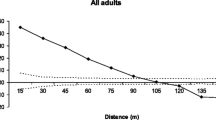
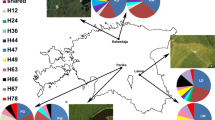
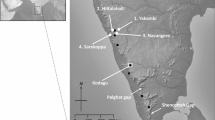
Abstract
Self-incompatible plants as well as common plant species are expected to be especially vulnerable to the deleterious effects of fragmentation on genetic diversity. Paypayrola blanchetiana (Violaceae) is a common, self-incompatible understory tree in the East Brazilian Atlantic forest. Its autochorous seed dispersal and occurrence in dense, well-separated clusters makes it an interesting model for studies of gene flow and genetic structuring on a small geographic scale. A previous study has found remarkably low fruit set and frequent seed abortions in this species in several populations situated in forest fragments. We tested the hypothesis that P. blanchetiana is affected by inbreeding and loss of genetic diversity in a fragmented landscape. Nine nuclear and three plastid microsatellite loci were genotyped for 285 individuals (149 adult trees and 136 saplings) across seven populations in five forest fragments in a 240-km2 sugarcane-rainforest matrix. We found a low to moderate genetic diversity in nuclear loci of P. blanchetiana, population structuring on a small geographical scale and high levels of inbreeding. Haplotype distributions confirmed that seed dispersal is very limited. There were, however, no signs for lower genetic diversity or higher inbreeding in populations situated in the smallest forest fragments. Furthermore, genetic diversity was not lower in the sapling cohort, which was created in post-fragmentation condition. Therefore, we may be witnessing the genetic consequences of this species’ biology, rather than immediate effects of fragmentation.



Similar content being viewed by others
References
Aguilar R, Quesada M, Ashworth L, Herrerias-Diego Y, Lobo J (2008) Genetic consequences of habitat fragmentation in plant populations: susceptible signals in plant traits and methodological approaches. Mol Ecol 17:5177–5188. https://doi.org/10.1111/j.1365-294X.2008.03971.x
André T, Lemes MR, Grogan J, Gribel R (2008) Post-logging loss of genetic diversity in a mahogany (Swietenia macrophylla King, Meliaceae) population in Brazilian Amazonia. Forest Ecol Manag 255:340–345. https://doi.org/10.1016/j.foreco.2007.09.055
Ballesteros-Mejia L, Lima NE, Lima-Ribeiro MS, Collevati RG (2016) Pollination mode and mating system explain patterns in genetic differentiation in Neotropical plants. PLoS One 11(7):e0158660
Bandelt HJ, Forster P, Rohl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16:37–48. https://doi.org/10.1093/oxfordjournals.molbev.a026036
Braun M, Dötterl S, Schlindwein C, Gottsberger G (2012) Can nectar be a disadvantage? Contrasting pollination natural histories of two woody Violaceae from the Neotropics. Int J Plant Sci 173:161–171. https://doi.org/10.1086/663167
Braun M, Esposito T, Huettel B, Pedrosa-Harand A (2019) Development and characterization of eleven microsatellite loci for the tropical understory tree Paypayrola blanchetiana Tul. (Violaceae). Mol Biol Rep 46:2529–2532. https://doi.org/10.1007/s11033-019-04622-z
Braun M, Piechowski D, Kazda M, Gottsberger G (2012) Fragment size and local flower density influence seed set of the understorey tree Paypayrola blanchetiana (Violaceae) in Brazilian Atlantic rain forest. J Trop Ecol 28:353–359. https://doi.org/10.1017/S0266467412000314
Browne L, Ottewell K, Karubian J (2015) Short-term genetic consequences of habitat loss and fragmentation for the neotropical palm Oenocarpus bataua. Heredity 115:389–395
Byers DL, Meagher TR (1992) Mate availability in small populations of plant species with homomorphic sporophytic self-incompatibility. Heredity 68:353–359
Chapuit M-P, Estoup A (2007) Microsatellite null alleles and estimation of population differentiation. Mol Biol Evol 24:621–631. https://doi.org/10.1093/molbev/msl191
Chase MR, Moller C, Kesseli R, Bawa KS (1996) Distant gene flow in tropical trees. Nature 383:398–399. https://doi.org/10.1038/383398a0
