med 51:1699–1706
Bansal A, Shuyan W, Hara T et al (2008) Biodisposition and metabolism of [18F]fluorocholine in 9 L glioma cells and 9 L glioma-bearing fisher rats. Eur J Nucl Med Mol Imaging 35:1192–1203
Talbot JN, Gutman F, Fartoux L et al (2006) PET/CT in patients with hepatocellular carcinoma using [18F]fluorocholine: preliminary comparison with [18F]FDG PET/CT. Eur J Nucl Med Mol Imaging 33:1285–1289
Lim J, Dorman E, Cabral C (2003) Automated production of[18F]FECh and [18F]FCH: preparation and use of [18F]fluoroalkane sulfonates and Fluoroalkylation agents [abstract]. J Label Compd Radiopharm 46:S46
Slaets D, De Vos F (2013) Comparison between kinetic modelling and graphical analysis for the quantification of [18F]fluoromethylcholine uptake in mice. Eur J Nucl Med Mol Imaging 3:66
Verwer EE, Oprea-Lager DE, van den Eertwegh AJ et al (2015) Quantification of 18F-fluorocholine kinetics in patients with prostate cancer. J Nucl Med 56:365–371
Takesh M (2013) Kinetic modeling application to 18F-fluoroethylcholine positron emission tomography in patients with primary and recurrent prostate cancer using two-tissue compartmental model. World J Nucl Med 12:101–110
Koc H, Mar MH, Ranasinghe A et al (2002) Quantitation of choline and its metabolites in tissues and foods by liquid chromatography/electrospray ionization-isotope dilution mass spectrometry. Anal Chem 74:4734–4740
Zhao YY, Xiong Y, Curtis JM (2011) Measurement of phospholipids by hydrophilic interaction liquid chromatography coupled to tandem mass spectrometry: the determination of choline containing compounds in foods. J Chromatogr A 1218:5470–5479
Mimmi MC, Finato N, Pizzolato G et al (2013) Absolute quantification of choline-related biomarkers in breast cancer biopsies by liquid chromatography electrospray ionization mass spectrometry. Anal Cell Pathol 36:71–83
Peck B, Schug ZT, Zhang Q et al (2016) Inhibition of fatty acid desaturation is detrimental to cancer cell survival in metabolically compromised environments. Cancer Metab 4:6
Plagemann PG (1969) Choline metabolism and membrane formation in rat hepatoma cells grown in suspension culture. II. Phosphatidylcholine synthesis during growth cycle and fluctuation of mitochondrial density. J Cell Biol 42:766–781
Glunde K, Bhujwalla ZM, Ronen SM (2011) Choline metabolism in malignant transformation. Nature Rev Cancer 11:835–848
Price DT, Coleman RE, Liao RP et al (2002) Comparison of [18F]fluorocholine and [18F]fluorodeoxyglucose for positron emission tomography of androgen dependent and androgen independent prostate cancer. J Urol 168:273–280
Schulze A, Harris AL (2012) How cancer metabolism is tuned for proliferation and vulnerable to disruption. Nature 491:364–373
Santos CR, Schulze A (2012) Lipid metabolism in cancer. FEBS J 279:2610–2623
Salem N, Kuang Y, Corn D et al (2011) [(methyl)1-11C]-acetate metabolism in hepatocellular carcinoma. Mol Imaging Biol 13:140–151
Ho CL, Yu SC, Yeung DW (2003) 11C-acetate PET imaging in hepatocellular carcinoma and other liver masses. J Nucl Med 44:213–221
Li L, Che L, Wang C, Blecha JE et al (2016) [11C]acetate PET imaging is not always associated with increased lipogenesis in hepatocellular carcinoma in mice. Mol Imaging Biol 18:360–367
Deford-Watts LM, Mintz A, Kridel SJ (2013) The potential of 11C-acetate PET for monitoring the fatty acid synthesis pathway in tumors. Curr Pharm Biotechnol 14:300–312
Guo D, Bell EH, Mischel P, Chakravarti A (2014) Targeting SREBP-1-driven lipid metabolism to treat cancer. Curr Pharm Des 20:2619–2626
Kast HR, Nguyen CM, Anisfeld AM et al (2001) CTP: phosphocholine cytidylyltransferase, a new sterol- and SREBP-responsive gene. J Lipid Res 42:1266–1272
Murakami Y, Kubo S, Tamori A et al (2015) Comprehensive analysis of transcriptome and metabolome analysis in intrahepatic cholangiocarcinoma and hepatocellular carcinoma. Sci Rpt 5:16294
Kwee SA, Hernandez B, Chan O, Wong L (2012) Choline kinase alpha and hexokinase-2 protein expression in hepatocellular carcinoma: association with survival. PLoS One 7:e46591
Bansal S, Berk M, Alkhouri N et al (2014) Stearoyl-CoA desaturase plays an important role in proliferation and chemoresistance in human hepatocellular carcinoma. J Surg Res 186:29–38
Acknowledgments
This work was substantially supported by US National Institutes of Health/National Cancer Institute grant R01CA161209-05.
Author Contributions
Conceived and designed experiments: SAK, MMS, YK, AF, LC, and LLW. Performed the experiments: SAK, AF, LC, and KM. Analyzed the data: SAK, MMS, YK, AF, LC, KM, and LLW. Contributed materials/analysis tools: SAK, MMS, YK, AF, LC, and LLW. Writing and final manuscript approval: SAK, MMS, YK, AF, LC, KM, and LLW.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Financial Support
This work was supported by NIH/NCI grant R01CA161209–05.
Electronic supplementary material
ESM 1
(PDF 157 kb)
Rights and permissions
About this article
Cite this article
Kwee, S.A., Sato, M.M., Kuang, Y. et al. [18F]Fluorocholine PET/CT Imaging of Liver Cancer: Radiopathologic Correlation with Tissue Phospholipid Profiling. Mol Imaging Biol 19, 446–455 (2017). https://doi.org/10.1007/s11307-016-1020-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-016-1020-3