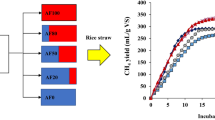
Abstract
Biomass from agriculture, forestry, and urban wastes is a potential renewable organic resource for energy generation. Many investigations have demonstrated that anaerobic fungi and methanogens could be co-cultured to degrade lignocellulose for methane generation. Thus, this study aimed to evaluate the effect of natural anaerobic fungi-methanogens co-culture on the methane production and lignocellulosic degradation of wastes from rice, corn and sugarcane. Hu sheep rumen digesta was used to develop a natural anaerobic fungi-methanogen co-culture. The substrates were rice straw (RS), rich husk (RH), corn stover (CS), corn cobs (CC), and sugarcane baggage (SB). Production of total gas and methane, metabolization rate of reducing sugar, glucose, and xylose, digestibility of hemicellulose and cellulose, activity of carboxymethylcellulase and xylanase, and concentrations of total acid and acetate were highest (P < 0.05) in CC, moderate (P < 0.05) in RS and CS, and lowest (P < 0.05) in SB and RH. The pH, lactate and ethanol were lowest (P < 0.05) in CC, moderate (P < 0.05) in RS and CS, and lowest (P < 0.05) SB and RH. Formate was lowest (P < 0.05) in CC, RS and CS, moderate (P < 0.05) in SB, and lowest (P < 0.05) in RH. Therefore, this study indicated that the potential of methane production and lignocellulosic degradation by natural anaerobic fungi-methanogens co-culture were highest in CC, moderate in RS and CS, and lowest in SB and RH.
Graphical abstract








Similar content being viewed by others
Data availability
The data used in the current research are available upon reasonable request from the corresponding author.
References
Abraham A, Mathew AK, Sindhu R, Pandey A, Binod P (2016) Potential of rice straw for bio-refining: an overview. Bioresour Technol 215:29–36. https://doi.org/10.1016/j.biortech.2016.04.011
Amin FR, Khalid H, Zhang H, Rahman SU, Zhang R, Liu G, Chen C (2017) Pretreatment methods of lignocellulosic biomass for anaerobic digestion. AMB Express 7:72. https://doi.org/10.1186/s13568-017-0375-4
AOAC (1990) Official methods of analysis. 15th Edition, Association of official analytical chemist, Washington DC.
Boxma B, Voncken F, Jannink S, van Alen T, Akhmanova A, van Weelden SW, van Hellemond JJ, Ricard G, Huynen M, Tielens AGM, Hackstein JHP (2004) The anaerobic chytridiomycete fungus Piromyces sp. E2 produces ethanol via pyruvate:formate lyase and an alcohol dehydrogenase E. Mol Microbiol 51:1389–1399. https://doi.org/10.1046/j.1365-2958.2003.03912.x
Cao G, Zhang X, Wang D, Zhang X (2005) Inventory of emissions of pollutants from open burning crop residue. J Agro-Environ Sci 24:800–804
Cheng Y, Zhu W (2009) Diversity analysis of anaerobic fungi in the co-cultures with or without methanogens by amplified ribosomal intergenic spacer analysis. Wei Sheng Wu Xue Bao 49:0504–0511
Cheng YF, Edwards JE, Allison GG, Zhu WY, Theodorou MK (2009) Diversity and activity of enriched ruminal cultures of anaerobic fungi and methanogens grown together on lignocellulose in consecutive batch culture. Bioresour Technol 100:4821–4828. https://doi.org/10.1016/j.biortech.2009.04.031
Cheng YF, Jin W, Mao SY, Zhu WY (2013) Production of citrate by anaerobic fungi in the presence of co-culture methanogens as revealed by 1H NMR spectrometry. Asian-Australas J Anim Sci 26:1416–1423. https://doi.org/10.5713/ajas.2013.13134
Cheng YF, Shi Q, Sun R, Liang D, Li YF, Li Y, Jin W, Zhu WY (2018) The biotechnological potential of anaerobic fungi on fiber degradation and methane production. World J Microbiol Biotechnol 34:155. https://doi.org/10.1007/s11274-018-2539-z
Edgardo A, Carolina P, Manuel R, Juanita F, Baeza J (2008) Selection of thermotolerant yeast strains Saccharomyces cerevisiae for bioethanol production. Enzyme Microb Technol 43:120–123. https://doi.org/10.1016/j.enzmictec.2008.02.007
FAOSTAT (2018) Food and Agriculture Organization of the United Nations. Retrieved on 29 December 2023 from https://www.fao.org/faostat/en/#data/QV
Frei M (2013) Lignin: characterization of a multifaceted crop component. Sci World J 436517. https://doi.org/10.1155/2013/436517
Gruber L (2009) Chemical composition, analyses and relevance of plant cell walls in the nutrition of ruminant livestock. Uebers Tierernaehr 37:45–86