Chen C, Lu RS, Zhu SS, Tamaki I, Qiu YX (2017) Population structure and historical demography of Dipteronia dyeriana (Sapindaceae), an extremely narrow palaeoendemic plant from China: implications for conservation in a biodiversity hot spot. Heredity 119:95–106. https://doi.org/10.5061/dryad.t8q1g
Cheon, K-S, Jang, S-K, Kim, K-A, Yang, J-C, Yoo, K-O (2015) The first complete chloroplast genome sequence from Violaceae (Viola seoulensis). GenBank: KP749924.1
Collevatti RG, Grattapaglia D, Hay JD (2001a) Population genetic structure of the endangered tropical tree species Caryocar brasiliense, based on variability at microsatellite loci. Mol Ecol 10:349–356. https://doi.org/10.1046/j.1365-294x.2001.01226.x
Collevatti RG, Grattapaglia D, Hay JD (2001b) High resolution microsatellite based analysis of the mating system allows the detection of significant biparental inbreeding in Caryocar brasiliense, an endangered tropical tree species. Heredity 86:60–67. https://doi.org/10.1046/j.1365-2540.2001.00801.x
Collevatti RG, Leoi LCT, Leite SA, Gribel R (2009) Contrasting patterns of genetic structure in Caryocar (Caryocaraceae) congeners from flooded and upland Amazonian forests. Biol J Linn Soc 98:278–290. https://doi.org/10.1111/j.1095-8312.2009.01287.x
Cornuet JM, Luikart G (1997) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144:2001–2014
Degen B, Bandou E, Caron H (2004) Limited pollen dispersal and biparental inbreeding in Symphonia globulifera in French Guiana. Heredity 93:585–591. https://doi.org/10.1038/sj.hdy.6800560
Dick CW, Hardy OJ, Jones FA, Petit RJ (2008) Spatial scales of pollen and seed-mediated gene flow in tropical rainforest trees. Trop Plant Biol 1:20–32. https://doi.org/10.1007/s12042-007-9006-6
Domínguez CA, Abarca CA, Eguiarte LE, Molina-Freaner F (2005) Local genetic differentiation among populations of the mass-flowering tropical shrub Erythroxylum havanense (Erythroxylaceae). New Phytol 166:663–672. https://doi.org/10.1111/j.1469-8137.2005.01359.x
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Duminil J, Hardy OJ, Petit RJ (2009) Plant traits correlated with generation time directly affect inbreeding depression and mating system and indirectly genetic structure. BMC Evol Biol 9:177. https://doi.org/10.1186/1471-2148-9-177
Dutech C, Seiter J, Petronelli P, Joly HI, Jarne P (2002) Evidence of low gene flow in a neotropical clustered tree species in two rainforest stands of French Guiana. Mol Ecol 11:725–738. https://doi.org/10.1046/j.1365-294x.2002.01475.x
Earl DA, von Holdt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. https://doi.org/10.1007/s12686-011-9548-7
Eliades N-G, Eliades DG (2009) HAPLOTYPE ANALYSIS: software for analysis of haplotypes data. Distributed by the authors. Forest Genetics and Forest Tree Breeding, Georg-Augst University Goettingen, Germany
Ennos RA (1994) Estimating the relative rates of pollen and seed migration among plant-populations. Heredity 72:250–259
Evanno G, Regnaut S, Goudet J (2005) Detecting the numbers of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567. https://doi.org/10.1111/j.1755-0998.2010.02847.x
Ferreira ME, Grattapaglia D (1995) Introdução ao uso de marcadores moleculares em análise genética. EMBRAPA-CENARGEN, Brasília. https://doi.org/10.13140/2.1.3757.8881
Galindo-Leal C, Camara IG (2003) Atlantic forest hotspot status: an overview. In: Galindo-Leal C, Camara IDG (eds) The Atlantic Forest of South America – biodiversity status, threats and outlook. Island Press, Washington, pp 3–11
Garcia C, Arroy JM, Godoy JA, Jordano P (2005) Mating patterns, pollen dispersal, and the ecological maternal neighbourhood in a Prunus mahaleb L. population. Mol Ecol 14:1821–1830. https://doi.org/10.1111/j.1365-294X.2005.02542.x
Goetze M, Palma-Silva C, Martini Zanella C, Bered F (2016) East-to-west genetic structure in populations of Aechmea calyculata (Bromeliaceae) from the southern Atlantic rainforest of Brazil. Bot J Linn Soc 181:477–490. https://doi.org/10.1111/boj.12416
Goudet J (1995) FSTAT (version 1.2): a computer program to calculate F-statistics. J Hered 86:485–486
