Abstract
Purpose
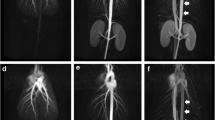
With the goal of facilitating tracer kinetic analysis in small-animal planar fluorescence imaging, an experimental method for characterizing tracer arterial input functions is presented. The proposed method involves exposing the common carotid arteries by surgical dissection, which can then be imaged directly during tracer injection and clearance.
Procedures
Arterial concentration curves of IRDye-700DX-carboxylate, IRDye-800CW-EGF, and IRDye-800CW conjugated to anti-EGFR Affibody are recovered from athymic female mice (n = 12) by directly imaging exposed vessels. Images were acquired with two imaging protocols: a slow-kinetics approach (temporal resolution = 45 s) to recover the arterial curves from two tracers simultaneously, and a fast-kinetics approach (temporal resolution = 500 ms) to characterize the first-pass peak of a single tracer. Arterial input functions obtained by the carotid imaging technique, as well as plasma curves measured by blood sampling were fit with a biexponential pharmacokinetic model.
Results
Pharmacological fast- and slow-phase rate constants recovered with the proposed method were 0.37 ± 0.26 and 0.007 ± 0.001 min−1, respectively, for the IRDye700DX-C. For the IRDye800CW-EGF, the rate constants were 0.11 ± 0.13 and 0.003 ± 0.002 min−1. These rate constants did not differ significantly from those calculated previously by blood sampling, as determined by an F test; however, the between-subject variability was four times lower for arterial curves recovered using the proposed technique, compared with blood sampling.
Conclusions
The proposed technique enables the direct characterization of arterial input functions for kinetic analysis. As this method requires no additional instrumentation, it is immediately deployable in commercially available planar fluorescence imaging systems.





Similar content being viewed by others
References
Frangioni JV (2003) In vivo near-infrared fluorescence imaging. Curr Opin Chem Biol 7:626–634
Gao X, Cui Y, Levenson RM et al (2004) In vivo cancer targeting and imaging with semiconductor quantum dots. Nat Biotechnol 22:969–976
Olson ES, Jiang T, Aguilera TA et al (2010) Activatable cell penetrating peptides linked to nanoparticles as dual probes for in vivo fluorescence and MR imaging of proteases. Proc Natl Acad Sci U S A 107:4311–4316
St Lawrence K, Verdecchia K, Elliott J et al (2013) Kinetic model optimization for characterizing tumour physiology by dynamic contrast-enhanced near-infrared spectroscopy. Phys Med Biol 58:1591–1604
Davis SC, Samkoe KS, Tichauer KM et al (2013) Dynamic dual-tracer MRI-guided fluorescence tomography to quantify receptor density in vivo. Proc Natl Acad Sci U S A 110:9025–9030
Tichauer KM, Samkoe KS, Klubben WS et al (2012) Advantages of a dual-tracer model over reference tissue models for binding potential measurement in tumors. Phys Med Biol 57:6647–6659
Tichauer KM, Samkoe KS, Sexton KJ et al (2011) In vivo quantification of tumor receptor binding potential with dual-reporter molecular imaging. Mol Imaging Biol 14:584–592
Solomon M, White BR, Nothdruft RE et al (2011) Video-rate fluorescence diffuse optical tomography for in vivo sentinel lymph node imaging. Biomed Opt Express 2:3267–3277
Nguyen QT, Tsien RY (2013) Fluorescence-guided surgery with live molecular navigation—a new cutting edge. Nat Rev Cancer 13:653–662
Meier P, Zierler KL (1954) On the theory of the indicator-dilution method for measurement of blood flow and volume. J Appl Physiol 6:731–744
Brix G, Ravesh MS, Zwick S et al (2011) On impulse response functions computed from dynamic contrast-enhanced image data by algebraic deconvolution and compartmental modeling. Phys Med 28:119–128
St Lawrence KS, Lee TY (1998) An adiabatic approximation to the tissue homogeneity model for water exchange in the brain: I. Theoretical derivation. J Cereb Blood Flow Metab 18:1365–1377
Schabel MC (2012) A unified impulse response model for DCE-MRI. Magn Reson Med 68:1632–1646
Cenic A, Nabavi DG, Craen RA et al (1999) Dynamic CT measurement of cerebral blood flow: a validation study. AJNR Am J Neuroradiol 20:63–73
Elliott JT, Diop M, Lee TY, Lawrence KS (2012) Model-independent dynamic constraint to improve the optical reconstruction of regional kinetic parameters. Opt Lett 37:2571–2573
Logan J, Fowler JS, Volkow ND et al (1996) Distribution volume ratios without blood sampling from graphical analysis of PET data. J Cereb Blood Flow Metab 16:834–840
Lammertsma AA, Hume SP (1996) Simplified reference tissue model for PET receptor studies. Neuroimage 4:153–158
Liu JT, Helms MW, Mandella MJ et al (2009) Quantifying cell-surface biomarker expression in thick tissues with ratiometric three-dimensional microscopy. Biophys J 96:2405–2414
Pogue BW, Samkoe KS, Hextrum S et al (2010) Imaging targeted-agent binding in vivo with two probes. J Biomed Opt 15:030513
Samkoe KS, Sexton K, Tichauer KM et al (2012) High vascular delivery of EGF, but low receptor binding rate is observed in AsPC-1 tumors as compared to normal pancreas. Mol Imaging Biol 14:472–479
Johnson RA, Waddelow TA, Caro J et al (1989) Chronic exposure to tumor necrosis factor in vivo preferentially inhibits erythropoiesis in nude mice. Blood 74:130–138
Coleman TF, Li YY (1996) An interior trust region approach for nonlinear minimization subject to bounds. Siam J Optim 6:418–445
Beck JV, Arnold KJ (1977) Parameter estimation in engineering and science. Wiley, New York
Parker GJ, Roberts C, Macdonald A et al (2006) Experimentally-derived functional form for a population-averaged high-temporal-resolution arterial input function for dynamic contrast-enhanced MRI. Magn Reson Med 56:993–1000
LI-COR (2013) Pearl® Impulse Operator’s Manual. LI-COR, Inc., Lincoln
Acknowledgments
This work has been funded by NIH research grants R01CA109558 (BWP and JRG), R01CA156177 (BWP and KSS), U54CA151662 (BWP and KJS), and by a Canadian Institutes of Health Research Postdoctoral Fellowship (JTE).
Conflict of Interest
The authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elliott, J.T., Tichauer, K.M., Samkoe, K.S. et al. Direct Characterization of Arterial Input Functions by Fluorescence Imaging of Exposed Carotid Artery to Facilitate Kinetic Analysis. Mol Imaging Biol 16, 488–494 (2014). https://doi.org/10.1007/s11307-013-0715-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-013-0715-y