Abstract
Introduction
The maternal body often faces unique physiological challenges in amino acid metabolism due to the continuous requirement of nutrients and substrates for fetal development and additional energy stores for labor and lactation during pregnancy.
Objective
The aims of the present study is to find out the metabolites involved in amino acid metabolism in a large longitudinal healthy pregnant cohort and provide baseline data for future studies of pregnancy and disease from in utero environmental stress factors.
Method
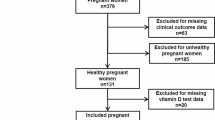
We conducted a UPLC-QTOFMS based-urine metabolomics study to investigate the dynamic amino acid metabolic profiles and pathways of 232 healthy pregnant women in their first, second and third trimesters. After multivariate classification to select the metabolites with the strongest contributions to dynamic alterations in normal pregnancy, we applied the method of standard deviation step (SDSD) down for statistical significance analysis to enhance the value of metabolites in clinical practice.
Results
Kynurenic acid, an endogenous antagonist of N-methyl-d-aspartate receptors, increased significantly in middle pregnancy. l-aspartyl-4-phosphate, a potential marker for lower tolerance against fatigue of human body, decreased significantly in the third trimester. Cysteinylglycine, a pyrolysis product of glutathione, significantly increased in late pregnancy. These findings presented a novel insight into normal pregnancy-related regulation of the generation of excitatory neurotransmitter receptor antagonists, maternal fatigue, oxidative stress and so on.
Conclusion
This normal pregnancy related amino acid metabolic profile as well as the pathways information might be valuable to explore the complex mechanisms of physiological metabolic challenge in amino acid metabolism with the potential capacity to generate a novel hypothesis, which in turn could provide an ideal start for a large-scale epidemiological study of women who subsequently develop diseases, e.g., gestational depression.



Similar content being viewed by others
References
Angueira, A. R., Ludvik, A. E., Reddy, T. E., Wicksteed, B., Lowe, W. L., Jr., & Layden, B. T. (2015). New insights into gestational glucose metabolism: Lessons learned from 21st century approaches. Diabetes, 64(2), 327–334.
Badawy, A. A. (2015). Tryptophan metabolism, disposition and utilization in pregnancy. Bioscience Reports, 35(5), e00261.
Cheng, C. Y., Chou, Y. H., Wang, P., Tsai, J. M., & Liou, S. R. (2014). Survey of trend and factors in perinatal maternal fatigue. Nursing & Health Sciences,. doi:10.1111/nhs.12149.
Carone, D., Loverro, G., Greco, P., Capuano, F., & Selvaggi, L. (1993). Lipid peroxidation products and antioxidant enzymes in red blood cells during normal and diabetic pregnancy. European Journal of Obstetrics, Gynecology, and Reproductive Biology, 51(2), 103–109.
Ciska, E., & Pathak, D. R. (2004). Glucosinolate derivatives in stored fermented cabbage. Journal of Agriculture and Food Chemistry, 52(26), 7938–7943.
Di Cianni, G., Miccoli, R., Volpe, L., Lencioni, C., & Del Prato, S. (2003). Intermediate metabolism in normal pregnancy and in gestational diabetes. Diabetes Metabolism Research and Reviews, 19(4), 259–270.
Diaz, S. O., Barros, A. S., Goodfellow, B. J., Duarte, I. F., Carreira, I. M., Galhano, E., et al. (2013). Following healthy pregnancy by nuclear magnetic resonance (NMR) metabolic profiling of human urine. Journal of Proteome Research, 12(2), 969–979.
Duggleby, S. L., & Jackson, A. A. (2002). Protein, amino acid and nitrogen metabolism during pregnancy: How might the mother meet the needs of her fetus? Current Opinion in Clinical Nutrition and Metabolic Care, 5(5), 503–509.
Dunn, W. B., Broadhurst, D., Begley, P., Zelena, E., Francis-McIntyre, S., Anderson, N., et al. (2011). Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nature Protocols, 6(7), 1060–1083.
Fanos, V., Atzori, L., Makarenko, K., Melis, G. B., & Ferrazzi, E. (2013). Metabolomics application in maternal-fetal medicine. BioMed Research International, 2013, 720514.
