Abstract
The classical “low latitude–high defense” hypothesis is seldom supported by empirical evidence. In this context, we tested latitudinal patterns in the leaf defense traits of deciduous broadleaved (DB) and evergreen broadleaved (EGB) tree species, which are expected to affect herbivore diversity. We examined the co-occurrence of leaf defense traits (tannin and phenol content, leaf mechanical strength, leaf dry matter content, leaf mass per area, and leaf thickness) in 741 broadleaved tree species and their correlations with species geographical range in East Asian island flora. We discovered contrasting latitudinal defense strategy gradients in DB and EGB tree species. DB species employed chemical defenses (increasing tannin and phenol content) at higher latitudes and physical defenses (softer and thinner leaves) at lower latitudes, whereas EGB tree species exhibited opposite latitudinal defense patterns. The “low latitude high defense” hypothesis included a paradoxical aspect in chemical and physical defense traits across broadleaved tree species. To reconcile paradoxical defense strategies along the latitudinal gradient, we conclude that interactive correlations among leaf traits are controlled by leaf longevity, which differs between DB and EGB tree species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biological interactions among organisms are believed widely to intensify at lower latitudes (Lewinsohn and Roslin 2008), leading to the development of latitudinal diversity gradient (LDG) hypotheses to explain large–scale biodiversity patterns (Dobzhansky 1950; MacArthur 1972; Pennings and Silliman 2005). As plants and herbivores comprise at least 40% of global terrestrial biodiversity (Price 2002), evaluation of the consequences of plant–herbivore interaction on the LDG should attract much interest (Marquis et al. 2012). Several studies have examined the “low latitude high defense” (LLHD) hypothesis (Bolser and Hay 1996) which posits that plant species distributed at lower latitudes will show higher degrees of defense (Dobzhansky 1950, Coley and Aide 1991, Schmitt et al. 1995). Although these studies have been conducted with the aim of confirming a latitudinal gradient in defense strength (Moles et al. 2011), few have found support for the hypothesis.
Moles et al. (2011) performed a meta–analysis of defense trait data, both chemical (tannins and phenols, flavonoids, alkaloids etc.) and physical (physical toughness, extrafloral nectaries etc.) defense traits, across a wide range of latitude, and found conflicting trends in response to predictions based on the LLHD hypothesis, with increasing and decreasing gradients, as well as nonsignificant trends, in plant defense traits across latitudes. One pitfall of this study is the large bias caused by variation in various plant functional types among individual studies (Sitch et al. 2003), which reduced the statistical power to detect or identify specific latitudinal gradients in the targeted trait variables. For the broadleaved (BL) tree species, several functional types (or groups), e.g., trees or shrubs, N fixers or not, are recognized (Wright et al. 2004; Kattge et al. 2011). Among them, the contrast between deciduous broadleaved (DB) and evergreen broadleaved (EGB) tree species should be the most evident (Wright et al. 2004). For example, Wright et al. (2005) showed clear contrast between DB and EGB tree species in leaf longevity along latitude. Thus, the separation of these functional types, namely DB vs. EGB, might clarify the conflicting trends in latitudinal gradient in plant defense traits. On the other hand, many empirical studies were based on trait data for particular plant taxa in phylogenetically narrow ranges (see supplemental tables in Moles et al. 2011; Anstett et al. 2016; see also Moles et al. 2013), which may make the detection of latitudinal gradients in defense strategies difficult.
As the defense strategies of each plant species, particular set of multiple defense traits were observed. Agrawal and Fishbein (2006) proposed the defense syndrome concept, in which several sets or combinations of defense traits are selected convergently as a syndrome. For example, they found three defense strategies in Asclepias plants: high physical and chemical defense with high nutrition, tolerance/escape, and low nutritional quality. Therefore, multiple functional traits should be examined simultaneously to explore plant defense strategies relevant to herbivores (Levin and York 1978). Thus, a comprehensive dataset including the entire species assemblage in the focal region for multiple sets of functional leaf traits should help us to rigorously examine the LLHD hypothesis (see also Anstett et al. 2016).
