Abstract
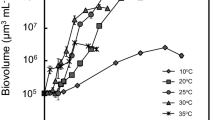
The dependence of competitive interactions on abiotic conditions is attracting increasing interest in the face of globally rising temperatures and altered biogeochemical cycles of major nutrients. In a microcosm experiment involving a natural inoculum of benthic microalgae, temperature and nutrient supply ratios were manipulated in order to test three main hypotheses: (1) temperature and nutrient supply ratios determine species composition and diversity of the assemblage, (2) the identity of the dominating species depends on nutrient supply and temperature, and (3) higher temperature leads to faster competitive exclusion and thus more rapid decline in species richness. Over a period of 7 weeks, algal biomass reached an equilibrium carrying capacity, with was higher at colder temperatures and intermediate N:P supply ratios (N:P = 16). Initial growth rate increased with temperature and under high P-supply. Species richness in the stationary phase of the experiment decreased with increasing temperature, reflecting a higher extinction rate in the warmer treatments, which were also characterized by higher dominance of single species. Thus, increasing temperature both altered the identity of the dominating species and accelerated competitive displacement. This experiment thus indicates that warming might influence outcome and temporal dynamics in species interactions, and thereby eventually local diversity.




Similar content being viewed by others
References
Admiraal W, Peletier H, Brouwer T (1984) The seasonal succession patterns of diatom species on an intertidal mudflat: an experimental analysis. Oikos 42:30–40
Bertness MD, Ewanchuk PJ (2002) Latitudinal and climate-driven variation in the strength and nature of biological interactions in New England salt marshes. Oecologia 132:392–401
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB (2004) Toward a metabolic theory of ecology. Ecology 85:1771–1789
Brzezinski MA (1985) The Si:C:N ratio of marine diatoms: interspecific variability and the effect of some environmental variables. J Phycol 21:347–357
Burgmer T, Hillebrand H (2011) Temperature mean and variance alter phytoplankton biomass and biodiversity in a long-term microcosm experiment. Oikos doi: 10.1111/j.1600-0706.2010.19301.x
Callaway RM, Brooker RW, Choler P, Kikvidze Z, Lortie CJ, Michalet R, Paolini L, Pugnaire FL, Newingham B, Aschehoug ET, Armas C, Kikodze D, Cook BJ (2002) Positive interactions among alpine plants increase with stress. Nature 417:844–848
Chesson P, Huntly N (1997) The roles of harsh and fluctuating conditions in the dynamics of ecological communities. Am Nat 150:519–553
DeNicola DM (1996) Periphyton responses to temperature at different ecological levels. In: Stevenson RJ, Bothwell ML, Lowe RL (eds) Algal ecology—freshwater benthic ecosystems, Academic, London, pp 149–181
Edwards M, Richardson AJ (2004) Impact of climate change on marine pelagic phenology and trophic mismatch. Nature 430:881–884
Fong P, Zedler JB (1993) Temperature and light effects on the seasonal succession of algal communities in shallow coastal lagoons. J Exp Mar Biol Ecol 171:259–272
Fujimoto N, Sudo R, Sugiura N, Inamori Y (1997) Nutrient limited growth of Microcystis aeruginosa and Phormidium tenue and competition under various N:P supply ratios and temperatures. Limnol Oceanogr 42:250–256
Hillebrand H, Sommer U (1997) Response of epilithic microphytobenthos of the western baltic sea to in situ experiments with nutrient enrichment. Mar Ecol Prog Ser 160:35–46
Hillebrand H, Sommer U (1999) The nutrient stoichiometry of benthic microalgal growth: redfield proportions are optimal. Limnol Oceanogr 44:440–446
Hillebrand H, Sommer U (2000) Effect of continuous nutrient enrichment on microalgae colonizing hard substrates. Hydrobiologia 426:185–192
Hillebrand H, Duerselen CD, Kirschtel DB, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J Phycol 35:403–424
Hillebrand H et al (2007) Consumer versus resource control of producer diversity depends on ecosystem type and producer community structure. Proc Natl Acad Sci USA 104:10904–10909
Hillebrand H, Borer ET, Bracken MES, Cardinale BJ, Cebrian J, Cleland EE, Elser JJ, Gruner DS, Harpole WS, Ngai JT et al (2009) Herbivore metabolism and stoichiometry each constrain herbivory at different organizational scales across ecosystems. Ecol Lett 12:516–527
Hobbie SE, Shevtsova A, Chapin FS (1999) Plant response to species removal and experimental warming in Alaskan tussock tundra. Oikos 84:417–434
Jöhnk KD, Huisman J, Sharples J, Sommeijer B, Visser PM, Stroom JM (2008) Summer heatwaves promote blooms of harmful cyanobacteria. Glob Chang Biol 14:495–512