Goodpaster, A. M., Romick-Rosendale, L. E., & Kennedy, M. A. (2010). Statistical significance analysis of nuclear magnetic resonance-based metabonomics data. Analytical Biochemistry, 401(1), 134–143.
Glinoer, D. (1999). What happens to the normal thyroid during pregnancy? Thyroid, 9(7), 631–635.
Grün, J. P., Meuris, S., De Nayer, P., & Glinoer, D. (1997). The thyrotrophic role of human chorionic gonadotrophin (hCG) in the early stages of twin (versus single) pregnancies. Clinical Endocrinology (Oxf), 46(6), 719–725.
Hoertel, N., López, S., Peyre, H., Wall, M. M., González-Pinto, A., Limosin, F., et al. (2015). Are symptom features of depression during pregnancy, the postpartum period and outside the peripartum period distinct? Results from a nationally representative sample using item response theory (IRT). Depression and Anxiety, 32(2), 129–140.
Kalhan, S. C., Rossi, K. Q., Gruca, L. L., Super, D. M., & Savin, S. M. (1998). Relation between transamination of branched-chain amino acids and urea synthesis: evidence from human pregnancy. American Journal of Physiology, 275(3 Pt 1), E423–E431.
Luan, H., Meng, N., Liu, P., Feng, Q., Lin, S., Fu, J., et al. (2014). Pregnancy-induced metabolic phenotype variations in maternal plasma. Journal of Proteome Research, 13(3), 1527–1536.
Lindsay, K. L., Hellmuth, C., Uhl, O., Buss, C., Wadhwa, P. D., Koletzko, B., et al. (2015). Longitudinal metabolomic profiling of amino acids and lipids across healthy pregnancy. PLoS One, 10(12), e0145794.
Liang, Q., Xu, W., Hong, Q., Xiao, C., Yang, L., Ma, Z., et al. (2015). Rapid comparison of metabolites in humans and rats of different sexes using untargeted UPLC-TOFMS and an in-house software platform. European Journal of Mass Spectrometry (Chichester, Eng), 21(6), 801–821.
Little, R. E., & Gladen, B. C. (1999). Levels of lipid peroxides in uncomplicated pregnancy: A review of the literature. Reproductive Toxicology, 13(5), 347–352.
Lowe, W. L., Jr., & Karban, J. (2014). Genetics, genomics and metabolomics: New insights into maternal metabolism during pregnancy. Diabetic Medicine, 31(3), 254–262.
Miller, A. L. (2008). The methylation, neurotransmitter, and antioxidant connections between folate and depression. Alternative Medicine Review, 13(3), 216–226.
Meissen, J. K., Yuen, B. T., Kind, T., Riggs, J. W., Barupal, D. K., et al. (2012). Induced pluripotent stem cells show metabolomic differences to embryonic stem cells in polyunsaturated phosphatidylcholines and primary metabolism. PLoS One, 7(10), e46770.
Michnovicz, J. J., & Bradlow, H. L. (1990). Induction of estradiol metabolism by dietary indole-3-carbinol in humans. Journal of the National Cancer Institute, 82(11), 947–949.
Maintz, L., Schwarzer, V., Bieber, T., van der Ven, K., & Novak, N. (2008). Effects of histamine and diamine oxidase activities on pregnancy: A critical review. Human Reproduction Update, 14(5), 485–495.
Nicholson, J. K. (2006). Global systems biology, personalized medicine and molecular epidemiology. Molecular Systems Biology, 2, 52.
Nicholson, J. K., Connelly, J., Lindon, J. C., & Holmes, E. (2002). Metabonomics: A platform for studying drug toxicity and gene function. Nature Reviews Drug Discovery, 1(2), 153–161.
Nicholson, J. K., Lindon, J. C., & Holmes, E. (1999). Metabonomics: Understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica, 29(11), 1181–1189.
Ozkan, Y., Yardim-Akaydin, S., Erdem, A., & Simşek, B. (2012). Variability of total thiol compounds, oxidative and nitrosative stress in uncomplicated pregnant women and nonpregnant women. Archives of Gynecology and Obstetrics, 285(5), 1319–1324.