To test the LLHD hypothesis, we compiled a dataset of plant defense traits by thoroughly sampling BL tree species across the East Asian continental archipelago from the Hokkaido to Iriomote Islands (Maeshiro et al. 2013; Kusumoto et al. 2014; Shiono et al. 2015). In the present study, we first examined trait co–occurrence with respect to the chemical and mechanical defense strategies of each plant species using ordination. We then explored the latitudinal gradients of multiples of leaf defense trait of DB and EGB tree species by path analysis. Our goal is to clarify which leaf traits show the latitudinal gradients, and how are the directions of the gradient both for DB and EGB tree species. Although the dominant theory is the LLHD (Dobzhansky 1950; Coley and Aide 1991), we expect that much complex trends will be observed depends on the plant functional types as well as the leaf defense traits examined.
Materials and methods
Plant functional traits and latitudinal species distribution
We examined 272 EGB and 469 DB tree species, a total of 741 BL tree species. We focused on leaf traits that might function as defense against herbivores: tannin and phenol contents (%), leaf mechanical strength (g cm−2), leaf dry matter content (LDMC, %), leaf mass per area (LMA, g cm−2), and leaf thickness (µm). To build a dataset of leaf defense traits, we collected five replicate trees over the distributional range of each species across the East Asian islands, including the Ryukyu Islands. The latitudinal range of samplings was from 24°N to 45°N. Four shoots with leaves were collected from each five-tree, and average values of each tree individual were calculated (Shiono et al. 2015). In the field, we harvested shoots with healthy and fully mature leaves. In the laboratory, we visually inspected the collected shoots to exclude immature and senescent leaves and carefully selected the samples for chemical analysis. LDMC and leaf thickness were measured following the protocols of Cornelissen et al. (2003). Leaf mechanical strength was measured using a “penetrometer” (Feeny 1970). Although Aranwela et al. (1999) showed the bias of using different area of fracture surface of punching rod (Onoda et al. 2011), in the present study, all the mechanical strength of leaves was measured by the standardized penetrometer made by M. Murakami. The diameter of steel punching rod was 3 mm, thus the area of fracture surface was 7.07 mm2 and punch perimeter was 9.42 mm. A proanthocyanidin assay was performed to determine tannin concentrations, using a commercially available quebracho powder as the standard (Julkunen-Tiitto 1985). The Folin–Ciocalteu method was used to determine the total phenol content (Waterman and Mole 1994) with a tannic acid (Wako Co. Ltd., Tokyo, Japan) serving as the standard. The distribution data for woody plant species were collected from botanical literature on the flora of Japan. Data collection methods are described in detail in Kubota et al. (2015).
Statistical analysis
The relationships between leaf defense traits and the distributional range of each plant species were examined for BL tree species as a whole, and separately for DB and EGB tree species. The average values of five replicates for each tree species were used for the following analyses. Although intraspecific variations in leaf defense traits, and even intraspecific gradients in these traits along latitude, were reported (Moles et al. 2013), the shortage of within species replicates prevent us to examine these trends. To assess multivariate relationships among leaf defense traits, we performed principal component analysis (PCA) which deals with collinearity among multiple leaf traits (Pearse and Hipp 2012). Then, we developed full SEM model a priori based on the latitudinal effects on all the leaf defense traits and possible correlations among the traits (Fig. 1). The fit of SEM was assessed by a X 2 goodness-of-fit test of the model, the root mean square error of approximation (RMSEA) and the comparative fit index (CFI). A satisfactory model fit was indicated by: (1) a non-significant X 2 goodness-of-fit test (α = 0.05), (2) CFI > 0.9, and (3) lower 90% confidence intervals (CIs) of RMSEA < 0.05 (Zhang et al. 2013, Blackburn et al. 2016). Based on species distributional data, we analyzed the relationships between latitude (1° intervals) and the trait values of species distributed at the focal latitudes. The integer values of latitude at the lower limits was given for the all analysis. All variables used in path analyses were standardized (Legendre and Legendre 1998) by subtracting the mean and dividing by the standard deviation.