Hamrick JL, Murawski DA, Nason JD (1993) The influence of seed dispersal mechanisms on the genetic structure of tropical tree populations. Vegetatio 107:281–297
Jha S, Dick CW (2010) Native bees mediate long-distance pollen dispersal in a shade coffee landscape mosaic. PNAS 107:13760–13764. https://doi.org/10.1073/pnas.1002490107
Kramer AT, Ison JL, Ashley MV, Howe AF (2008) The paradox of forest fragmentation genetics. Conserv Biol 22:878–885. https://doi.org/10.1111/j.1523-1739.2008.00944.x
Krutovsky KV, Neale DB (2005) Forest genomics and new molecular genetic approaches to measuring and conserving adaptive genetic diversity in forest trees. In: Geburek T, Turok J (eds) Conservation and management of forest genetic resources in Europe. Arbora Publishers, Zvolen, pp 369–390
Lachenaud P, Zhang D (2008) Genetic diversity and population structure in wild stands of cacao trees (Theobroma cacao L.) in French Guiana. Ann For Sci 65:310. https://doi.org/10.1051/forest:2008011
Lasso E, Dalling JW, Bermingham E (2011) Strong spatial genetic structure in five tropical Piper species: should the Baker–Fedorov hypothesis be revived for tropical shrubs? Ecol Evol 1:502–516. https://doi.org/10.1002/ece3.40
Lexer C, Marthaler F, Humbert S, Barbará T, de la Harpe M, Bossolini E, Paris M, Martinelli G, Versieux LM (2016) Gene flow and diversification in a species complex of Alcantarea inselberg bromeliads. Bot J Linn Soc 181:505–520. https://doi.org/10.1111/boj.12372
Lowe AJ, Boshier D, Ward M, Bacles CFE, Navarro C (2005) Genetic resource impacts of habitat loss and degradation; reconciling empirical evidence and predicted theory for neotropical trees. Heredity 95:255–273. https://doi.org/10.1038/sj.hdy.6800725
Mao LH, Zhou XL, Fang YM (2016) Genetic diversity and population structure of Castanopsis eyrei based on simple sequence repeat markers. Genet Mol Res 15:1–8. https://doi.org/10.4238/gmr.15028256
Martini AM, Fiaschi P, Amorim AM, da Paixa OJL (2007) A hot-point within a hot-spot: a high diversity site within Brazil’s Atlantic Forest. Biodivers Conserv 16:3111–3128. https://doi.org/10.1007/s10531-007-9166-6
Melo A, Franceschinelli EV (2016) Gene flow and fine-scale spatial genetic structure in Cabralea canjerana (Meliaceae), a common tree species from the Brazilian Atlantic forest. J Trop Ecol 32:135–145. https://doi.org/10.1017/S0266467416000067
Minasiewicz J, Znaniecka JM, Górniak M, Kawiński A (2018) Spatial genetic structure of an endangered orchid Cypripedium calceolus (Orchidaceae) at a regional scale: limited gene flow in a fragmented landscape. Conserv Genet 19:1449–1460. https://doi.org/10.1007/s10592-018-1113-4
Moraes-Filho RM, Bonifácio-Anacleto F, Alzate-Marin AL (2015) Fragmentation effects and genetic diversity of the key semidecidual forest species Metrodorea nigra in Southwestern Brazil. Genet Mol Res 14:3509–3524. https://doi.org/10.4238/2015.April.15.15
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Nattero J, Malerba R, Medel R, Coccucci A (2011) Factors affecting pollinator movement and plant fitness in a specialized pollination system. Plant Syst Evol 296:77–85. https://doi.org/10.1007/s00606-011-0477-4
Nei M (1978) Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 89:583–590
Opedal ØH, Falahati-Anbaran M, Albertsen E, Armbruster WS, Pérez-Barrales R, Stenøien HK, Pélabon C (2017) Euglossine bees mediate only limited long-distance gene flow in a tropical vine. New Phytol 213:1898–1908. https://doi.org/10.1111/nph.14380
Palma-Silva C, Lexer C, Paggi GM, Barbará T, Bered F, Bodanese-Zanettini MH (2009) Range-wide patterns of nuclear and chloroplast DNA diversity in Vriesea gigantea (Bromeliaceae), a neotropical forest species. Heredity 103:503–512. https://doi.org/10.1038/hdy.2009.116
Piry S, Luikart G, Cornuet JM (1999) BOTTLENECK: a computer program for detecting recent reductions in the effective size using allele frequency data. Heredity 90:502–503. https://doi.org/10.1093/jhered/90.4.502
