Abstract
Introduction
Our untargeted metabolic data unveiled that Acyl-CoAs undergo dephosphorylation, however little is known about these novel metabolites and their physiology/pathology relevance.
Objectives
To understand the relationship between acyl-CoAs dephosphorylation and energy status as implied in our previous work, we seek to investigate how ischemia (energy depletion) triggers metabolic changes, specifically acyl-CoAs dephosphorylation in this work.
Methods
Rat hearts were isolated and perfused in Langendorff mode for 15 min followed by 0, 5, 15, and 30 minutes of global ischemia. The heart tissues were harvested for metabolic analysis.
Results
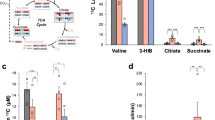
As expected, ATP and phosphocreatine were significantly decreased during ischemia. Most short- and medium-chain acyl-CoAs progressively increased with ischemic time from 0 to 15 min, whereas a 30-minute ischemia did not lead to further change. Unlike other acyl-CoAs, propionyl-CoA accumulated progressively in the hearts that underwent ischemia from 0 to 30 min. Progressive dephosphorylation occurred to all assayed acyl-CoAs and free CoA regardless their level changes during the ischemia.
Conclusion
The present work further confirms that dephosphorylation of acyl-CoAs is an energy-dependent process and how this dephosphorylation is mediated warrants further investigations. It is plausible that dephosphorylation of acyl-CoAs and limited anaplerosis are involved in ischemic injuries to heart. Further investigations are warranted to examine the mechanisms of acyl-CoA dephosphorylation and how the dephosphorylation is possibly involved in ischemic injuries.






Similar content being viewed by others
References
Askenasy, N. (2001). Glycolysis protects sarcolemmal membrane integrity during total ischemia in the rat heart. Basic Research In Cardiology, 96, 612–622.
Broderick, T. L., Paulson, D. J., & Gillis, M. (2004). Effects of propionyl-carnitine on mitochondrial respiration and post-ischaemic cardiac function in the ischaemic underperfused diabetic rat heart. Drugs In R&D, 5, 191–201.
Buja, L. M. (2022). Pathobiology of Myocardial Ischemia and Reperfusion Injury: Models, Modes, Molecular Mechanisms, Modulation and Clinical Applications.Cardiol Rev.
Deng, S., Zhang, G. F., Kasumov, T., Roe, C. R., & Brunengraber, H. (2009). Interrelations between C4 ketogenesis, C5 ketogenesis, and anaplerosis in the perfused rat liver. Journal of Biological Chemistry, 284, 27799–27807.
Feuvray, D., & Leblond, Y. (1984). Metabolism of long chain fatty acids in the normal and pathologic heart: effects of ischemia. Diabete Et Metabolisme, 10, 316–323.
Folch, J., Lees, M., & Sloane Stanley, G. H. (1957). A simple method for the isolation and purification of total lipides from animal tissues. Journal of Biological Chemistry, 226, 497–509.
Jennings, R. B. (2013). Historical perspective on the pathology of myocardial ischemia/reperfusion injury. Circ Res, 113, 428–438.
Kerner, J., Minkler, P. E., Lesnefsky, E. J., & Hoppel, C. L. (2014). Fatty acid chain elongation in palmitate-perfused working rat heart: mitochondrial acetyl-CoA is the source of two-carbon units for chain elongation. Journal Of Biological Chemistry, 289, 10223–10234.
Li, Q., Zhang, S., Berthiaume, J. M., Simons, B., & Zhang, G. F. (2014). Novel approach in LC-MS/MS using MRM to generate a full profile of acyl-CoAs: discovery of acyl-dephospho-CoAs. Journal of Lipid Research, 55, 592–602.
Li, Q., Deng, S., Ibarra, R. A., Anderson, V. E., Brunengraber, H., & Zhang, G. F. (2015). Multiple mass isotopomer tracing of acetyl-CoA metabolism in Langendorff-perfused rat hearts: channeling of acetyl-CoA from pyruvate dehydrogenase to carnitine acetyltransferase. Journal of Biological Chemistry, 290, 8121–8132.
Lindsay, R. T., Dieckmann, S., Krzyzanska, D., Manetta-Jones, D., West, J. A., Castro, C., Griffin, J. L., & Murray, A. J. (2021). beta-hydroxybutyrate accumulates in the rat heart during low-flow ischaemia with implications for functional recovery. Elife 10.
Marquis, N. R., & Fritz, I. B. (1965). The distribution of Carnitine, Acetylcarnitine, and Carnitine Acetyltransferase in Rat Tissues. Journal Of Biological Chemistry, 240, 2193–2196.
Millington, D. S., & Stevens, R. D. (2011). Acylcarnitines: analysis in plasma and whole blood using tandem mass spectrometry. Methods In Molecular Biology, 708, 55–72.
Newman, J. C., & Verdin, E. (2017). beta-hydroxybutyrate: A Signaling Metabolite. Annual Review Of Nutrition, 37, 51–76.
Nowbar, A. N., Gitto, M., Howard, J. P., Francis, D. P., & Al-Lamee, R. (2019). Mortality from ischemic heart disease. Circulation. Cardiovascular Quality And Outcomes, 12, e005375.