The distribution of leaf defense traits across latitude for deciduous and evergreen broad–leaved tree species. The signs (+, −) after the trait codes show the direction of latitudinal trends in the path analysis (Fig. 2), respectively. LDMC shows leaf dry mass content, and LMA shows leaf mass per area
All analyses were conducted in the R 3.3.2 statistical platform (R Development Core Team 2016). The SEMs were calculated with the R package “lavaan 0.5–16” (Rosseel, 2012), and the packages “stats” and “MASS” (R Development Core Team 2016) were used for PCA and other analyses.
Results
Relationships among functional leaf traits
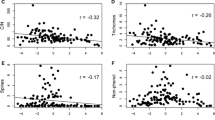
Among the BL tree species overall, the PCA results showed that 31% of the total variance was explained by the first axis and 27% was explained by the second axis. In a separate PCA of DB species, 31% of the total variance was explained by the first axis, which distinguished species with low LMA values from those with high LDMC values. The second axis, which distinguished species with high mechanical defense trait values from those with high tannin and phenol content values, explained 27% of the variation. The PCA of EGB species showed that 29% of the total variance was explained by the first axis, which separated species with low LMA values from those with high leaf mechanical strength (Fig. S1, Table 1). The second axis, which ordinated species with high chemical defense trait values and those with low mechanical defense trait values, explained 27% of the variation. The vectors for chemical and mechanical defense traits were at right angles to each other for DB and EGB tree species, indicating the independence of those variables.
Path analysis
The effects of latitude on leaf traits contrasted markedly between DB and EGB species (Figs. 2, 3). Positive latitudinal effects on tannin and phenol content were detected in DB species, whereas a negative effect on tannin and no effect on phenol was detected in EGB species. We also observed contrasting latitudinal effects on leaf mechanical strength and thickness; these effects were negative for DB species and positive in leaf mechanical strength and no effect in leaf thickness for EGB species. Effects on LDMC were positive in DB and EGB species.
Results of a structural equation model (SEM) depicting hypothesized causal relationships among leaf defense traits and the effects of latitude on them. The positive effects or interactions are indicated by solid lines, while the negative effects or interactions are indicated by broken lines. The dashed lines show the non–significant paths. The deciduous (DB) and evergreen (EGB) tree species were separately examined. LDMC shows leaf dry mass content, and LMA shows leaf mass per area. Standardized coefficients are provided for each path with significant (P < 0.05) effect. For DB; df = 1, P value (X 2) = 0.150 (indicating close model-data fit). For EGB; df = 2, P value (X 2) = 0.249 (again indicating close model-data fit)
Schematic relationships of latitudinal gradients of leaf longevity, chemical defense, and physical defense between deciduous (DB, dashed lines) and evergreen (EGB, dotted lines) broad leaved trees species. The pattern shown in the panel for leaf longevity was derived from the Kikuzawa et al. (2013)
Upon removal of the effects of latitude on individual leaf traits, the interactive correlations among leaf traits were fairly similar in DB and EGB species (Fig. 2). These trends were consistent even when the phylogenetic bias was removed using phylogenetic independent contrasts (PICs; Table S1; Felsenstein 1985).
Discussion
We found significant latitudinal gradients in leaf defense traits in both EGB and DB tree species. However, EGB and DB tree species demonstrated contrasting latitudinal gradients in mechanical and chemical defense traits. Among the mechanical defense traits, EGB species showed increasing trends in leaf mechanical strength and thickness along the latitudinal gradient, with tougher and thicker leaves occurring at higher latitudes. DB species showed the opposite trend, with softer and thinner leaves occurring at higher latitudes. Although Onoda et al. (2011) also observed the positive trends in leaf mechanical strength along latitude for the woody species, the present analysis on EGB and DB tree species showed contrasting trends between them. Among the chemical defense traits, EGB species exhibited a decreasing trend along the latitudinal gradient in tannin content, whereas DB species showed increasing trends with latitude in tannin and phenol content. These results indicate greater chemical defenses at lower latitudes in EGB species and greater physical defenses at lower latitudes in DB species. Thus, the LLHD hypothesis should accommodate a paradoxical aspect in the latitudinal gradients in chemical and physical defense traits across DB and EGB tree species.
