Abstract
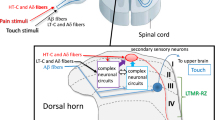
P2X3 monomeric receptors (P2X3Rs) and P2X2/3 heteromeric receptors (P2X2/3Rs) in primary sensory neurons and microglial P2X4 monomeric receptors (P2X4Rs) in the spinal dorsal horn (SDH) play important roles in neuropathic pain. In particular, P2X4R in the spinal microglia during peripheral nerve injury (PNI), experimental autoimmune neuritis, and herpes models are useful to explore the potential strategies for developing new drugs to treat neuropathic pain. Recently, novel P2X4 antagonists, NP-1815-PX and NC-2600, were developed, which demonstrated potent and specific inhibition against rodent and human P2X4Rs. The phase I study of NC-2600 has been completed, and no serious side effects were reported. The roles played by purinergic receptors in evoking neuropathic pain provide crucial insights into the pathogenesis of neuropathic pain.




Similar content being viewed by others
References
Burnstock G (2006) Purinergic P2 receptors as targets for novel analgesics. Pharmacol Ther 110:433–454
Aldskogius H, Kozlova EN (1998) Central neuron–glial and glial–glial interactions following axon injury. Prog Neurobiol 55:1–26
Carson MJ (2002) Microglia as liaisons between the immune and central nervous systems: functional implications for multiple sclerosis. Glia 40:218–231
Eikelenboom P, Bate C, Van Gool WA, Hoozemans JJ, Rozemuller JM, Veerhuis R, Williams A (2002) Neuroinflammation in Alzheimer’s disease and prion disease. Glia 40:232–239
Colburn RW, Rickman AJ, Deleo JA (1999) The effect of site and type of nerve injury on spinal glial activation and neuropathic pain behavior. Exp Neurol 157:289–304
Sweitzer S, Martin D, Deleo JA (2001) Intrathecal interleukin-1 receptor antagonist in combination with soluble tumor necrosis factor receptor exhibits an anti-allodynic action in a rat model of neuropathic pain. Neuroscience 103:529–539
Garden GA (2002) Microglia in human immunodeficiency virusassociated neurodegeneration. Glia 40:240–251
Inoue K (2007) P2 receptors and chronic pain. Purinergic Signal 3:135–144
Cho T, Chaban VV (2012) Expression of P2X3 and TRPV1 receptors in primary sensory neurons from estrogen receptors-α and estrogen receptor-β knockout mice. Neuroreport 23:530–534
Inoue K, Tsuda M (2018) Microglia in neuropathic pain: cellular and molecular mechanisms and therapeutic potential. Nat Rev Neurosci 19:138–152
Collo G, North RA, Kawashima E, Merlo-Pich E, Neidhart S, Surprenant A, Buell G (1996) Cloning OF P2X5 and P2X6 receptors and the distribution and properties of an extended family of ATP-gated ion channels. J Neurosci 16:2495–2507
Rae MG, Rowan EG, Kennedy C (1998) Pharmacological properties of P2X3-receptors present in neurones of the rat dorsal root ganglia. Br J Pharmacol 124:176–180
Ueno S, Tsuda M, Iwanaga T, Inoue K (1999) Cell type-specific ATP-activated responses in rat dorsal root ganglion neurons. Br J Pharmacol 126:429–436
Bonventre JV, Huang Z, Taheri MR, O'Leary E, Li E, Moskowitz MA, Sapirstein A (1997) Reduced fertility and postischaemic brain injury in mice deficient in cytosolic phospholipase A2. Nature 390:622–625
Hirabayashi T, Shimizu T (2000) Localization and regulation of cytosolic phospholipase a(2). Biochim Biophys Acta 1488:124–138