Pinto, J., Barros, A. S., Domingues, M. R., Goodfellow, B. J., Galhano, E., Pita, C., et al. (2015). Following healthy pregnancy by NMR metabolomics of plasma and correlation to urine. Journal of Proteome Research, 14(2), 1263–1274.
Pershing, M. L., Bortz, D. M., Pocivavsek, A., Fredericks, P. J., Jørgensen, C. V., Vunck, S. A., et al. (2015). Elevated levels of kynurenic acid during gestation produce neurochemical, morphological, and cognitive deficits in adulthood: Implications for schizophrenia. Neuropharmacology, 90, 33–41.
R Core Team. (2015). R: A language and environment for statistical computing. R foundation for statistical computing. Vienna, Austria. https://www.R-project.org/.
Senna, A. A., Zedan, M., el-Salam, G. E., & el-Mashad, A. I. (2008). Study of plasma adrenomedullin level in normal pregnancy and preclampsia. Medscape Journal of Medicine, 10(2), 29.
Shoji, H., & Koletzko, B. (2007). Oxidative stress and antioxidant protection in the perinatal period. Current Opinion in Clinical Nutrition and Metabolic Care, 10(3), 324–328.
Sublette, M. E., Galfalvy, H. C., Fuchs, D., Lapidus, M., Grunebaum, M. F., Oquendo, M. A., et al. (2011). Plasma kynurenine levels are elevated in suicide attempters with major depressive disorder. Brain, Behavior, and Immunity, 25(6), 1272–1278.
Shannon, P., Markiel, A., Ozier, O., Baliga, N. S., Wang, J. T., Ramage, D., et al. (2003). Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Research, 13(11), 2498–2504.
Smith, T. K., Lund, E. K., Clarke, R. G., Bennett, R. N., & Johnson, I. T. (2005). Effects of Brussels sprout juice on the cell cycle and adhesion of human colorectal carcinoma cells (HT29) in vitro. Journal of Agriculture and Food Chemistry, 53(10), 3895–3901.
Ueland, P. M., Mansoor, M. A., Guttormsen, A. B., Müller, F., Aukrust, P., Refsum, H., et al. (1996). Reduced, oxidized and protein-bound forms of homocysteine and other aminothiols in plasma comprise the redox thiol status—A possible element of the extracellular antioxidant defense system. Journal of Nutrition, 126(4 Suppl), 1281S–1284S.
Wang, B., Shi, Z., Weber, G. F., & Kennedy, M. A. (2013). Introduction of a new critical p value correction method for statistical significance analysis of metabonomics data. Analytical and Bioanalytical Chemistry, 405(26), 8419–8429.
Want, E. J., Wilson, I. D., Gika, H., Theodoridis, G., Plumb, R. S., Shockcor, J., et al. (2010). Global metabolic profiling procedures for urine using UPLC-MS. Nature Protocols, 5(6), 1005–1018.
Wang, X., Xie, G., Wang, X., Zhou, M., Yu, H., Lin, Y., et al. (2015). Urinary metabolite profiling offers potential for differentiation of liver-kidney yin deficiency and dampness-heat internal smoldering syndromes in posthepatitis B cirrhosis patients. Evidence Based Complementary Alternative Medicine, 2015, 464969.
Young, K. D., Drevets, W. C., Dantzer, R., Teague, T. K., Bodurka, J., & Savitz, J. (2016). Kynurenine pathway metabolites are associated with hippocampal activity during autobiographical memory recall in patients with depression. Brain, Behavior, and Immunity, S0889–1591(16), 30098-8.
Funding
This work was supported by the National Natural Science Foundation of China (21437002, 81372959, 81402649), the R&D Special Fund for Public Welfare Industry (Environment) (201309048), and the Fundamental Research Funds for the Central Universities, HUST (2016YXZD043).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the Ethics Committees of the Tongji Medical College, Huazhong University of Science and Technology, and the Study Hospital of the Maternal and Child Health Hospital of Wuhan City in China, as well as with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Additional information
Shunqing Xu is the primary corresponding author.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, M., Liang, Q., Li, H. et al. Normal pregnancy-induced amino acid metabolic stress in a longitudinal cohort of pregnant women: novel insights generated from UPLC-QTOFMS-based urine metabolomic study. Metabolomics 12, 131 (2016). https://doi.org/10.1007/s11306-016-1067-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-016-1067-9