One possible explanation for these trends involves trade–offs between leaf defense traits along latitudinal gradients, which would limit total costs of defense against herbivores and might lead to contrasting patterns in latitudinal trends in chemical and mechanical defense traits (Eichenberg et al. 2015). In the present study, many pairs of leaf defense traits showed significant positive correlations in path analyses (Fig. 2), PCA (Fig. S1) and PIC (Table S1); these correlations were detected more frequently than in Moles et al. (2013) who observed them in only three of 45 pairwise comparisons. Moles et al. (2013) argued that this low incidence of significant correlations in pairwise comparisons might partly be explained by the bias in the measurement of defense traits, but not the allocation of resources that might drive trade–offs, in the majority of included studies. However, the present results show much clearer trends in correlations among leaf defense traits, likely due to the separate analyses of DB and EGB tree species and the comprehensive dataset of leaf defense traits obtained by thorough sampling (see “Discussion” in Moles et al. 2013).
One possible explanation for the contrasting trends in physical and chemical defense traits shown in both DB and EGB tree species is “trade-off” between these traits. Classical examinations on defense strategies considered defenses as singleton strategies, and assumed the trade–offs among different antiherbivore strategies (Steward and Keeler 1988; Herms and Mattson 1992). However, in the present study, we can observe the independent trends between physical and chemical leaf defense traits in PCA (Fig. S1, Table 1). These suggested that the contrasting trends of these leaf traits along the latitude were not trade-off but independent responses between physical and chemical leaf defense traits. These paradoxical trends in defense strategies observed in DB and EGB tree species could be explained by trends in leaf longevity along temperature gradients. Wright et al. (2005) and Kikuzawa et al. (2013) found decreasing trends in leaf longevity among EGB tree species along a temperature gradient, and opposing trends in DB tree species (Fig. 1). They also detected a positive correlation between leaf longevity and leaf mass per area, which is generally correlated positively with leaf thickness, a surrogate for physical defense. Thus, EGB-specific higher physical defense (or DB-specific lower physical defense) at higher latitudes likely is a by-product of or reflects a correlation with the leaf longevity gradient along the temperature gradient (Fig. 1). Latitudinal gradients in leaf defense traits could be driven not only by herbivory, but also by abiotic conditions, e.g., soil fertility or UV radiations (Moles et al. 2011). Although this study examined multiple defense traits simultaneously across a wide range of species, broader and more consistent measurements of plant functional traits may be needed to obtain a better understanding of plant defense strategies.
Furthermore, the correlative patterns among leaf defense traits were consistent between DB and EGB tree species when the covariate effect of latitude was removed by path analysis (Fig. 2). These results suggest the existence of a fixed core structure in multiple defense traits. LMA showed consistent positive correlations with other leaf defense traits, suggesting that it has a defensive function or just correlate with them. A leaf economics spectrum study indicated the existence of exploitative plant species, with fast growth associated with thinner leaves and lower LMA (Wright et al. 2004). Thus, species with lower LMA may abandon this trait to defend themselves from herbivores, and grow faster to escape herbivory. Conversely, LDMC showed consistent positive correlations with other leaf defense traits, suggesting that higher LDMC is a surrogate for a higher defense strategy in these species. Many previous studies have shown that LDMC is a surrogate for a variety of leaf properties (e.g., Shipley et al. 2006); higher LDMC is correlated with lower water content (Niinemets 2001) and lower soil fertility (Rusch et al. 2009). Positive correlations of LDMC with plant mechanical strength and resistance to biotic agents have also been observed (Chave et al. 2009, Preston et al. 2006). These showed the common core structures in defense traits of DB and EGB tree species and also specific responses to environmental gradients between these tree species.