Haitjema CH, Solomon KV, Henske JK, Theodorou MK, O’Malley MA (2014) Anaerobic gut fungi: advances in isolation, culture, and cellulolytic enzyme discovery for biofuel production. Biotechnol Bioeng 111:1471–1482. https://doi.org/10.1002/bit.25264
Hu WL, Wang JK, Lv JM, Guo YQ, Liu JX (2006) Rapid gas chromatogram determination of methane, organic acid in in vitro ruminal fermentation products. J Zhejiang Uni Agri Life Sci 32:217–221
Jin W (2009) Isolation and identification of anaerobic fungi and their associated mehanogens from herbivorous animals and their characteristics of in vitro fermentation. Nanjing Agri Uni
Jin W, Cheng YF, Mao SY, Zhu WY (2011) Isolation of natural cultures of anaerobic fungi and indigenously associated methanogens from herbivores and their bioconversion of lignocellulosic materials to methane. Bioresour Technol 102:7925–7931. https://doi.org/10.1016/j.biortech.2011.06.026
Khelaifia S, Drancourt M (2012) Susceptibility of archaea to antimicrobial agents: applications to clinical microbiology. Clin Microbiol Infect 18:841–848. https://doi.org/10.1111/j.1469-0691.2012.03913.x
Kumar P, Barrett DM, Delwiche MJ, Stroeve P (2009) Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind Eng Chem Res 48:3713–3729. https://doi.org/10.1021/ie801542g
Li F, Wang J (2013) Estimation of carbon emission from burning and carbon sequestration from biochar producing using crop straw in China. Trans Chin Soc Agri Eng 29:1–7
Li Y, Jin W, Cheng Y, Zhu W (2016) Effect of the associated methanogen methanobrevibacter thaueri on the dynamic profile of end and intermediate metabolites of anaerobic fungus Piromyces sp. F1. Curr Microbiol 73:434–441. https://doi.org/10.1007/s00284-016-1078-9
Li Y, Jin W, Mu C, Cheng Y, Zhu W (2017) Indigenously associated methanogens intensified the metabolism in hydrogenosomes of anaerobic fungi with xylose as substrate. J Basic Microbiol 57:933–940. https://doi.org/10.1002/jobm.201700132
Li Y, Cheng Y, Zhu W (2018) Enhancing the resistance of anaerobic fungus Piromyces sp. F1 to nitrovin by co-culture with Methanobrevibacter Thauer F1. Microbiol Chin 45:111–119. https://doi.org/10.13344/j.microbiol.china.170143
Li Y, Sun M, Li Y, Cheng Y, Zhu W (2020) Co-cultured methanogen improved the metabolism in the hydrogenosome of anaerobic fungus as revealed by GC/MS analysis. Asian-Australas J Anim Sci 33:1948–1956. https://doi.org/10.5713/ajas.19.0649
Li Y, Meng Z, Xu Y, Shi Q, Ma Y, Aung M, Cheng Y, Zhu W (2021) Interactions between anaerobic fungi and methanogens in the rumen and their biotechnological potential in biogas production from lignocellulosic materials. Microorganisms 9:1–17. https://doi.org/10.3390/microorganisms9010190
Lowe SE, Theodorou MK, Trinci APJ (1987a) Growth and fermentation of an anaerobic rumen fungus on various carbon-sources and effect of temperature on development. Appl Environ Microbiol 53:1210–1215. https://doi.org/10.1128/aem.53.6.1210-1215.1987
Lowe SE, Theodorou MK, Trinci AP (1987b) Cellulases and xylanase of an anaerobic rumen fungus grown on wheat straw, wheat straw holocellulose, cellulose, and xylan. Appl Environ Microbiol 53:1216–1223. https://doi.org/10.1128/aem.53.6.1216-1223.1987
Ma J, Zhong P, Li Y, Sun Z, Sun X, Aung M, Hao L, Cheng Y, Zhu W (2022) Hydrogenosome, pairing anaerobic Fungi and H2-Utilizing microorganisms based on metabolic ties to facilitate Biomass utilization. J Fungi 8:338. https://doi.org/10.3390/jof8040338
Martínez-Gutiérrez E (2018) Biogas production from different lignocellulosic biomass sources: advances and perspectives. 3 Biotech 8:233–286. https://doi.org/10.1007/s13205-018-1257-4
Nakashimada Y, Srinivasan K, Nurakami M, Nishio N (2000) Direct conversion of cellulose to methane by anaerobic fungi Neocallimastix frontalis and defined methanogens. Biotechnol Lett 22:223–227. https://doi.org/10.1023/A:1005666428494
Patinvoh RJ, Osadolor OA, Chandolias K, Sárvári HI, Taherzadeh MJ (2017) Innovative pretreatment strategies for biogas production. Bioresour Technol 224:13–24. https://doi.org/10.1016/j.biortech.2016.11.083
Sawatdeenarunat C, Surendra KC, Takara D, Oechsner H, Khanal SK (2015) Anaerobic digestion of lignocellulosic biomass: challenges and opportunities. Bioresour Technol 178:178–186. https://doi.org/10.1016/j.biortech.2014.09.103
