Abstract
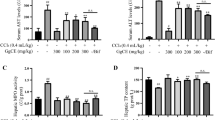
Diammonium glycyrrhizinate (DG), a constitutent of Glycyrrhiza uralensis, has a protective effect on hepatic injury, hepatisis and cirrhosis. To date, the mechanism has been poorly understood, especially at the metabolic level. A metabolomic profiling study was performed to characterize the carbon tetrachloride (CCl4) induced global metabolic alteration and the protective effects of DG in Sprague-Dawley rats. Urinary and hepatic tissue metabolic profiling revealed that CCl4 perturbed the amino acid metabolism (alanine, glycine, leucine), tricarboxylic acid cycle (citrate), lipid metabolism (unsaturated fatty acids) and gut microbiota related metabolites. Our results also indicated that DG was able to attenuate CCl4 perturbed metabolic pathways and ameliorated biochemical markers of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and Total cholesterol (TCHO). This global metabolomic approach also revealed full metabolic recovery takes longer than apparent and conventional histological and biochemical markers.






Similar content being viewed by others
Abbreviations
- DG:
-
Diammonium glycyrrhizinate
- NC:
-
Normal control
- TCHO:
-
Total cholesterol
- AST:
-
Aspartate aminotransferase
- ALT:
-
Alanine aminotransferase
- MDA:
-
Malondiadehyde
- SOD:
-
Superoxide dismutase
- GSH-px:
-
Glutathion peroxidase
- ECF:
-
Ethylchloroformate
- PCA:
-
Principal component analysis
- PLS-DA:
-
Partial least squares discriminant analysis
- PC:
-
Principal component
- GSH:
-
Reduced glutathione
- GSSG:
-
Oxidized glutathione
- DOPAC:
-
3,4-Dihydroxy-phenylacetate
References
Abdel-Salam, O. M., Sleem, A. A., & Morsy, F. A. (2007). Effects of biphenyldimethyl-dicarboxylate administration alone or combined with silymarin in the CCL4 model of liver fibrosis in rats. ScientificWorldJournal, 7, 1242–1255.
Asatoor, A. (1968). The origin of urinary tyramine. Formation in tissues and by intestinal microorganisms. Clinica Chimica Acta, 22, 223–229.
Bao, Y., Zhao, T., Wang, X., Qiu, Y., Su, M., Jia, W., et al. (2009). Metabonomic variations in the drug-treated type 2 diabetes mellitus patients and healthy volunteers. Journal of Proteome Research, 8, 1623–1630.
Beauchamp, G. K., Keast, R. S., Morel, D., Lin, J., Pika, J., Han, Q., et al. (2005). Phytochemistry: ibuprofen-like activity in extra-virgin olive oil. Nature, 437, 45–46.
Beger, R. D., Sun, J., & Schnackenberg, L. K. (2010). Metabolomics approaches for discovering biomarkers of drug-induced hepatotoxicity and nephrotoxicity. Toxicology and Applied Pharmacology, 243, 154–166.
Boll, M., Weber, L. W., Becker, E., & Stampfl, A. (2001). Pathogenesis of carbon tetrachloride-induced hepatocyte injury bioactivation of CCI4 by cytochrome P450 and effects on lipid homeostasis. Z Naturforsch C, 56, 111–121.
Brosnan, J. T. (2003). Interorgan amino acid transport and its regulation. Journal of Nutrition, 133, 2068S–2072S.
Catala, A. (2009). Lipid peroxidation of membrane phospholipids generates hydroxy-alkenals and oxidized phospholipids active in physiological and/or pathological conditions. Chemistry and Physics of Lipids, 157, 1–11.
Chen, C., Krausz, K. W., Shah, Y. M., Idle, J. R., & Gonzalez, F. J. (2009). Serum metabolomics reveals irreversible inhibition of fatty acid beta-oxidation through the suppression of PPARalpha activation as a contributing mechanism of acetaminophen-induced hepatotoxicity. Chemical Research in Toxicology, 22, 699–707.
Cherkashina, D. V., & Petrenko, A. Y. (2006). Hepatoprotective effect of fetal tissue cytosol and its thermostable fraction in rats with carbon tetrachloride-induced hepatitis. Bulletin of Experimental Biology and Medicine, 141, 544–547.
Choudry, H. A., PAN, M., Karinch, A. M., & Souba, W. W. (2006). Branched-chain amino acid-enriched nutritional support in surgical and cancer patients. Journal of Nutrition, 136, 314S–318S.
Feng, C., Wang, H., Yao, C., Zhang, J., & Tian, Z. (2007). Diammonium glycyrrhizinate, a component of traditional Chinese medicine Gan-Cao, prevents murine T-cell-mediated fulminant hepatitis in IL-10- and IL-6-dependent manners. International Immunopharmacology, 7, 1292–1298.
Fodor, O., Schvartz, M., Suciu, A., Barbarino, F., Duca, S., & Lezeu, R. (1976). Protecting action of aspartate on the hepatic changes induced by D-galactosamine. Arzneimittelforschung, 26, 855–858.
Froh, M., Zhong, Z., Walbrun, P., Lehnert, M., Netter, S., Wiest, R., et al. (2008). Dietary glycine blunts liver injury after bile duct ligation in rats. World Journal of Gastroenterology, 14, 5996–6003.