Based on the examination of a comprehensive dataset of BL from East Asian flora, we discovered complex latitudinal trends in leaf defense traits involving higher chemical defense at lower latitudes for EGB species and higher physical defense at lower latitudes for DB species. In a meta–analysis derived from various empirical studies of leaf traits across functional types (e.g., tree, shrub, and herbaceous species) and in different regions, Moles et al. (2011) reported no such trend. Our evaluation of LLHD hypothesis that was characterized by a paradoxical aspect in chemical and physical defense traits suggests the importance of more detailed exploration of separate plant functional groups to test this hypothesis.
Change history
25 July 2018
The article “A paradox of latitudinal leaf defense strategies in deciduous and evergreen broadleaved trees”, written by Saihanna Saihanna, Tomoe Tanaka, Yu Okamura, Buntarou Kusumoto, Takayuki Shiono, Toshihide Hirao, Yasuhiro Kubota, and Masashi Murakami, was originally published electronically on the publisher’s internet portal (currently SpringerLink) on 01 June 2018 without open access.
References
Agrawal AA, Fishbein M (2006) Plant defense syndromes. Ecology 87:s132–s149
Anstett DN, Nunes KA, Baskett C, Kotanen PM (2016) Sources of controversy surrounding latitudinal patterns in herbivory and defense. Trends Ecol Evol 31:789–802
Aranwela N, Sanson G, Read J (1999) Methods of assessing leaf–fracture properties. The New Phytol 144:369–383
Blackburn TM, Delean S, Pyšek P, Cassey P (2016) On the island biogeography of aliens: a global analysis of the richness of plant and bird species on oceanic islands. Global Ecol Biogeogr 25:859–868
Bolser RC, Hay ME (1996) Are tropical plants better defended? palatability and defenses of temperate vs. tropical seaweeds. Ecology 77:2269–2286
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12:351–366
Coley PD, Aide TM (1991) A comparison of herbivory and plant defenses in temperate and tropical broad–leaved forests. In: Price PW, Lewinsohn TM, Fernandes GW, Benson WW (eds) Plant–animal interactions: evolutionary ecology in tropical and temperate regions. Wiley, New York, pp 25–49
Cornelissen JHC, Lavorel S, Garnier E, Díaz S, Buchmann N, Gurvich DE, Reich PB, ter Steege H, Morgan HD, van der Heijden MGA, Pausas JG, Poorter H (2003) A handbook of protocols for standardized and easy measurement of plant functional traits worldwide. Australian J Bot 51:335–380
Development Core Team R (2016) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna
Dobzhansky T (1950) Evolution in the Tropics. Am Sci 38:209–221
Eichenberg D, Purschke O, Ristok C, Wessjohann L, Bruelheide H (2015) Trade–offs between physical and chemical carbon-based leaf defense: of intraspecific variation and trait evolution. J Ecol 103:1667–1679
Feeny P (1970) Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth caterpillars. Ecology 51:565–581
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Herms DA, Mattson WJ (1992) The dilemma of plants: to grow or defend. Quat Rev Biol 67:283–335
Julkunen-Tiitto R (1985) Phenolic constituents in the leaves of northern willows: methods for the analysis of certain phenolics. J Agr Food Chem 33:213–217