Sawatdeenarunat C, Nguyen D, Surendra KC, Shrestha S, Rajendran K, Oechsner H, Khanal SK (2016) Anaerobic biorefinery: current status, challenges, and opportunities. Bioresour Technol 215:304–313. https://doi.org/10.1016/j.biortech.2016.03.074
Silveira RL, Stoyanov SR, Gusarov S, Skaf MS, Kovalenko A (2015) Supramolecular interactions in secondary plant cell walls: effect of lignin chemical composition revealed with the molecular theory of solvation. J Phys Chem Lett 6:206–211. https://doi.org/10.1021/jz502298q
Solomon KV, Haitjema CH, Henske JK, Gilmore SP, Borges-Rivera D, Lipzen A, Brewer HM, Purvine SO, Wright AT, Theodorou MK, Grigoriev IV, Regev A, Thompson DA, O’Malley MA (2016) Early-branching gut fungi possess a large, comprehensive array of biomass-degrading enzymes. Science 351:1192–1195. https://doi.org/10.1126/science.aad1431
Su HF, Liu G, He MX, Tan FR (2015) A biorefining process: sequential, combinational lignocellulose pretreatment procedure for improving biobutanol production from sugarcane bagasse. Bioresour Technol 187:149–160. https://doi.org/10.1016/j.biortech.2015.03.107
Sun YZ, Mao SY, Jie C, Zhou LF, Zhu WY (2005) Effect of substrates with different lignin content on rumen fungal attachment and fermentation in the rumen of goats. Acta Pratac Sci 3:56–61
Thangavelu K, Desikan R, Taran OP, Uthandi S (2018) Delignification of corncob via combined hydrodynamic cavitation and enzymatic pretreatment: process optimization by response surface methodology. Biotechnol Biofuels 11:203. https://doi.org/10.1186/s13068-018-1204-y
Theodorou MK, Williams BA, Dhanoa MS, Mcallan AB, France J (1994) A simple gas-production method using a pressure transducer to determine the fermentation kinetics of ruminant feeds. Anim Feed Sci Technol 48:185–197. https://doi.org/10.1016/0377-8401(94)90171-90176
Theodorou MK, Zhu WY, Rickers A, Nielsen BB, Gull K, Trinci APJ (1996) Biochemistry and ecology of anaerobic fungi. In: Howard DH, Miller JD (eds) The Mycota VI: Human and Animal relationships. Springer-, pp 265–295
Van Soest PJ, Robertson JB, Lewis BA (1991) Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci 74:3583–3597. https://doi.org/10.3168/jds.S0022-0302(91)78551-2
Xu N, Liu S, Xin F, Zhou J, Jia H, Xu J, Jiang M, Dong W (2019) Biomethane production from lignocellulose: Biomass recalcitrance and its impacts on anaerobic digestion. Front Bioeng Biotechnol 7:1–12. https://doi.org/10.3389/fbioe.2019.00191
Yang S, Zhang Y, Yue W, Wang W, Wang YY, Yuan TQ, Sun RC (2016) Valorization of lignin and cellulose in acid-steam-exploded corn stover by a moderate alkaline ethanol post-treatment based on an integrated biorefinery concept. Biotechnol Biofuels 9:238. https://doi.org/10.1186/s13068-016-0656-1
Yu Q, Zhuang X, Wang W, Qi W, Wang Q, Tan X, Kong X, Yuan Z (2016) Hemicellulose and lignin removal to improve the enzymatic digestibility and ethanol production. Biomass Bioenerg 94:105–109. https://doi.org/10.1016/j.biombioe.2016.08.005
Zhu W, Mao S, Wang Q, Wen Y (2001) Study on the screening of anaerobic fungi by in vitro fermentation. J Nanjing Agric Univ 3:44–48
Acknowledgements
We would like to thank Zhenxiang Meng and Yihan Xue for their help when conducting experiments.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Numbers; 3237200386; 32061143034).
Author information
Authors and Affiliations
Contributions
Conceptualization, Writing – original draft: Y.Y.K. and M.A.; Methodology: Y.YK., M.A., Y.X. and Y.L.; investigation: Y.Y.K., M.A., Y.X., Y.Z., Y.L. and Z.S.; formal analysis: Y.Y.K., M.A., Y.X., Y.Z., Y.L. and Z.S.; data curation: Y.Y.K., M.A. and Z.S.; project administration: Y.C.; funding acquisition: W.Z. and Y.C.; Conceptualization, Project administration, Supervision, Writing – review and editing: Y.C. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval
All international, national, and/or institutional animal care and usage criteria were followed.
Competing interests
The authors state that they do not have any competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kyawt, Y.Y., Aung, M., Xu, Y. et al. Methane production and lignocellulosic degradation of wastes from rice, corn and sugarcane by natural anaerobic fungi-methanogens co-culture. World J Microbiol Biotechnol 40, 109 (2024). https://doi.org/10.1007/s11274-024-03938-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-024-03938-8