Abstract
Introduction
Genital tuberculosis (GTB) in women is one of the common causes of infertility in emerging countries. As an intracellular pathogen, Mycobacterium tuberculosis in the endometrium significantly alters the host metabolism in dormant GTB cases. Nuclear magnetic resonance (NMR) based metabolic profiling has emerged as a useful tool for identification of biomarkers in biological fluids.
Objective
To investigate NMR based serum metabolic profile of dormant GTB women as compared to controls.
Methods
Dormant GTB women (n = 26) and unexplained infertile women (controls; n = 26), healthy proven fertile women undergoing voluntary sterilization (n = 25) and women undergoing recurrent spontaneous miscarriage (RSM) (n = 27) were included in the study. 700 MHz proton NMR spectra of serum collected from these patients were recorded. Multivariate analysis including principal component analysis, partial least squares discriminant analysis and orthogonal projection to latent structure-discriminant analysis was applied to all the spectra. Association of dysregulated serum metabolites with our earlier findings related to altered endometrial tissue metabolites in dormant GTB women was studied using multiple correlation analysis.
Results
This study indicates a clear metabolic differentiation between women with dormant GTB and controls. Metabolites including 3-hydroxybutyrate, succinate, citrate, acetate, l-glutamine, l-lysine, glutamate, l-threonine and 1-methyl histidine were found to be significantly upregulated in serum of women with dormant GTB compared with controls. Pearson’s correlation analysis showed a significant correlation between the expression of endometrial tissue and serum metabolites.
Conclusions
The set of identified metabolites may be considered as candidate markers for the diagnosis of dormant GTB and help clinicians in early therapeutic management.



Similar content being viewed by others
References
Banerjee, P., Dutta, M., Srivastava, S., Joshi, M., Chakravarty, B., & Chaudhury, K. (2014). (1)H NMR serum metabonomics for understanding metabolic dysregulation in women with idiopathic recurrent spontaneous miscarriage during implantation window. Journal of Proteome Research, 13, 3100–3106.
Banerjee, P., Jana, S. K., Pasricha, P., Ghosh, S., Chakravarty, B., & Chaudhury, K. (2013). Proinflammatory cytokines induced altered expression of cyclooxygenase-2 gene results in unreceptive endometrium in women with idiopathic recurrent spontaneous miscarriage. Fertility and Sterility, 99, 179–187.
Bertini, I., Cacciatore, S., Jensen, B. V., Schou, J. V., Johansen, J. S., Kruhøffer, M., et al. (2012). Metabolomic NMR fingerprinting to identify and predict survival of patients with metastatic colorectal cancer. Cancer Research, 72, 356–364.
Bhanu, N. V., Singh, U. B., Chakraborty, M., Suresh, N., Arora, J., Rana, T., et al. (2005). Improved diagnostic value of PCR in the diagnosis of female genital tuberculosis leading to infertility. Journal of Medical Microbiology, 54, 927–931.
Bose, M. (2011). Female genital tract tuberculosis: how long will it elude diagnosis. Indian Journal of Medical Research, 134, 13–14.
Bujak, R., Garcia-Alvarez, A., Ruperez, F. J., Nuno-Ayala, M., Garcia, A., Ruiz-Cabello, J., et al. (2014). Metabolomics reveals metabolite changes in acute pulmonary embolism. Journal of Proteome Research, 13, 805–816.
Cao, M., Zhao, L., Chen, H., Xue, W., & Lin, D. (2012). NMR-based metabolomic analysis of human bladder cancer. Analytical Sciences, 28, 451–456.
Dam, P., Shirazee, H. H., Goswami, S. K., Ghosh, S., Ganesh, A., Chaudhury, K., et al. (2006). Role of latent genital tuberculosis in repeated IVF failure in the Indian clinical setting. Gynecologic and Obstetric Investigation, 61, 223–227.
Das, P., Ahuja, A., & Gupta, S. D. (2008). Incidence, etiopathogenesis and pathological aspects of genitourinary tuberculosis in India: A journey revisited. Indian Journal of Urology, 24, 356–361.