Lin LL, Wartmann M, Lin AY, Knopf JL, Seth A, Davis RJ (1993) cPLA2 is phosphorylated and activated by MAP kinase. Cell 72:269–278
Hasegawa S, Kohro Y, Tsuda M, Inoue K (2009) Activation of cytosolic phospholipase A2 in dorsal root ganglion neurons by Ca2+/calmodulin-dependent protein kinase II after peripheral nerve injury. Mol Pain 5:22
Schievella AR, Regier MK, Smith WL, Lin LL (1995) Calcium-mediated translocation of cytosolic phospholipase A2 to the nuclear envelope and endoplasmic reticulum. J Biol Chem 270:30749–30754
Stahelin RV, Rafter JD, Das S, Cho W (2003) The molecular basis of differential subcellular localization of C2 domains of protein kinase C-alpha and group IVa cytosolic phospholipase A2. J Biol Chem 278:12452–12460
Barclay J, Patel S, Dorn G, Wotherspoon G, Moffatt S, Eunson L, Abdel'al S, Natt F, Hall J, Winter J, Bevan S, Wishart W, Fox A, Ganju P (2002) Functional downregulation of P2X3 receptor subunit in rat sensory neurons reveals a significant role in chronic neuropathic and inflammatory pain. J Neurosci 22:8139–8147
Honore P, Mikusa J, Bianchi B, McDonald H, Cartmell J, Faltynek C, Jarvis MF (2002) TNP-ATP, a potent P2X(3) receptor antagonist, blocks acetic acid- induced abdominal constriction in mice: comparison with reference analgesics. Pain 96:99–105
Tsuda M, Koizumi S, Kita A, Shigemoto Y, Ueno S, Inoue K (2000) Mechanical allodynia caused by intraplantar injection of P2X receptor agonist in rats: involvement of heteromeric P2X2/3 receptor signaling in capsaicin-insensitive primary afferent neurons. J Neurosci 20:RC90
Bouvier MM, Evans ML, Benham CD (1991) Calcium influx induced by stimulation of ATP receptors on neurons cultured from rat dorsal root ganglia. Eur J Neurosci 3:285–291
North RA, Surprenant A (2000) Pharmacology of cloned P2X receptors. Annu Rev Pharmacol Toxicol 40:563–580
Jarvis MF, Khakh BS (2009) ATP-gated P2X cation-channels. Neuropharmacol 56:208–215
Hasegawa S, Kohro Y, Shiratori M, Ishii S, Shimizu T, Tsuda M, Inoue K (2010) Role of PAF receptor in proinflammatory cytokine expression in the dorsal root ganglion and tactile allodynia in a rodent model of neuropathic pain. PLoS One 5:e10467
Schafers M, Svensson CI, Sommer C, Sorkin LS (2003) Tumor necrosis factor-alpha induces mechanical allodynia after spinal nerve ligation by activation of p38 MAPK in primary sensory neurons. J Neurosci 23:2517–2521
Kawasaki Y, Xu ZZ, Wang X, Park JY, Zhuang ZY, Tan PH, Gao YJ, Roy K, Corfas G, Lo EH, Ji RR (2008) Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain. Nat Med 14:331–336
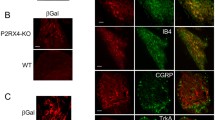
Tsuda M, Shigemoto-Mogami Y, Koizumi S, Mizokoshi A, Kohsaka S, Salter MW, Inoue K (2003) P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424:778–783
Ulmann L, Hatcher JP, Hughes JP, Chaumont S, Green PJ, Conquet F, Buell GN, Reeve AJ, Chessell IP, Rassendren F (2008) Up-regulation of P2X4 receptors in spinal microglia after peripheral nerve injury mediates BDNF release and neuropathic pain. J Neurosci 28:11263–11268
Trang T, Beggs S, Wan X, Salter MW (2009) P2X4-receptor-mediated synthesis and release of brain-derived neurotrophic factor in microglia is dependent on calcium and p38-mitogen-activated protein kinase activation. J Neurosci 29:3518–3528
Coull JA, Beggs S, Boudreau D, Boivin D, Tsuda M, Inoue K, Gravel C, Salter MW, De Koninck Y (2005) BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature 438:1017–1021
