Abstract
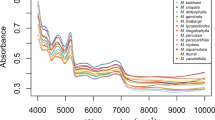
Fourier transform infrared spectroscopy was used to characterise highland and lowland populations of Polygonum minus Huds. grown in different controlled environments. A thermal perturbation technique of two-dimensional correlation infrared spectroscopy (2D-IR) correlation spectra was applied to establish differences between the populations. The absorption peaks at 3,480 cm−1 (hydroxyl group), 2,927 cm−1 (methyl group), 1,623 cm−1 (carbonyl group), and 1,068 cm−1 (C–O group) were particularly powerful in separating the populations. These peaks, which indicate the presence of carbohydrate, terpenes, amide and flavonoids were more intense for the highland populations than lowland populations, and increased in environments with a higher temperature. Wavenumbers (1,634, 669 cm−1) and (1,634, 1,555 cm−1) in the 2D-IR correlation spectra provided fingerprint signals to differentiate plants grown at different temperatures. This study demonstrates that IR fingerprinting, which combines mid-IR spectra and 2D-IR correlation spectra, can directly discriminate different populations of P. minus and the effects of temperature.





Similar content being viewed by others
References
Allwood, J. W., Ellis, D. I., & Goodacre, R. (2008). Metabolomic technologies and their application to the study of plants and plant–host interactions: A review. Physiologia Plantarum, 132, 117–135.
Baharum, S. N., Bunawan, H., Ghani, M. A., Mustapha, W. A. W., & Noor, N. M. (2010). Analysis of the chemical composition of the essential oil of Polygonum minus Huds. using two-dimensional gas chromatography-time-of-flight mass spectrometry (GC-TOF MS). Molecules, 15, 7006–7015.
Beekes, M., Lasch, P., & Naumann, D. (2007). Analytical applications of Fourier transform-infrared (FT-IR) spectroscopy in microbiology and prion research. Veterinary Microbiology, 123, 305–319.
Bunawan, H., Chee, Y. C., Md-Zain, B. M., Baharum, S. N., & Noor, N. M. (2011a). Molecular Systematics of Polygonum minus Huds. based on ITS Sequences. International Journal of Molecular Sciences, 12, 7626–7634.
Bunawan, H., Talip, N., & Noor, N. M. (2011b). Foliar anatomy and micromorphology of Polygonum minus Huds. and their taxonomic implications. Australian Journal of Crop Science, 5(2), 123–127.
Dixon, R. A., & Paiva, N. L. (1995). Stress-induced phenylpropanoid metabolism. Plant Cell, 7, 1085–1097.
Gianfagna, T. J., Carter, C. D., & Sacalis, J. N. (1992). Temperature and photoperiod influence trichome density and sesquiterpene content of Lycopersicon hirsutum f. hirsutum. Plant Physiology, 100, 1403–1405.
Gomez-Romero, M., Segura-Carretero, A., & Fernandez-Gutierrez, (2010). Metabolite profiling and quantification of phenolic compounds in methanol extracts of tomato fruit. Phytochemistry, 71, 1848–1864.
Hayashi, H. (2001). Plant Temperature Stress. doi:10.1038/npg.els.0001320.
Huda-Faujan, N., Noriham, A., Norrakiah, A. S., & Babji, A. S. (2007). Antioxidative activities of water extracts of some Malaysian herbs. ASEAN Food Journal, 14(1), 61–68.
Ikeda, T., Kanaya, S., Kobayashi, A., Yonetani, T., & Fukusaki, E. (2007). Prediction of Japanese green tea ranking by Fourier transform near-infrared reflectance spectroscopy. Journal of Agricultural and Food Chemistry, 55, 9908–9912.
Janas, K. M., Cvikrova, M., Palagiewicz, A., & Eder, J. (2000). Alterations in phenylpropanoid content in soybean roots during low temperature acclimation. Plant Physiology and Biochemistry, 38, 587–593.
Jie, Z., Xiaodong, J., Tianlai, L., & Zaiqiang, Y. (2012). Effect of moderately-high temperature stress on photosynthesis and carbohydrate metabolism in tomato (Lycopersico esculentum L.) leaves. African Journal of Agricultural Research, 7(3), 487–492.
Kell, D. B. (2004). Metabolomics and systems biology: making sense of the soup. Current Opinion in Microbiology, 7, 296–307.
Kemsley, E. K., Belton, P. S., McCann, M. C., Ttofis, S., Wilson, R. H., & Delgadillo, I. (1994). A rapid method for the authentication of vegetable matter using Fourier transform infrared spectroscopy. Food Control, 5, 241–243.
Lafta, A. M., & Lorenzen, J. H. (1995). Effect of high temperature on plant growth and carbohydrate metabolism in potato. Plant Physiology, 109(2), 637–643.
Li, Y. M., Sun, S. Q., Zhou, Q., et al. (2004). Identification of American ginseng from different regions using FT-IR and two-dimensional correlation IR spectroscopy. Vibrational Spectroscopy, 36, 227–232.
Lyons, J. M. (1973). Chilling injury in plants. Annual Review of Plant Physiology, 24, 445–466.