De Graaf, R. A., & Behar, K. L. (2003). Quantitative 1H NMR spectroscopy of blood plasma metabolites. Analytical Chemistry, 75, 2100–2104.
Dutta, M., Joshi, M., Srivastava, S., Lodh, I., Chakravarty, B., & Chaudhury, K. (2012). A metabonomics approach as a means for identification of potential biomarkers for early diagnosis of endometriosis. Molecular BioSystems, 8, 3281–3287.
Fernie, A. R., Trethewey, R. N., Krotzky, A. J., & Willmitzer, L. (2004). Metabolite profiling: From diagnostics to systems biology. Nature Reviews Molecular Cell Biology, 5, 763–769.
Frediani, J. K., Jones, D. P., Tukvadze, N., Uppal, K., Sanikidze, E., Kipiani, M., et al. (2014). Plasma metabolomics in human pulmonary tuberculosis disease: A pilot study. PLoS ONE, 9, e108854.
Garris, D. R., & Smith, C. (1983). Diabetes-associated endometrial disruption in the ketonuric, diabetic Chinese hamster. Gynecologic and Obstetric Investigation, 16, 86–96.
Hedjazi, L., Gauguier, D., Zalloua, P. A., Nicholson, J. K., Dumas, M. E., & Cazier, J. B. (2015). mQTL.NMR: an integrated suite for genetic mapping of quantitative variations of (1)H NMR-based metabolic profiles. Analytical Chemistry, 87, 4377–4384.
Jindal, U. N. (2006). An algorithmic approach to female genital tuberculosis causing infertility. International Journal of Tuberculosis and Lung Disease, 10, 1045–1050.
Jindal, U. N., Bala, Y., Sodhi, S., Verma, S., & Jindal, S. (2010). Female genital tuberculosis: early diagnosis by laparoscopy and endometrial polymerase chain reaction. International Journal of Tuberculosis and Lung Disease, 14, 1629–1634.
Jindal, U. N., Verma, S., & Bala, Y. (2012). Favorable infertility outcomes following antitubercular treatment prescribed on the sole basis of a positive polymerase chain reaction test for endometrial tuberculosis. Human Reproduction, 27, 1368–1374.
Karnovsky, A., Weymouth, T., Hull, T., Tarcea, V. G., Scardoni, G., Laudanna, C., et al. (2012). Metscape 2 bioinformatics tool for the analysis and visualization of metabolomics and gene expression data. Bioinformatics, 28, 373–380.
Kostara, C. E., Papathanasiou, A., Psychogios, N., Cung, M. T., Elisaf, M. S., Goudevenos, J., et al. (2014). NMR-based lipidomic analysis of blood lipoproteins differentiates the progression of coronary heart disease. Journal of Proteome Research, 13, 2585–2598.
Kulshrestha, V., Kriplani, A., Agarwal, N., Singh, U. B., & Rana, T. (2011). Genital tuberculosis among infertile women and fertility outcome after antitubercular therapy. International Journal of Gynecology and Obstetrics, 113, 229–234.
Leroy, J. L., Vanholder, T., Opsomer, G., Van Soom, A., & de Kruif, A. (2006). The in vitro development of bovine oocytes after maturation in glucose and beta-hydroxybutyrate concentrations associated with negative energy balance in dairy cows. Reproduction in Domestic Animals, 41, 119–123.
Lim, T. K., Gough, A., Chin, N. K., & Kumarasinghe, G. (2000). Relationship between estimated pretest probability and accuracy of automated Mycobacterium tuberculosis assay in smear-negative pulmonary tuberculosis. Chest, 118, 641–647.
Liu, F., Yang, M., Wang, X., Yang, S., Gu, J., Zhou, J., et al. (2014). Acetylome analysis reveals diverse functions of lysine acetylation in Mycobacterium tuberculosis. Molecular and Cellular Proteomics, 13, 3352–3366.
