Abstract
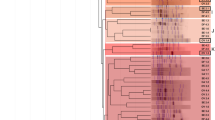
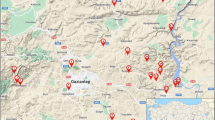
The introduced species Acacia saligna is a very promiscuous host as it can be efficiently nodulated with a wide range diversity of rhizobia taxa, including both fast and slow-growing strains. Fourteen nitrogen (N)-fixing bacteria were isolated from root nodules of wild Acacia saligna growing in distinct geographic locations in Morocco and were examined for their symbiotic efficiency and phenotypic properties. Multivariate tools, such as principal component analysis (PCA) and hierarchical clustering analysis (HCA), were used to study the correlation between phenotypic and symbiotic variables and discriminate and describe the similarities between different isolated bacteria with respect to all the phenotypic and symbiotic variables. Phenotypic characterization showed a variable response to extreme temperature, salinity and soil pH. At the plant level, the nodulation, nitrogen fixation, and the shoot and root dry weights were considered. The obtained results show that some of the tested isolates exhibit remarkable tolerances to the studied abiotic stresses while showing significant N2 fixation, indicating their usefulness as effective candidates for the inoculation of acacia trees. The PCA also allowed showing the isolates groups that present a similarity with evaluated phenotypic and symbiotic parameters. The genotypic identification of N2-fixing bacteria, carried out by the 16S rDNA approach, showed a variable genetic diversity among the 14 identified isolates, and their belonging to three different genera, namely Agrobacterium, Phyllobacterium and Rhizobium.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- CFU:
-
Colony Forming Unit
- HCA:
-
Hierarchical clustering analysis
- HgCl2 :
-
Mercury chloride
- NaCl:
-
Sodium chloride
- PCA:
-
Principal component analysis
- KNO3 :
-
Potassium nitrate
- PCR:
-
Polymerase chain reaction
- PGA:
-
Peptone–glucose–agar
- RDW:
-
Root dry weight
- RE:
-
Relative efficiency
- SDW:
-
Shoot dry weight
- YMA:
-
Yeast, Mannitol Agar
References
Amrani S, Noureddine NE, Bhatnagar T et al (2010) Phenotypic and genotypic characterization of rhizobia associated with Acacia saligna (Labill.) Wendl. in nurseries from Algeria. Syst Appl Microbiol 33:44–51. https://doi.org/10.1016/J.SYAPM.2009.09.003
Arora NK (2018) Bioremediation: a green approach for restoration of polluted ecosystems. Environ Sustain 14(1):305–307. https://doi.org/10.1007/S42398-018-00036-Y
Assefa F (1993) Nodulation and nitrogen fixation by Rhizobium and Bradyrhizobium spp of some indigenous tree legumes of Ethiopia—ERef Bayreuth. Universität Bayreuth, Bayreuth
Ba S, Willems A, De Lajudie P et al (2002) Symbiotic and taxonomic diversity of rhizobia isolated from Acacia tortilis subsp. raddiana in Africa. Syst Appl Microbiol 25:130–145. https://doi.org/10.1078/0723-2020-00091
Bakhoum N, Fall D, Fall F et al (2018) Senegalia senegal (synonym: Acacia senegal), its importance to sub-Saharan Africa, and its relationship with a wide range of symbiotic soil microorganisms. South African J Bot 119:362–368. https://doi.org/10.1016/J.SAJB.2018.10.007
Basu S, Kumar G (2020) Nitrogen fixation in a legume-rhizobium symbiosis: the roots of a success story. Plant Microbe Symbiosis. https://doi.org/10.1007/978-3-030-36248-5_3
Beck D, Meteron L, Afandi F (1993) Practical rhizobium legume technology manual. Manual No. 9. International Center for Agricultural Research in the Dry Areas, Aleppo
Berrada H, Nouioui I, Houssaini MI et al (2012) Phenotypic and genotypic characterizations of rhizobia isolated from root nodules of multiple legume species native of Fez, Morocco. Afr J Microbiol Res 6:5314–5324. https://doi.org/10.5897/AJMR11.1505
Botsford JL, Lewis TA (1990) Osmoregulation in Rhizobium meliloti: production of glutamic acid in response to osmotic stress. Appl Environ Microbiol 56:488–494. https://doi.org/10.1128/AEM.56.2.488-494.1990
Boukhatem ZF, Domergue O, Bekki A et al (2012) Symbiotic characterization and diversity of rhizobia associated with native and introduced acacias in arid and semi-arid regions in Algeria. FEMS Microbiol Ecol 80:534–547. https://doi.org/10.1111/J.1574-6941.2012.01315.X
Bridges CC (1966) Hierarchical cluster analysis. Psychol Rep 18:851–854. https://doi.org/10.2466/PR0.1966.18.3.851
Broughton WJ, Dilworth MJ (1971) Control of leghaemoglobin synthesis in snake beans. Biochem J 125(4):1075–1080
Cardinale M, Lanza A, Bonnì ML et al (2008) Diversity of rhizobia nodulating wild shrubs of Sicily and some neighbouring islands. Arch Microbiol 1904(190):461–470. https://doi.org/10.1007/S00203-008-0394-2
Crisóstomo JA, Rodríguez-Echeverría S, Freitas H (2013) Co-introduction of exotic rhizobia to the rhizosphere of the invasive legume Acacia saligna, an intercontinental study. Appl Soil Ecol 64:118–126. https://doi.org/10.1016/J.APSOIL.2012.10.005
