Abstract
Purpose
Given that carotid vasa vasorum neovascularization is associated with increased risk for stroke and cardiac events, the present in vivo study was designed to investigate molecular imaging of carotid artery vasa vasorum neovascularization via target-specific contrast-enhanced ultrasound (CEU) micro-imaging.
Procedures
Molecular imaging was performed in male transgenic rats with carotid artery disease and non-transgenic controls using dual endothelin1/VEGFsp receptor (DEspR)-targeted microbubbles (MBD) and the Vevo770 micro-imaging system and CEU imaging software.
Results
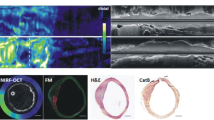
DEspR-targeted CEU-positive imaging exhibited significantly higher contrast intensity signal (CIS)-levels and pre-/post-destruction CIS-differences in seven of 13 transgenic rats, in contrast to significantly lower CIS-levels and differences in control isotype-targeted microbubble (MBC)-CEU imaging (n = 8) and in MBD CEU-imaging of five non-transgenic control rats (P < 0.0001). Ex vivo immunofluorescence analysis demonstrated binding of MBD to DEspR-positive endothelial cells; and association of DEspR-targeted increased contrast intensity signals with DEspR expression in vasa vasorum neovessel and intimal lesions. In vitro analysis demonstrated dose-dependent binding of MBD to DEspR-positive human endothelial cells with increasing %cells bound and number of MBD per cell, in contrast to MBC or non-labeled microbubbles (P < 0.0001).
Conclusion
In vivo DEspR-targeted molecular imaging detected increased DEspR-expression in carotid artery lesions and in expanded vasa vasorum neovessels in transgenic rats with carotid artery disease. Future studies are needed to determine predictive value for stroke or heart disease in this transgenic atherosclerosis rat model and translational applications.





Similar content being viewed by others
References
Ruiz-Opazo N, Hirayama K, Akimoto K (1998) Herrera, V.LM. Molecular characterization of a dual endothelin-1/angiotensin II receptor. Mol Med 4:96–108
Herrera VLM, Ponce LRB, Bagamasbad PD, VanPelt BD, Didishvili T, Ruiz-Opazo N (2005) Embryonic lethality in Dear gene-deficient mice: new player in angiogenesis. Physiol Genomics 23:257–268
Ferrara N, Carver-Moore K, Chen H et al (1996) Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 380:439–442
Carmeliet P, Ferreira V, Breir G et al (1996) Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature 380:435–439
Dunmore BJ, McCarthy MJ, Naylor AR, Brindle NP (2007) Carotid plaque instability and ischemic symptoms are linked to immaturity of microvessels within plaques. J Vasc Surg 45:155–159
Giannoni MF, Vicenzini E, Citone M et al (2009) Contrast carotid ultrasound for the detection of unstable plaques with neoangiogenesis: a pilot study. Eur J Vasc Endovasc Surg. doi:10.10.16/j.ejvs.2008.12.028
Vincenzini E, Giannoni MF, Benedetti-Valentini F, Lenzi GL (2009) Imaging of carotid plaque angiogenesis. Cerbrovasc Dis 27(Suppl 2):48–54
Kaufmann BA, Lindner JR (2007) Molecular imaging with targeted contrast ultrasound. Curr Opin Biotech 18:11–16
Lindner JR (2009) Molecular imaging of cardiovascular disease with contrast-enhanced ultrasonography. Nat Rev Cardiol 6:475–481
Ellegala DB, Leong-Poi H, Carpenter JE et al (2003) Imaging tumor angiogenesis with contrast ultrasound and microbubbles targeted to alpha(v)beta3. Circulation 108:336–341
Leong-Poi H, Christiansen J, Heppner P et al (2005) Assessment of endogenous and therapeutic arteriogenesis by contrast ultrasound molecular imaging of integrin expression. Circulation 111:3248–3254
Willmann JH, Paulmurugan R, Chen K et al (2008) US imaging of tumor angiogenesis with microbubbles targeted to vascular endothelial growth factor receptor type 2 in mice. Radiology 246:508–518
Weller GE, Lu E, Csikari MM et al (2003) Ultrasound imaging of acute cardiac transplant rejection with microbubbles targeted to intercellular adhesion molecule-1. Circulation 108:218–224