Malik, S. (2003). Genital tuberculosis and implantation in assisted reproduction. Reviews in Gynaecological Practice, 3, 160–164.
Nambi, S., Gupta, K., Bhattacharyya, M., Ramakrishnan, P., Ravikumar, V., Siddiqui, N., et al. (2013). Cyclic AMP-dependent protein lysine acylation in mycobacteria regulates fatty acid and propionate metabolism. Journal of Biological Chemistry, 288, 14114–14124.
Paton, N. I., Chua, Y. K., Earnest, A., & Chee, C. B. (2004). Randomized controlled trial of nutritional supplementation in patients with newly diagnosed tuberculosis and wasting. American Journal of Clinical Nutrition, 80, 460–465.
Roy, H., & Roy, S. (2003). Use of polymerase chain reaction for diagnosis of endometrial tuberculosis in high risk subfertile women in an endemic zone. Journal of Obstetrics and Gynecology of India, 53, 260–263.
Rozati, R., Roopa, S., & Naga Rajeshwari, C. (2006). Evaluation of women with infertility and genital tuberculosis. Journal of Obstetrics and Gynecology of India, 56, 423–426.
Shin, J. H., Yang, J. Y., Jeon, B. Y., Yoon, Y. J., Cho, S. N., Kang, Y. H., et al. (2011). (1)H NMR-based metabolomic profiling in mice infected with mycobacterium tuberculosis. Journal of Proteome Research, 10, 2238–2247.
Shrivastava, G., Bajpai, T., Bhatambare, G. S., & Patel, K. B. (2014). Genital tuberculosis: Comparative study of the diagnostic modalities. Journal of Human Reproductive Sciences, 7, 30–33.
Somashekar, B. S., Amin, A. G., Rithner, C. D., Troudt, J., Basaraba, R., Izzo, A., et al. (2011). Metabolic profiling of lung granuloma in Mycobacterium tuberculosis infected guinea pigs: ex vivo 1H magic angle spinning NMR studies. Journal of Proteome Research, 10, 4186–4195.
Somashekar, B. S., Amin, A. G., Tripathi, P., MacKinnon, N., Rithner, C. D., Shanley, C. A., et al. (2012). Metabolomic signatures in guinea pigs infected with epidemic-associated W-Beijing strains of Mycobacterium tuberculosis. Journal of Proteome Research, 11, 4873–4884.
Subramani, E., Jothiramajayam, M., Dutta, M., Chakravorty, D., Joshi, M., Srivastava, S., et al. (2016a). NMR-based metabonomics for understanding the influence of dormant female genital tuberculosis on metabolism of the human endometrium. Human Reproduction, 31, 854–865.
Subramani, E., Madogwe, E., Ray, C. D., Dutta, S. K., Chakravarty, B. N., Bordignon, V., et al. (2016b). Dysregulated LIF and its receptor regulated STAT3 pathway: A possible cause for repeated implantation failure in women with dormant genital tuberculosis? Fertility and Sterility, 105, 1076–1084.
Thangappah, R. B. P., Paramasivan, C. N., & Narayanan, S. (2011). Evaluating PCR, culture and histopathology in the diagnosis of female genital tuberculosis. Indian Journal of Medical Research, 134, 40–46.
Weiner, J., Parida, S. K., Maertzdorf, J., Black, G. F., Repsilber, D., Telaar, A., et al. (2012). Biomarkers of inflammation, immunosuppression and stress with active disease are revealed by metabolomic profiling of tuberculosis patients. PLoS ONE, 7, e40221.
Wevers, R. A., Engelke, U., & Heerschap, A. (1994). High-resolution 1H-NMR spectroscopy of blood plasma for metabolic studies. Clinical Chemistry, 40, 1245–1250.
Wishart, D. S., Knox, C., Guo, A. C., Eisner, R., Young, N., Gautam, B., et al. (2009). HMDB: A knowledgebase for the human metabolome. Nucleic Acids Research, 37, D603–D610.