Klanderud K, Totland O (2007) The relative role of dispersal and local interactions for alpine plant community diversity under simulated climate warming. Oikos 116:1279–1288
O’Connor MI, Piehler MF, Leech DM, Anton A, Bruno JF (2009) Warming and resource availability shift food web structure and metabolism. PLoS Biol 7(8):e1000178. doi:10.1371/journal.pbio.1000178
Parmesan C (2006) Ecological and evolutionary responses to recent climate change. Annu Rev Ecol Evol Syst 37:637–669
Sommer U (1994a) Are marine diatoms favoured by high si:N ratios? Mar Ecol Prog Ser 115:309–315
Sommer U (1994b) The impact of light intensity and daylength on silicate and nitrate competition among marine phytoplankton. Limnol Oceanogr 39:1680–1688
Sommer U (1996) Nutrient competition experiments with periphyton from the Baltic Sea. Mar Ecol Prog Ser 140:161–167
Sommer U, Lengfellner K (2008) Climate change and the timing, magnitude, and composition of the phytoplankton spring bloom. Glob Chang Biol 14:1199–1208
Tilman D, Kiesling R, Sterner R, Kilham SS, Johnson FA (1986) Green, bluegreen and diatom algae: taxonomic differences in competitive ability for phosphorus, silicon and nitrogen. Arch Hydrobiol 106:473–485
Tirok K, Gaedke U (2007) The effect of irradiance, vertical mixing and temperature on spring phytoplankton dynamics under climate change: long-term observations and model analysis. Oecologia 150:625–642
Tylianakis JM, Didham RK, Bascompte J, Wardle DA (2008) Global change and species interactions in terrestrial ecosystems. Ecol Lett 11:1351–1363
Vijverberg J, Vos M (2006) Predator-released compounds, ambient temperature and 5 competitive exclusion among differently sized Daphnia species. Freshw Biol 51:756–767
Walker MD, Wahren CH, Hollister RD, Henry GHR, Ahlquist LE, Alatalo JM, Bret-Harte MS, Calef MP, Callaghan TV, Carroll AB, Epstein HE, Jonsdottir IS, Klein JA, Magnusson B, Molau U, Oberbauer SF, Rewa SP, Robinson CH, Shaver GR, Suding KN, Thompson CC, Tolvanen A, Totland O, Turner PL, Tweedie CE, Webber PJ, Wookey PA (2006) Plant community responses to experimental warming across the tundra biome. Proc Natl Acad Sci USA 103:1342–1346
Watermann F, Hillebrand H, Gerdes G, Krumbein WE, Sommer U (1999) Competition between benthic cyanobacteria and diatoms as influenced by different grain sizes and temperatures. Mar Ecol Prog Ser 187:77–87
Acknowledgments
This paper is a reassessment of data obtained from an experiment in a project funded by the German Science Foundation (DFG So145/15-1). The new analysis is based on ideas developed during the Aquashift priority program funded by the DFG (DFG Hi 848/3-2).
Author information
Authors and Affiliations
Corresponding author
Appendix 1
Appendix 1
Species found in the inocula of the two experiments are listed. Besides the species names and classes (BAC Bacillariphyceae, RHO Rhodophyceae, CYA Cyanobacteria), the percentage contribution to the inocula is given (r rare, <0.1% of inoculums biovolume).
Species | Class | % |
|---|---|---|
Achnanthesbrevipes C.A. Ag. | BAC | r |
Achnantheslongipes C.A. Ag. | BAC | r |
Aglaothamnionbyssoides (Arn.) L’Hardy-Halos & Rueness | RHO | r |
Amphoracoffeaeformis (C.A. Ag.) Kützing | BAC | 0.2 |
Asterionellaformosa Hassall | BAC | 0.2 |
Berkeleyarutilans (Trentepohl) Grunow | BAC | 0.5 |
Chaetocerossimplex Ostenfeld | BAC | r |
Cocconeisscutellum Ehrenberg | BAC | r |
Cylindrothecaclosterium (Ehrenberg) Reiman & Lewin | BAC | r |
Cymbellapusilla Kützing | BAC | r |
Diatomavulgare Bory | BAC | r |
Hasleacrucigera (W. Smith) Simonsen | BAC | r |
Licmophorahyalina (Kützing) Grunow | BAC | r |
Licmophoraparadoxa (Lyngbye) C.A. Ag. | BAC | r |
Melosiramoniliformis (O.F. Müller) C.A. Ag. | BAC | 73.7 |
Melosiranummuloides C.A. Ag. | BAC | 13.0 |
Navicula cf. cincta (Ehrenberg) Ralfs | BAC | 1.7 |
Navicula cf. perminuta Grunow | BAC | 1.3 |
Navicularadiosa Kützing | BAC | r |
Nitzschiacapitellata Hustedt | BAC | 0.5 |
Nitzschiagracilis Hantzsch | BAC | r |
Nitzschiamicrocephala Grunow | BAC | r |
Nitzschiasigma (Kützing) W. Smith | BAC | r |
Odontellaaurita (Lyngbye) C.A. Ag. | BAC | 8.7 |
Pseudanabaena sp. | CYA | r |
Skeletonemacostatum (Greville) Cleve | BAC | r |
Spirulinasubsalsa Oersted | CYA | r |
Stauroneis simulans (Donkin) Ross | BAC | r |
Tabulariafasciculata (C.A. Ag.) Williams & Round | BAC | r |
Tryblionellaapiculata Gregory | BAC | r |
About this article
Cite this article
Hillebrand, H. Temperature mediates competitive exclusion and diversity in benthic microalgae under different N:P stoichiometry. Ecol Res 26, 533–539 (2011). https://doi.org/10.1007/s11284-011-0810-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-011-0810-y