Kattge J, Diaz S, Lavorel S, Prentice IC, Leadley P, Bönisch G, Garnier E, Westoby M, Reich PB, Wright IJ, Cornelissen JHC, Violle C, Harrison SP, van Bodegom PM, Reichstein M, Enquist BJ, Soudzilovskaia NA, Ackerly DD, Anand M, Atkin O, Bahn M, Baker TR, Baldocchi D, Bekker R, Blanco C, Blonder B, Bond WJ, Bradstock R, Bunker DE, Casanoves F, Cavender-Bares J, Chambers JQ, Chapin FS, Chave J, Coomes D, Cornwell WK, Craine JM, Dobrin BH, Duarte L, Durka W, Elser J, Esser G, Estiarte M, Fagan WF, Fang J, FernándezMéndez F, Fidelis A, Finegan B, Flores O, Ford H, Frank D, Freschet GT, Fyllas NM, Gallagher RV, Green WA, Gutierrez AG, Hickler T, Higgins S, Hodgson JG, Jalili A, Jansen S, Joly C, Kerkhoff AJ, Kirkup D, Kitajima K, Kleyer M, Klotz S, Knops JMH, Kramer K, Kühn I, Kurokawa H, Laughlin D, Lee TD, Leishman M, Lens F, Lenz T, Lewis SL, Lloyd J, Llusiá J, Louault F, Ma S, Mahecha MD, Manning P, Massad T, Medlyn B, Messier J, Moles AT, Müller SC, Nadrowski K, Naeem S, Niinemets Ü, Nöllert S, Nüske A, Ogaya R, Oleksyn J, Onipchenko VJ, Onoda Y, Ordoñez J, Overbeck G, Ozinga WA, Patiño S, Paula S, Pausas JG, Peñuelas J, Phillips OL, Pillar V, Poorter H, Poorter L, Poschlod P, Prinzing A, Proulx R, Rammig A, Reinsch S, Reu B, Sack L, Salgado-Negret B, Sardans J, Shioder S, Shipley B, Siefert A, Sosinski E, Soussana JF, Swaine E, Swenson N, Thompson K, Thornton P, Waldram M, Weiher E, White M, White S, Wright SJ, Yguel B, Zaehle S, Zanne AE, Wirth C (2011) TRY–a global database of plant traits. Global Chang Biol 17:2905–2935
Kikuzawa K, Onoda Y, Wright IJ, Reich PB (2013) Mechanisms underlying global temperature-related patterns in leaf longevity. Global Ecol Biogeogr 22:982–993
Kubota Y, Shiono T, Kusumoto B (2015) Role of climate and geohistorical factors in driving plant richness patterns and endemicity on the east Asian continental islands. Ecography 38:639–648
Kusumoto B, Shiono T, Miyoshi M, Maeshiro R, Fujii S, Kuuluvainen T, Kubota Y (2014) Functional response of plant communities to clearcutting: management impacts differ between forest vegetation zones. J Appl Ecol 52:171–180
Legendre P, Legendre L (1998) Numerical ecology. Second English edition. Elsevier Science BV, Amsterdam
Levin DA, York BM (1978) The toxicity of plant alkaloids: an ecogeographic perspective. Biochem Syst Ecol 6:61–76
Lewinsohn TM, Roslin T (2008) Four ways towards tropical herbivore megadiversity. Ecol Lett 11:398–416
MacArthur R (1972) Geographical ecology: patterns in the distribution of species. Harper and Rowe, NY
Maeshiro R, Kusumoto B, Fujii SJ, Shiono T, Kubota Y (2013) Using tree functional diversity to evaluate management impacts in a subtropical forest. Ecosphere 4: art70
Marquis RJ, Ricklefs RE, Abdala-Roberts L (2012) Testing the low latitude/high defense hypothesis for broad-leaved tree species. Oecologia 169:811–820
Moles AT, Bonser SP, Poore AGB, Wallis IR, Foley WJ (2011) Assessing the evidence for latitudinal gradients in plant defense and herbivory. Funct Ecol 25:380–388
Moles AT, Wallis IR, Foley WJ, Warton DI, Stegen JC, Bisigato AJ, Cella-Pizarro L, Clark CJ, Cohen PS, Cornwell WK, Edwards W, Ejrnæs R, Gonzales-Ojeda T, Graae BJ, Hay G, Lumbwe FC, Magaña-Rodríguez B, Moore BD, Peri PL, Poulsen JR, Veldtman R, von Zeipel H, Andrew NR, Boulter SL, Borer ET, Campón FF, Coll M, Farji-Brener AG, De Gabriel J, Jurado E, Kyhn LA, Low B, Mulder CP, Reardon-Smith K, Rodríguez-Velázquez J, Seabloom EW, Vesk PA, van Cauter A, Waldram MS, Zheng Z, Blendinger PG, Enquist BJ, Facelli JM, Knight T, Majer JD, Martínez-Ramos M, McQuillan P, Prior LD (2013) Putting plant resistance traits on the map: a test of the idea that plants are better defended at lower latitudes. New Phytol 191:777–788