Zhao, X., Chen, J., Ye, L., & Xum, G. (2014). Serum metabolomics study of the acute graft rejection in human renal transplantation based on liquid chromatography mass spectrometry. Journal of Proteome Research, 13, 2567–2659.
Zhou, A., Ni, J., Xu, Z., Wang, Y., Lu, S., Sha, W., et al. (2013). Application of (1)H NMR spectroscopy-based metabolomics to sera of tuberculosis patients. Journal of Proteome Research, 12, 4642–4649.
Zhu, Y., Guo, Z., Zhang, L., Zhang, Y., Chen, Y., Nan, J., et al. (2015). System-wide assembly of pathways and modules hierarchically reveal metabolic mechanism of cerebral ischemia. Scientific Reports, 5, 17068.
Acknowledgments
The authors are thankful to all the volunteers participated in this study. One of the authors, E Subramani, is acknowledging to IIT Kharagpur and MHRD, Govt. of India for fellowship.
Funding
His work was partly funded by Indian Council of Medical Research, Govt. of India (No.5/7/372/09-RHN; KC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Authors do not have any conflict of interest.
Ethical approval
All procedures performed in the present study involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11306_2016_1042_MOESM1_ESM.tif
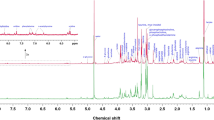
Supplementary material 1 Representative T2-edited CPMG NMR spectra (δ 0.0−9.0) of serum from women with dormant GTB. Metabolites are indicated as follows: 1-TSP, 2-Lipids, 3-L-Isoleucine, 4-L-Leucine, 5-L-Valine, 6-3-Hydroxybutyric acid, 7-Lactate, 8-L-Alanine, 9-Adipic acid, 10-L-Lysine, 11-Acetate, 12-L-Proline, 13-n-Acetyl glycoproteins, 14-L-Glutamine, 15-Acetone, 16-D-Glutamate, 17-Citrate, 18-Creatine, 19-Creatinine, 20-Ornithine, 21-L-Carnitine, 22-D-Glucose, 23-Glycine, 24-L-Histidine, 25-L-Threonine, 26-L-Tyrosine, 27-L-Phenylalanine, 28-Formate, 29-Glycerol, 30-Betaine, 31-Succinate (TIFF 669 kb)
11306_2016_1042_MOESM2_ESM.tif
Supplementary material 2 Two-dimensional scatter plot of (A) PCA, (B) PLS-DA discriminates dormant GTB women, controls, proven fertile women and RSM cases. (C) Permutation test of PLS-DA model for validation (n = 200) (TIFF 1542 kb)
11306_2016_1042_MOESM3_ESM.tif
Supplementary material 3 Color map represents variable importance in projection (VIP) scores corresponding to the OPLS-DA model for dormant GTB and controls (TIFF 4830 kb)
11306_2016_1042_MOESM4_ESM.tif
Supplementary material 4 Scatter plot of OPLS-DA and its corresponding correlation coefficient plots obtained from (A) dormant GTB cases versus proven fertile women, (B) RSM cases versus proven fertile women (C) dormant GTB women versus RSM women (TIFF 2008 kb)
11306_2016_1042_MOESM5_ESM.tif
Supplementary material 5 The color bar represents VIP scores corresponding to OPLS-DA model of (A) dormant GTB cases versus proven fertile women, (B) RSM cases versus proven fertile women (C) dormant GTB women versus RSM women (TIFF 2711 kb)
11306_2016_1042_MOESM6_ESM.tif
Supplementary material 6 Compound network map of dysregulated metabolites. Blue hexagon represents input compound (potential metabolite markers). Pink hexagon indicated the related compounds and chemical reactions (TIFF 22000 kb)
Rights and permissions
About this article
Cite this article
Subramani, E., Dutta, M., Jothiramajayam, M. et al. Identification of serum metabolic markers for diagnosis of women with dormant genital tuberculosis. Metabolomics 12, 99 (2016). https://doi.org/10.1007/s11306-016-1042-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-016-1042-5