Goldstein, D. S., Eisenhofer, G., & Kopin, I. J. (2003). Sources and significance of plasma levels of catechols and their metabolites in humans. Journal of Pharmacology and Experimental Therapeutics, 305, 800–811.
Grizzi, F., Franceschini, B., Gagliano, N., Moscheni, C., Annoni, G., Vergani, C., et al. (2003). Mast cell density, hepatic stellate cell activation and TGF-beta1 transcripts in the aging Sprague-Dawley rat during early acute liver injury. Toxicologic Pathology, 31, 173–178.
Holecek, M., Tilser, I., Skopec, F., & Sprongl, L. (1996). Leucine metabolism in rats with cirrhosis. Journal of Hepatology, 24, 209–216.
Lin, Y., SI, D., Zhang, Z., & Liu, C. (2009). An integrated metabonomic method for profiling of metabolic changes in carbon tetrachloride induced rat urine. Toxicology, 256, 191–200.
Maillet, E. L., Margolskee, R. F., & Mosinger, B. (2009). Phenoxy herbicides and fibrates potently inhibit the human chemosensory receptor subunit T1R3. Journal of Medicinal Chemistry, 52, 6931–6935.
Nowak, A., & Libudzisz, Z. (2006). Influence of phenol, p-cresol and indole on growth and survival of intestinal lactic acid bacteria. Anaerobe, 12, 80–84.
Okamoto, T., & Okabe, S. (2000). Carbon tetrachloride treatment induces anorexia independently of hepatitis in rats. International Journal of Molecular Medicine, 6, 181–183.
Pan, L., Qiu, Y., Chen, T., Lin, J., Chi, Y., Su, M., et al. (2010). An optimized procedure for metabonomic analysis of rat liver tissue using gas chromatography/time-of-flight mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis, 52, 589–596.
Qiu, Y., Su, M., Liu, Y., Chen, M., GU, J., Zhang, J., et al. (2007). Application of ethyl chloroformate derivatization for gas chromatography-mass spectrometry based metabonomic profiling. Analytica Chimica Acta, 583, 277–283.
Rechner, A. R., Smith, M. A., Kuhnle, G., Gibson, G. R., Debnam, E. S., Srai, S. K., et al. (2004). Colonic metabolism of dietary polyphenols: influence of structure on microbial fermentation products. Free Radical Biology and Medicine, 36, 212–225.
Robertson, D. G., Reily, M. D., Sigler, R. E., Wells, D. F., Paterson, D. A., & Braden, T. K. (2000). Metabonomics: evaluation of nuclear magnetic resonance (NMR) and pattern recognition technology for rapid in vivo screening of liver and kidney toxicants. Toxicological Sciences, 57, 326–337.
Sun, J., Schnackenberg, L. K., & Beger, R. D. (2009). Studies of acetaminophen and metabolites in urine and their correlations with toxicity using metabolomics. Drug Metabolism Letters, 3, 130–136.
Vulimiri, S. V., Berger, A. & Sonawane, B. (2010). The potential of metabolomic approaches for investigating mode(s) of action of xenobiotics: Case study with carbon tetrachloride. Mutation Research, Accession number: 20188855, epub ahead of print.
Wang, X., Su, M., Qiu, Y., NI, Y., Zhao, T., Zhou, M., et al. (2007). Metabolic regulatory network alterations in response to acute cold stress and ginsenoside intervention. Journal of Proteome Research, 6, 3449–3455.
Wang, X., Zhao, T., Qiu, Y., Su, M., Jiang, T., Zhou, M., et al. (2009). Metabonomics approach to understanding acute and chronic stress in rat models. Journal of Proteome Research, 8, 2511–2518.
Weber, L. W., Boll, M., & Stampfl, A. (2003). Hepatotoxicity and mechanism of action of haloalkanes: Carbon tetrachloride as a toxicological model. Critical Reviews in Toxicology, 33, 105–136.
Wiklund, S., Johansson, E., Sjostrom, L., Mellerowicz, E. J., Edlund, U., Shockcor, J. P., et al. (2008). Visualization of GC/TOF-MS-based metabolomics data for identification of biochemically interesting compounds using OPLS class models. Analytical Chemistry, 80, 115–122.
Xu, X., Ling, Q., Gao, F., He, Z. L., Xie, H. Y., & Zheng, S. S. (2009). Hepatoprotective effects of marine and kuhuang in liver transplant recipients. American Journal of Chinese Medicine, 37, 27–34.
Yuan, H., Ji, W. S., Wu, K. X., Jiao, J. X., Sun, L. H., & Feng, Y. T. (2006). Anti-inflammatory effect of Diammonium Glycyrrhizinate in a rat model of ulcerative colitis. World Journal of Gastroenterology, 12, 4578–4581.
Acknowledgments
This work was financially supported by the National Basic Research Program of China (2007CB914700), the National Science and Technology Major Project (2009ZX10005-020) and the National Natural Science Foundation of China Grant 30901997, 20775048 and the International Collaborative Project of Chinese Ministry of Science and Technology (2006DFA02700).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, X., Lin, J., Chen, T. et al. Metabolic profiling reveals the protective effect of diammonium glycyrrhizinate on acute hepatic injury induced by carbon tetrachloride. Metabolomics 7, 226–236 (2011). https://doi.org/10.1007/s11306-010-0244-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11306-010-0244-5