Niinemets U (2001) Global–scale climatic controls of leaf dry mass per area, density, and thickness in trees and shrubs. Ecology 82:453–469
Onoda Y et al (2011) Global patterns of leaf mechanical properties. Ecol Lett 14:301–312
Pearse IS, Hipp AL (2012) Global patterns of leaf defenses in oak species. Evolution 66:2272–2286
Pennings SC, Silliman BR (2005) Linking biogeography and community ecology: latitudinal variation in plant–herbivore interaction strength. Ecology 86:2310–2319
Preston KA, Cornwell WK, Denoyer JL (2006) Wood density and vessel traits as distinct correlates of ecological strategy in 51 California coast range angiosperms. New Phytol 170:807–818
Price PW (2002) Resource-driven terrestrial interaction webs. Ecol Res 17:241–247
Rosseel Y (2012) Lavaan: an R package for structural equation modeling. J Stat Softw 48:1–36
Rusch GM, Skarpe C, Halley DJ (2009) Plant traits link hypothesis about resource—use and response to herbivory. Basic Appl Ecol 10:466–474
Schmitt TM, Hay ME, Lindquist N (1995) Constraints on chemically mediated coevolution: multiple functions for seaweed secondary metabolites. Ecology 76:107–123
Shiono T, Kusumoto B, Maeshiro R, Fujii SJ, Götzenberger L, Bello F, Kubota Y (2015) Climatic drivers of trait assembly in woody plants in Japan. J Biogeogr 42:1176–1186
Shipley B, Lechowicz MJ, Wright I, Reich PB (2006) Fundamental trade-offs generating the worldwide leaf economics spectrum. Ecology 87:535–541
Sitch S, Smith B, Prentice IC, Arneth A, Bondeau A, Cramer W, Kaplan JO, Levis S, Lucht W, Sykes MT, Thonicke K, Venevsky S (2003) Evaluation of ecosystem dynamics, plant geography and terrestrial carbon cycling in the LPJ dynamic global vegetation model. Global Chang Biol 9:161–185
Steward JL, Keeler KH (1988) Are there trade-offs among antiherbivore defenses in Ipomoea (Convolvulaceae)? Oikos 53:79–86
Waterman PG, Mole S (1994) Extraction and chemical quantification. In: Waterman PG, Mole S (eds) Analysis of phenolic plant metabolites. Blackwell Science, Oxford, pp 66–103
Wright IJ, Reich PB, Westoby M, Ackerly DD (2004) The worldwide leaf economics spectrum. Nature 428:821–827
Wright IJ, Reich PB, Cornelissen JHC, Falster DS, Groom PK, Hikosaka K, Lee W, Lusk CH, Niinemets Ü, Oleksyn J, Osada N, Poorter H, Warton DI, Westoby M (2005) Modulation of leaf economic traits and trait relationships by climate. Global Ecol Biogeogr 14:411–421
Zhang J, Kissling DW, He F (2013) Local forest structure, climate and human disturbance determine regional distribution of boreal bird species richness in Alberta, Canada. J Biogeogr 40:1131–1142
Acknowledgements
We are grateful to the two anonymous reviewers for helpful comments. This study was supported by the Japan Society for the Promotion of Science (no. 22405006, no. 15H04424, no.15K16153, no. 15K14607) and Program for Advancing Strategic International Networks to Accelerate the Circulation of Talented Researchers, the Japan Society for the Promotion of Science to YK.
Author information
Authors and Affiliations
Corresponding authors
Additional information
The original version of this article was revised due to a retrospective Open Access order.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Saihanna, S., Tanaka, T., Okamura, Y. et al. A paradox of latitudinal leaf defense strategies in deciduous and evergreen broadleaved trees. Ecol Res 33, 1011–1017 (2018). https://doi.org/10.1007/s11284-018-1614-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-018-1614-0