Abstract
Introduction
Onion (Allium cepa) represents one of the most important horticultural crops and is used as food, spice and medicinal plant almost worldwide. Onion bulbs accumulate a broad range of primary and secondary metabolites which impact nutritional, sensory and technological properties.
Objectives
To complement existing analytical methods targeting individual compound classes this work aimed at the development and validation of an analytical workflow for comprehensive metabolite profiling of onion bulbs.
Method
Metabolite profiling was performed by liquid chromatography coupled with electrospray ionization quadrupole time-of-flight mass spectrometry (LC/ESI-QTOFMS). For annotation of metabolites accurate mass tandem mass spectrometry experiments were carried out.
Results
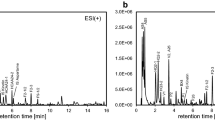
On the basis of LC/ESI-QTOFMS and two chromatographic methods an analytical workflow was developed which facilitates profiling of polar and semi-polar onion metabolites including fructooligosaccharides, proteinogenic amino acids, peptides, S-substituted cysteine conjugates, flavonoids and saponins. To minimize enzymatic conversion of S-alk(en)ylcysteine sulfoxides, a sample preparation and extraction protocol for fresh onions was developed comprising cryohomogenization and a low-temperature quenching step. A total of 123 metabolites were annotated and characterized by chromatographic and tandem mass spectral data. For validation, recovery rates and matrix effects were determined for 15 model compounds. Repeatability and linearity were assessed for more than 80 endogenous metabolites.
Conclusion
As exemplarily demonstrated by comparative metabolic analysis of six onion cultivars the established analytical workflow in combination with targeted and non-targeted data analysis strategies can be successfully applied for comprehensive metabolite profiling of onion bulbs.






Similar content being viewed by others
References
Aoyagi, M., Kamoi, T., Kato, M., Sasako, H., Tsuge, N., & Imai, S. (2011). Structure and bioactivity of thiosulfinates resulting from suppression of lachrymatory factor synthase in onion. Journal of Agricultural and Food Chemistry, 59(20), 10893–10900.
Arnault, I., Christides, J. P., Mandon, N., Haffner, T., Kahane, R., & Auger, J. (2003). High-performance ion-pair chromatography method for simultaneous analysis of alliin, deoxyalliin, allicin and dipeptide precursors in garlic products using multiple mass spectrometry and UV detection. Journal of Chromatography A, 991(1), 69–75.
Bacon, J. R., Moates, G. K., Ng, A., Rhodes, M. J. C., Smith, A. C., & Waldron, K. W. (1999). Quantitative analysis of flavour precursors and pyruvate levels in different tissues and cultivars of onion (Allium cepa). Food Chemistry, 64(2), 257–261.
Bernaert, N., Goetghebeur, L., De Clercq, H., De Loose, M., Daeseleire, E., Van Pamel, E., Van Bockstaele, E., Van Droogenbroeck, B. (2012). Influence of cultivar and harvest time on the amounts of isoalliin and methiin in leek (Allium ampeloprasum var. porrum). Journal of Agricultural and Food Chemistry, 60(44), 10910–10919.
Block, E. (1992). The organosulfur chemistry of the genus allium—Implications for the organic-chemistry of sulfur. Angewandte Chemie—International Edition, 31(9), 1135–1178.
Bonaccorsi, P., Caristi, C., Gargiulli, C., & Leuzzi, U. (2008). Flavonol glucosides in Allium species: A comparative study by means of HPLC-DAD-ESI-MS-MS. Food Chemistry, 107(4), 1668–1673.
Corea, G., Fattorusso, E., Lanzotti, V., Capasso, R., & Izzo, A. A. (2005). Antispasmodic saponins from bulbs of red onion, Allium cepa L. var. Tropea. Journal of Agricultural and Food Chemistry, 53(4), 935–940.
Corzo-Martinez, M., Corzo, N., & Villamiel, M. (2007). Biological properties of onions and garlic. Trends in Food Science & Technology, 18(12), 609–625.
Das, S., Das, S., Bhattacharyab, P., Saha, A., De, B. (2015). Gas chromatography-mass spectrometry based metabolic profiling of onion varieties of india. Current Metabolomics, 3(1), 32–41.