Ohsuzu, F., Bessho, M., Yanagida, S., Sakata, N., Takayama, E., & Nakamura, H. (1994). Comparative measurement of myocardial ATP and creatine phosphate by two chemical extraction methods and 31P-NMR spectroscopy. Journal Of Molecular And Cellular Cardiology, 26, 203–210.
Paulson, D. J., Schmidt, M. J., Traxler, J. S., Ramacci, M. T., & Shug, A. L. (1984). Improvement of myocardial function in diabetic rats after treatment with L-carnitine. Metabolism, 33, 358–363.
Ruiz, M., Labarthe, F., Fortier, A., Bouchard, B., Thompson Legault, J., Bolduc, V., Rigal, O., Chen, J., Ducharme, A., Crawford, P. A., Tardif, J. C., & Rosiers, D., C (2017). Circulating acylcarnitine profile in human heart failure: a surrogate of fatty acid metabolic dysregulation in mitochondria and beyond. American Journal Of Physiology Heart And Circulatory Physiology, 313, H768–H781.
Savic, D., Ball, V., Curtis, M. K., Fialho, S., Timm, M. D. L., Hauton, K. N., West, D., Griffin, J., Heather, J., L.C. and, & Tyler, D. J. (2021). L-Carnitine Stimulates In Vivo Carbohydrate Metabolism in the Type 1 Diabetic Heart as Demonstrated by Hyperpolarized MRI. Metabolites 11.
Shah, A. A., Craig, D. M., Sebek, J. K., Haynes, C., Stevens, R. C., Muehlbauer, M. J., Granger, C. B., Hauser, E. R., Newby, L. K., Newgard, C. B., Kraus, W. E., Hughes, G. C., & Shah, S. H. (2012). Metabolic profiles predict adverse events after coronary artery bypass grafting. Journal Of Thoracic And Cardiovascular Surgery, 143, 873–878.
Silva, M. F., Ijlst, L., Allers, P., Jakobs, C., Duran, M., de Almeida, I. T., & Wanders, R. J. (2004). Valproyl-dephosphoCoA: a novel metabolite of valproate formed in vitro in rat liver mitochondria. Drug Metabolism And Disposition, 32, 1304–1310.
Srinivasan, B., Baratashvili, M., van der Zwaag, M., Kanon, B., Colombelli, C., Lambrechts, R. A., Schaap, O., Nollen, E. A., Podgorsek, A., Kosec, G., Petkovic, H., Hayflick, S., Tiranti, V., Reijngoud, D. J., Grzeschik, N. A., & Sibon, O. C. (2015). Extracellular 4’-phosphopantetheine is a source for intracellular coenzyme A synthesis. Nature Chemical Biology, 11, 784–792.
Wang, J. M., Chu, Y., Li, W., Wang, X. Y., Guo, J. H., Yan, L. L., Ma, X. H., Ma, Y. L., Yin, Q. H., & Liu, C. X. (2014). Simultaneous determination of creatine phosphate, creatine and 12 nucleotides in rat heart by LC-MS/MS. Journal Of Chromatography. B, Analytical Technologies In The Biomedical And Life Sciences, 958, 96–101.
Wang, S., Wang, Z., Zhou, L., Shi, X., & Xu, G. (2017). Comprehensive analysis of Short-, Medium-, and long-chain acyl-coenzyme A by Online Two-Dimensional Liquid Chromatography/Mass Spectrometry. Analytical Chemistry, 89, 12902–12908.
Wang, Y., Christopher, B. A., Wilson, K. A., Muoio, D., McGarrah, R. W., Brunengraber, H., & Zhang, G. F. (2018). Propionate-induced changes in cardiac metabolism, notably CoA trapping, are not altered by l-carnitine. American Journal Of Physiology. Endocrinology And Metabolism, 315, E622–E633.
Zhang, G. F., Kombu, R. S., Kasumov, T., Han, Y., Sadhukhan, S., Zhang, J., Sayre, L. M., Ray, D., Gibson, K. M., Anderson, V. A., Tochtrop, G. P., & Brunengraber, H. (2009). Catabolism of 4-hydroxyacids and 4-hydroxynonenal via 4-hydroxy-4-phosphoacyl-CoAs. Journal Of Biological Chemistry, 284, 33521–33534.
Zhang, G. F., Jensen, M. V., Gray, S. M., El, K., Wang, Y., Lu, D., Becker, T. C., Campbell, J. E., & Newgard, C. B. (2021). Reductive TCA cycle metabolism fuels glutamine- and glucose-stimulated insulin secretion. Cell Metab, 33, 804–817e5.
Acknowledgements
This publication was made possible by AHA award 12GRNT12050453 to G.F.Z.
Funding
This study was supported by American Heart Association (Grant No. 12GRNT12050453)
Author information
Authors and Affiliations
Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work and approved it for publication.
Corresponding author
Ethics declarations
Conflict of interest
S.F.P. is currently employed by and owns stock in Merck.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, W., Berthiaume, J.M., Previs, S. et al. Ischemia promotes acyl-CoAs dephosphorylation and propionyl-CoA accumulation. Metabolomics 19, 12 (2023). https://doi.org/10.1007/s11306-023-01975-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-023-01975-2