Masuda T, Ozono Y, Mikuriya S, Kohro Y, Tozaki-Saitoh H, Iwatsuki K, Uneyama H, Ichikawa R, Salter MW, Tsuda M, Inoue K (2016) Dorsal horn neurons release extracellular ATP in neuropathic pain in a VNUT-dependent manner. Nat Commun 7:12529
Loeser JD (1986) Herpes zoster and postherpetic neuralgia. Pain 25:149–164
Takasaki I, Andoh T, Shiraki K, Kuraishi Y (2000) Allodynia and hyperalgesia induced by herpes simplex virus type-1 infection in mice. Pain 86:95–101
Sasaki A, Inomata Y, Serizawa K, Andoh T, Kuraishi Y (2013) Contribution of sensory C-fiber neuron injury to mechanical dynamic allodynia in a murine model of postherpetic neuralgia. Neuroreport 24:137–141
Takasaki I, Taniguchi K, Komatsu F, Sasaki A, Andoh T, Nojima H, Shiraki K, Hsu DK, Liu FT (2012) Kato I et al.:contribution of spinal galectin-3 to acute herpetic allodynia in mice. Pain 153:585–592
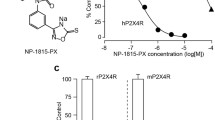
Matsumura Y, Yamashita T, Sasaki A, Nakata E, Kohno K, Masuda T, Tozaki-Saitoh H, Imai T, Kuraishi Y, Tsuda M, Inoue K (2016) A novel P2X4 receptor-selective antagonist produces anti-allodynic effect in a mouse model of herpetic pain. Sci Rep 6:32461
Imai T, Nakata E, Inoue K (2012) Inhibition of P2X4 receptor on spinal microglia attenuates mechanical allodynia in experimental autoimmune neuritis rats. Pain Res 27:27–36
Zhang Z, Zhang ZY, Fauser U, Schluesener HJ (2008) Mechanical allodynia and spinal up-regulation of P2X4 receptor in experimental autoimmune neuritis rats. Neuroscience 152:495–501
Nagata K, Imai T, Yamashita T, Tozaki-Saitoh H, Tsuda M, Inoue K (2009) Antidepressants inhibit P2X4 receptor function: a possible involvement in neuropathic pain relief. Mol Pain 5:20
Richards D, Gever JR, Ford AP, Fountain SJ (2019) Action of MK-7264 (Gefapixant) at human P2X3 and P2X2/3 receptors and in vivo efficacy in models of sensitization. Br J Pharmacol 176:2279–2291
Morice AH, Kitt MM, Ford AP, Tershakovec AM, Wu WC, Brindle K, Thompson R, Thackray-Nocera S, Wright C (2019) The effect of gefapixant, a P2X3 antagonist, on cough reflex sensitivity: a randomised placebo-controlled study. Eur Respir J 54:1900439
Finger TE, Danilova V, Barrows J, Bartel DL, Vigers AJ, Stone L, Hellekant G, Kinnamon SC (2005) ATP signaling is crucial for communication from taste buds to gustatory nerves. Science 310:1495–1499
Funding
This work was supported partly by JSPS KAKENHI Grant Numbers 25117013 and the Core Research for Evolutional Science and Technology (AMED-CREST) program from Japan Agency for Medical Research and Development. I also thank Drs. Makoto Tsuda, Hidetoshi Tozaki-Saitoh, Takahiro Masuda and many students for the experiments. I thank Lisa Giles, PhD, from Edanz Group (https://en-author-services.edanzgroup.com/ac) for suggesting to edit a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Kazuhide Inoue declares that he/she has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on A Tribute to Professor Geoff Burnstock.
Rights and permissions
About this article
Cite this article
Inoue, K. Nociceptive signaling of P2X receptors in chronic pain states. Purinergic Signalling 17, 41–47 (2021). https://doi.org/10.1007/s11302-020-09743-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-020-09743-w