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Raymond M, Rousset F (1995) GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Schlaepfer D, Braschler B, Rusterholz H-P, Bauer B (2018) Genetic effects of anthropogenic habitat fragmentation on remnant animal and plant populations: a meta-analysis. Ecosphere 9(10):e02488. https://doi.org/10.1002/ecs2.2488
Schuelke M (2000) An economic method for the fluorescent labeling of PCR fragments. Nat Biotechnol 18:233–234. https://doi.org/10.1038/72708
Sokal RR, Rohlf FI (1995) Biometry: the principles and practice of statistics in biological research. Freeman and Company, NewYork
Stacy EA, Hamrick JL, Nason JD, Hubbell SP, Foster RB, Condit R (1996) Pollen dispersal in low-density populations of three Neotropical tree species. Am Nat 148:275–298. https://doi.org/10.1086/285925
Steane DA, Jones RC, Vaillancourt RE (2005) A set of chloroplast microsatellite primers for Eucalyptus (Myrtaceae). Mol Ecol Notes 5:538–541. https://doi.org/10.1111/j.1471-8286.2005.00981.x
Trindade MB, Lins-e-Silva ACB, da Silva HP, Figueira SB, Schessl M (2008) Fragmentation of the Atlantic Rainforest in the Northern Coastal Region of Pernambuco, Brazil: recent changes and implications for conservation. Bioremediat Biodivers Bioavail 2:5–13
Valière N (2002) GIMLET: a computer program for analysing genetic individual identification data. Mol Ecol Resour 2:377–379
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) Micro-Checker: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538. https://doi.org/10.1111/j.1471-8286.2004.00684.x
Vranckx G, Jacquemyn H, Muys B, Honnay O (2012) Meta-analysis of susceptibility of woody plants to loss of genetic diversity through habitat fragmentation. Conserv Biol 26:228–237. https://doi.org/10.1111/j.1523-1739.2011.01778.x
Ward M, Dick CW, Gribel R, Lowe AJ (2005) To self or not to self…a review of outcrossing and pollen-mediated gene flow in neotropical trees. Heredity 95:246–254. https://doi.org/10.1038/sj.hdy.6800712
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370. https://doi.org/10.1111/j.1558-5646.1984.tb05657.x
Weising K, Gardner RC (1999) A set of conserved PCR primers for the analysis of simple sequence repeat polymorphisms in chloroplast genomes of dicotyledonous angiosperms. Genome 42:9–19
Wright S (1965) The interpretation of population structure by F-statistics with special regards to system of mating. Evolution 19:395–420. https://doi.org/10.1111/j.1558-5646.1965.tb01731.x
Young A, Boyle T, Brown T (1996) The population genetic consequences of habitat fragmentation for plants. Trends Ecol Evol 11:413–418. https://doi.org/10.1016/0169-5347(96)10045-8
Young AG, Pickup M (2010) Low S-allele numbers limit mate availability, reduce seed set and skew fitness in small populations of a self-incompatible plant. J Appl Ecol 47:541–548. https://doi.org/10.1111/j.1365-2664.2010.01798.x
Acknowledgments
The authors thank Usina São José S.A., municipality of Igarassú (Pernambuco) for facilitating collection of leaf material, and Dr. Theresa Liberal, Bruno Sampaio, and the Laboratory of Bioinformatics and Evolutionary Biology (Federal University of Pernambuco, Recife) for providing services of the ABI-3500 sequencing platform.
Data archiving statement
Genotype (SSR markers) and haplotype data have been deposited in the Tree Genes Database (acession-nr. TGDR 377; https://doi.org/10.5281/zenodo.4274480).
Funding
The study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (grant no. 300532/2016-4) and Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (grant no. APQ-0207-2.03/13).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interest.
Additional information
Communicated by N. Tomaru
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Population structure
Rights and permissions
About this article
Cite this article
Braun, M., Dantas, L., Esposito, T. et al. Strong genetic differentiation on a small geographic scale in the Neotropical rainforest understory tree Paypayrola blanchetiana (Violaceae). Tree Genetics & Genomes 16, 85 (2020). https://doi.org/10.1007/s11295-020-01477-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-020-01477-5