Downes, K., & Terry, L. A. (2010). A new acetonitrile-free mobile phase method for LC-ELSD quantification of fructooligosaccharides in onion (Allium cepa L.). Talanta, 82(1), 118–124.
El-Aasr, M., Fujiwara, Y., Takeya, M., Ikeda, T., Tsukamoto, S., Ono, M., Nakano, D., Okawa, M., Kinjo, J., Yoshimitsu, H., & Nohara, T. (2010). Onionin A from Allium cepa Inhibits Macrophage Activation. Journal of Natural Products, 73(7), 1306–1308.
Fattorusso, E., Iorizzi, M., Lanzotti, V., & Taglialatela-Scafati, O. (2002). Chemical composition of shallot (Allium ascalonicum Hort.). Journal of Agricultural and Food Chemistry, 50(20), 5686–5690.
Fritsch, R. M., & Keusgen, M. (2006). Occurrence and taxonomic significance of cysteine sulphoxides in the genus Allium L. (Alliaceae). Phytochemistry, 67(11), 1127–1135.
Goda, Y., Shibuya, M., & Sankawa, U. (1987). Inhibitors of the arachidonate cascade from Allium chinense and their effect on invitro platelet-aggregation. Chemical and Pharmaceutical Bulletin, 35(7), 2668–2674.
Griffiths, G., Trueman, L., Crowther, T., Thomas, B., & Smith, B. (2002). Onions—a global benefit to health. Phytotherapy Research, 16(7), 603–615.
Hansen, S. L. (2001). Content of free amino acids in onion (Allium cepa L.) as influenced by the stage of development at harvest abd long-term storage. Acta Agriculturae Scandinavica, Section B—Soil & Plant Science, 51, 77–83.
Harrison, S., Xue, H., Lane, G., Villas-Boas, S., & Rasmussen, S. (2012). Linear ion trap MS(n) of enzymatically synthesized 13C-labeled fructans revealing differentiating fragmentation patterns of beta (1–2) and beta (1–6) fructans and providing a tool for oligosaccharide identification in complex mixtures. Analytical Chemistry, 84(3), 1540–1548.
Jones, M. G., Hughes, J., Tregova, A., Milne, J., Tomsett, A. B., & Collin, H. A. (2004). Biosynthesis of the flavour precursors of onion and garlic. Journal of Experimental Botany, 55(404), 1903–1918.
Kubec, R., & Dadakova, E. (2009). Chromatographic methods for determination of S-substituted cysteine derivatives—a comparative study. Journal of Chromatography A, 1216(41), 6957–6963.
Lanzotti, V. (2006). The analysis of onion and garlic. Journal of Chromatography A, 1112(1–2), 3–22.
Lanzotti, V., Romano, A., Lanzuise, S., Bonanomi, G., & Scala, F. (2012). Antifungal saponins from bulbs of white onion, Allium cepa L. Phytochemistry, 74, 133–139.
Lim, T. K. (2015). Allium cepa. In Edible medicinal and non medicinal plants: Modified stems, roots, bulbs (Vol. 9, pp. 124–203). Dordrecht: Springer.
Liu, D. Q., Wu, L. M., Sun, M. J., & MacGregor, P. A. (2007). On-line H/D exchange LC-MS strategy for structural elucidation of pharmaceutical impurities. Journal of Pharmaceutical and Biomedical Analysis, 44(2), 320–329.
Ly, T. N., Hazama, C., Shimoyamada, M., Ando, H., Kato, K., & Yamauchi, R. (2005). Antioxidative compounds from the outer scales of onion. Journal of Agricultural and Food Chemistry, 53(21), 8183–8189.
Matikkala, E. J., & Virtanen, A. I. (1967). On quantitative determination of amino acids and gamma-glutamylpeptides of onion. Acta Chemica Scandinavica, 21(10), 2891–2893.
Nakabayashi, R., Sawada, Y., Yamada, Y., Suzuki, M., Hirai, M. Y., Sakurai, T., & Saito, K. (2013). Combination of liquid chromatography fourier transform ion cyclotron resonance-mass spectrometry with C-13-labeling for chemical assignment of sulfur-containing metabolites in onion bulbs. Analytical Chemistry, 85(3), 1310–1315.
Perez-Gregorio, R. M., Garcia-Falcon, M. S., Simal-Gandara, J., Rodrigues, A. S., & Almeida, D. P. F. (2010). Identification and quantification of flavonoids in traditional cultivars of red and white onions at harvest. Journal of Food Composition and Analysis, 23(6), 592–598.