Martz, F. O., Peltola, R., Julkunen-Tiitto, R., Fontanay, S., & Stark, S. (2009). Effect of latitude and altitude on the terpenoid and soluble phenolic composition of Juniper (juniperus communis) needles and evaluation of their antibacterial activity in the Boreal zone. Journal of Agricultural and Food Chemistry, 57, 9575–9584.
Miguel, A. R., Maria, M. R., Rosa, C., Nicolas, C., Juan, M. R., & Luis, R. (2007). Sucrolytic activities in cherry tomato fruits in relation to temperature and solar radiation. Scientia Horticulturae, 113, 244–249.
Naghdi-Badi, H., Dastpak, H. A., & Ziai, S. A. (2004). A Review of Psyllium Plant (Plantago ovata Forsk. and Plantago psyllium L.). Journal of Medicinal Plants, 3, 1–13.
Oliver, S. G., Winson, M. K., Kell, D. B., & Baganz, F. (1998). Systematic functional analysis of the yeast genome. Trends in Biotechnology, 16, 373–378.
Oquist, G. (1983). Effects of low-temperature on photosynthesis. Plant Cell and Environment, 6, 281–300.
Pavia, D. L., Lampman, G. M., & Kriz, G. S. (2001). Introduction to spectroscopy (3rd ed.). USA: Thomson Learning.
Ridley, H. N. (1967). The Flora of the Malay Peninsula. Ashford: L. Reeve & Co.
Rosenfeld, H. J., Aaby, K., & Lea, P. (2002). Influence of temperature and plant density on sensory quality and volatile terpenoids of carrot (Daucus carota L.) root. Journal of the Science of Food and Agriculture, 82(12), 1384–1390.
Roslan, N. D., Yusop, J. M., Baharum, S. N., et al. (2012). flavonoid biosynthesis genes putatively identified in the aromatic plant Polygonum minus via expressed sequences tag (EST) analysis. International Journal of Molecular Sciences, 13, 2692–2706.
Said, S. A., Fernandez, C., Greff, S., et al. (2011). Inter-population variability of terpenoid composition in leaves of Pistacia lentiscus L. from Algeria: A chemoecological approach. Molecules, 16(3), 2646–2657.
Sharkey, T. D., & Singsaas, E. L. (1995). Why plants emit isoprene. Nature, 374, 769.
Singsaas, E. L., Lerdau, M., Winter, K., & Sharkey, T. D. (1997). Isoprene increases thermotolerance of isoprene-emitting leaves. Plant Physiology, 115, 1413–1420.
Sun, S. Q., Zhou, Q., & Chen, J. B. (2011). Infrared spectroscopy for complex mixtures: application in food and traditional medicine. Beijing: Chemical Industry Press.
Suzuki, M., Kusano, M., Takahashi, H., et al. (2010). Rice-Arabidopsis FOX line screening with FT-NIR-based fingerprinting for GC-TOF/MS-based metabolite profiling. Metabolomics, 6(1), 137–145. doi:10.1007/s11306-009-0182-2.
Velikova, V., & Loreto, F. (2005). On the relationship between isoprene emission and thermotolerance in Phragmites australis leaves exposed to high temperatures and during the recovery from a heat stress. Plant, Cell and Environment, 28, 318–327.
Vokou, D., Kokkini, S., & Bessiere, J. M. (1993). Geographic-variation of Greek oregano (Origanum Vulgare ssp. Hirtum) essential oils. Biochemical Systematics and Ecology, 21, 287–295.
Wei, Y. M., Wang, L. H., Cao, F. L., Wei, S. Q., & Liang, Y. D. (2010). Variation and cluster analysis on leaf characters from different provenance sources of Polygonum multiflorum Thunb. Agricultural Science and Technology, 11, 94–98.
Wilks, P. (2006). NIR versus Mid-IR: How to choose. Spectroscopy, 21(4), 43–48.
Xu, C., Wang, Y., Chen, J., et al. (2013). Infrared macro-fingerprint analysis-through-separation for holographic chemical characterization of herbal medicine. Journal of Pharmaceutical and Biomedical Analysis, 74, 298–307.
Yaacob, K. B. (1987). Kesom oil: A natural source of aliphatic aldehydes. Perfumer and Flavorist, 12, 27–30.
Zhang, Z. X., Liu, P., Kang, H. J., Liao, C. C., Chen, Z. L., & Xu, G. D. (2008). A study of the diversity of different geographical populations of Emmenopterys henryi using FTIR based on principal component analysis and cluster analysis. Spectroscopy Spectral Analysis, 28(9), 2081–2086.
Acknowledgments
This research is supported by the Genomics and Molecular Biology Initiative of the Malaysia Genome Institute, Ministry of Science, Technology and Innovation (07-05-MGI-GMB 004), Research University Grant under the Arus Perdana (UKM-AP-BPB-14-2009) and Fundamental Research Grant Scheme (UKM-RB-06-FRGS0102-2009). The authors would like to thank the reviewers and Prof. Dr. Michael Burrell from University of Sheffield for their constructive comments throughout the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khairudin, K., Sukiran, N.A., Goh, HH. et al. Direct discrimination of different plant populations and study on temperature effects by Fourier transform infrared spectroscopy. Metabolomics 10, 203–211 (2014). https://doi.org/10.1007/s11306-013-0570-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11306-013-0570-5