Hauff P, Reinhardt M, Briel A, Debus N, Schirner M (2004) Molecular targeting of lymph nodes with l-selectin ligand-specific US contrast agent: a feasibility study in mice and dogs. Radiology 231:667–673
Kaufmann BA, Sanders JM, Davis C et al (2007) Molecular imaging of inflammation in atherosclerosis with targeted ultrasound detection of vascular cell adhesion molecule-1. Circulation 116:276–284
Christiansen JP, Leong-Poi H, Klibanov AL, Kaul S, Lindner JR (2002) Noninvasive imaging of myocardial reperfusion injury using leukocyte-targeted contrast echocardiography. Circulation 105:1764–1767
Villanueva FS, Wagner WR (2008) Ultrasound molecular imaging of cardiovascular disease. Nat Clin Pract Cardiovasc Med 5:S26–S32
Schumann PA, Christiansen JP, Quigley RM et al (2002) Targeted-microbubble binding selectively to GPIIb/IIIa receptors of platelet thrombi. Invest Radiol 37:587–593
Hamilton A, Huang SL, Warninck D et al (2002) Left ventricular thrombus enhancement after intravenous injection of echogenic immunoliposomes: studies in a new experimental model. Circulation 105:2772–2778
Lee S, Carr CL, Belcik TA et al (2008) Contrast-enhanced ultrasound characterization of inflammation and vasa vasoral proliferation caused by mural hemorrhage and platelet deposition. Circulation 118:S644 (Abstract 1074)
Decano JL, Viereck JC, McKee AC, Hamilton JA, Ruiz-Opazo N, Herrera VLM (2009) Early-life sodium exposure unmasks susceptibility to stroke in hyperlipidemic, hypertensive heterozygous Tg25 rats transgenic for human cholesteryl ester transfer protein. Circulation 119:1501–9
Herrera VLM, Tsikoudakis A, Didishvili T et al (2004) Analysis of gender-specific atherosclerosis susceptibility in transgenic[hCETP]25DS rat model. Atherosclerosis 177:9–18
Loveless ME, Li X, Huamani J et al (2008) A method for assessing the microvasculature in a murine tumor model using contrast-enhanced ultrasonography. J Ultrasound Med 27:1699–1709
Herrera VL, Viereck JC, Lopez-Guerra G et al (2009) 11.7 Tesla magnetic resonance microimaging of laryngeal tissue architecture. Laryngoscope 119:2187–94
Blindt R, Vogt F, Lamby D et al (2002) Characterization of differential gene expression in quiescent and invasive human arterial smooth muscle cells. J Vasc Res 39:340–352
Calliada F, Campani R, Bottinelli O, Bozzini A, Sommaruga MG (1998) Ultrasound contrast agents: basic principles. Eur J Radiol 27(Suppl 2):S157–160
Lankford M, Behm CZ, Yeh J, Klibanov AL, Robinson P, Lindner JR (2006) Effect of microbubble ligation to cells on ultrasound signal enhancement: implications for targeted imaging. Invest Radiol 41:721–728
Yasui K, Lee J, Tuziuti T, Towata A, Kozuka T, Iida Y (2009) Influence of the bubble–bubble interaction on destruction of encapsulated microbubbles under ultrasound. J Acoust Soc Am 126:973–982
Chomas JE, Dayton P, Allen J, Morgan K, Ferrara KW (2001) Mechanisms of contrast agent destruction. IEEE Trans Ultrason Ferroelectr Freq Control 48:232–248
Kaufmann BA, Carr CL, Belcik T et al (2010) Molecular imaging of the initial inflammatory response in atherosclerosis. Arterioscler Thromb Vasc Biol 30:54–59
Goertz DE, Frijlink ME, Tempel D et al (2007) Subharmonic contrast intravascular ultrasound for vasa vasorum imaging. Ultrasound Med Biol 33:1859–1872
Kaufmann BA (2009) Ultrasound molecular imaging of atherosclerosis. Cardiovasc Res 83:617–625
Acknowledgments
We acknowledge the Ultrasound Micro-Imaging Core established by the Department of Medicine. This study was supported by NIH RO1 AG32649 to Dr. Herrera.
Disclosures
Pending patent application by Boston University for DEspR.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
ESM 1
(DOC 30 kb)
Rights and permissions
About this article
Cite this article
Decano, J.L., Moran, A.M., Ruiz-Opazo, N. et al. Molecular Imaging of Vasa Vasorum Neovascularization via DEspR-targeted Contrast-enhanced Ultrasound Micro-imaging in Transgenic Atherosclerosis Rat Model. Mol Imaging Biol 13, 1096–1106 (2011). https://doi.org/10.1007/s11307-010-0444-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-010-0444-4