Ramos, F. A., Takaishi, Y., Shirotori, M., Kawaguchi, Y., Tsuchiya, K., Shibata, H., Higuti, T., Tadokoro, T., & Takeuchi, M. (2006). Antibacterial and antioxidant activities of quercetin oxidation products from yellow onion (Allium cepa) skin. Journal of Agricultural and Food Chemistry, 54(10), 3551–3557.
Schmidt, J. S., Nyberg, N. T., & Staerk, D. (2014). Assessment of constituents in Allium by multivariate data analysis, high-resolution alpha-glucosidase inhibition assay and HPLC-SPE-NMR. Food Chemistry, 161, 192–198.
Shiomi, N., Benkeblia, N., Onodera, S. (2005). The metabolism of the fructooligosaccharides in onion bulbs: a comprehensive review. Journal of Applied Glycoscience, 52(2), 121–127.
Shiomi, N., Onodera, S., Chatterton, N. J., & Harrison, P. A. (1991). Separation of fructooligosaccharide isomers by anion-exchange chromatography. Agricultural and Biological Chemistry, 55(5), 1427–1428.
Slimestad, R., Fossen, T., & Vagen, I. M. (2007). Onions: A source of unique dietary flavonoids. Journal of Agricultural and Food Chemistry, 55(25), 10067–10080.
Smith, C. A., Want, E. J., O’Maille, G., Abagyan, R., & Siuzdak, G. (2006). XCMS: Processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Analytical Chemistry, 78(3), 779–787.
Soininen, T. H., Jukarainen, N., Auriola, S. O. K., Julkunen-Tiitto, R., Karjalainen, R., & Vepsalainen, J. J. (2014). Quantitative metabolite profiling of edible onion species by NMR and HPLC-MS. Food Chemistry, 165, 499–505.
Stoll, A., & Seebeck, E. (1948). Über Alliin, die genuine Muttersubstanz des Knoblauchöls. Helvetica Chimica Acta, 31(1), 189–210.
Stoll, A., & Seebeck, E. (1949). Über den enzymatischen Abbau des Alliins und die Eigenschaften der Alliinase. Helvetica Chimica Acta, 32(1), 197–205.
Verspreet, J., Hansen, A. H., Dornez, E., Courtin, C. M., & Harrison, S. J. (2014). A new high-throughput LC-MS method for the analysis of complex fructan mixtures. Analytical and Bioanalytical Chemistry, 406(19), 4785–4788.
Xiao, H., & Parkin, K. (2006). Isolation and identification of phase II enzyme-inducing agents from nonpolar extracts of green onion (Allium spp.). Journal of Agricultural and Food Chemistry, 54(22), 8417–8424.
Yamazaki, Y., Tokunaga, T., & Okuno, T. (2005). Quantitative determination of eleven flavor precursors (S-alk(en)yl cysteine derivatives) in garlic with an HPLC method. Journal of the Japanese Society for Food Science and Technology-Nippon Shokuhin Kagaku Kogaku Kaishi, 52(4), 160–166.
Ziegert, K., Schütze, W., Schulz, H., Keusgen, M., Gun, F., & Keller, E. R. J. (2006). Efficient determination of cysteine sulphoxides in Allium plants applying new biosensor and HPLC-MS2 methods. Journal of Applied Botany and Food Quality, 80(1), 31–35.
Acknowledgements
We thank Astrid Hansen and Tania Geselle for excellent technical assistance. Roland Buchhorn (JKI) and his team are greatly acknowledged for performing the field trials.
Funding
This work was supported by funding from the state of Niedersachsen to Symrise AG (FKZ W380143435).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Christoph Böttcher, Andrea Krähmer and Hartwig Schulz declare that they have no conflict of interest. Melanie Stürtz and Sabine Widder are employees of Symrise AG holding IP on processing of onions.
Human and animal research information
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Böttcher, C., Krähmer, A., Stürtz, M. et al. Comprehensive metabolite profiling of onion bulbs (Allium cepa) using liquid chromatography coupled with electrospray ionization quadrupole time-of-flight mass spectrometry. Metabolomics 13, 35 (2017). https://doi.org/10.1007/s11306-017-1175-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-017-1175-1