Cummings SP, Gyaneshwar P, Vinuesa P et al (2009) Nodulation of Sesbania species by Rhizobium (Agrobacterium) strain IRBG74 and other rhizobia. Environ Microbiol 11:2510–2525. https://doi.org/10.1111/J.1462-2920.2009.01975.X
De Lajudie P, Willems A, Pot B et al (1994) Polyphasic taxonomy of rhizobia: Emendation of the genus Sinorhizobium and description of Sinorhizobium meliloti comb. nov., Sinorhizobium saheli sp. nov., and Sinorhizobium teranga sp. nov. Int J Syst Bacteriol 44:715–733. https://doi.org/10.1099/00207713-44-4-715/CITE/REFWORKS
De Lajudie P, Willems A, Nick G et al (1999) Agrobacterium bv. 1 Strains Isolated from Nodules of Tropical Legumes. Syst Appl Microbiol 22:119–132. https://doi.org/10.1016/S0723-2020(99)80035-6
De Meyer SE, De Beuf K, Vekeman B, Willems A (2015) A large diversity of non-rhizobial endophytes found in legume root nodules in Flanders (Belgium). Soil Biol Biochem 83:1–11. https://doi.org/10.1016/J.SOILBIO.2015.01.002
Diouf D, Samba-Mbaye R, Lesueur D et al (2007) Genetic diversity of Acacia seyal Del. rhizobial populations indigenous to Senegalese soils in relation to salinity and pH of the sampling sites. Microb Ecol 54:553–566. https://doi.org/10.1007/S00248-007-9243-0
Edwards U, Rogall T, Blockerl H et al (1989) Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res 17:7843–7853
El Hilali I, Brhada F, Thami Alami I, Filali Maltouf A (2007) Evidence of the presence of genetically different rhizobial strains in a single cluster of a lupinoid nodule, the case of Lupinus luteus. J Food, Agric Environ 5:352–359
Elbanna K, Elbadry M, Gamal-Eldin H (2009) Genotypic and phenotypic characterization of rhizobia that nodulate snap bean (Phaseolus vulgaris L.) in Egyptian soils. Syst Appl Microbiol 32:522–530. https://doi.org/10.1016/J.SYAPM.2009.07.006
Essendoubi M, Brhada F, Eljamali JE et al (2007) Osmoadaptative responses in the rhizobia nodulating Acacia isolated from south-eastern Moroccan Sahara. Environ Microbiol 9:603–611. https://doi.org/10.1111/J.1462-2920.2006.01176.X
Fentahun M, Akhtar MS, Muleta D, Lemessa F (2013) Isolation and characterization of nitrogen deficit Rhizobium isolates and their effect on growth of haricot bean. African J Agric Res 8:5942–5952. https://doi.org/10.5897/AJAR2012.6690
Fikri Benbrahim K, Berrada H, El Ghachtouli N et al (2014) Les acacias: des plantes fixatrices d’azote prometteuses pour le développement durable des zones arides et semi-arides [Acacia: Promising nitrogen fixing trees for sustainable development in arid and semi-arid areas]. Int J Innov Appl Stud 8:46–58
Fikri-Benbrahim K, Chraibi M, Lebrazi S et al (2017) Phenotypic and genotypic diversity and symbiotic effectiveness of rhizobia isolated from Acacia sp. grown in Morocco. J Agric Sci Technol 19:201–216
García-Fraile P, Mulas-García D, Peix A et al (2010) Phaseolus vulgaris is nodulated in northern Spain by Rhizobium leguminosarum strains harboring two nodC alleles present in American Rhizobium etli strains: biogeographical and evolutionary implications. Can J Microbiol 56:657–666. https://doi.org/10.1139/w10-048
Geetha SJ, Joshi SJ (2013) Engineering rhizobial bioinoculants: a strategy to improve iron nutrition. Sci World J. https://doi.org/10.1155/2013/315890
Gouffi K, Pica N, Pichereau V, Blanco C (1999) Disaccharides as a new class of nonaccumulated osmoprotectants for Sinorhizobium meliloti. Appl Environ Microbiol 65:1491–1500. https://doi.org/10.1128/AEM.65.4.1491-1500.1999/ASSET/371F56D9-CCCA-42A5-80B1-8A079398A979/ASSETS/GRAPHIC/AM0491711002.JPEG
Hoque MS, Broadhurst LM, Thrall PH (2011) Genetic characterization of root-nodule bacteria associated with Acacia salicina and A. stenophylla (Mimosaceae) across south-eastern Australia. Int J Syst Evol Microbiol 61:299–309. https://doi.org/10.1099/IJS.0.021014-0/CITE/REFWORKS
Hung M-H, Bhagwath AA, Shen F-T et al (2005) Indigenous rhizobia associated with native shrubby legumes in Taiwan. Pedobiologia (JENA) 49:577–584. https://doi.org/10.1016/j.pedobi.2005.06.002
Hungria M, Franco AA, Sprent JI (1993) New sources of high-temperature tolerant rhizobia for Phaseolus vulgaris L. Plant Soil 149:103–109. https://doi.org/10.1007/BF00010767
Jordan D (1984) Family III Rhizobiaceae Conn. 1938–254. In: Krieg NR, Holt JG (eds) Bergey’s manual of systematic bacteriology, vol 1. The Williams and Wilkins Co., Baltimore, pp 235–244
Küçük Ç, Kıvanç M (2010) Preliminary characterization of Rhizobium strains isolated from chickpea nodules. Afr J Biotechnol 7:772–775. https://doi.org/10.4314/ajb.v7i6.58526
Küçük Ç, Kivanç M, Kinaci E (2006) Characterization of Rhizobium sp. isolated from bean. Turkish J Biol 30:127–132
Kumar S, Stecher G, Li M et al (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lebrazi S, Niehaus K, Bednarz H et al (2020) Screening and optimization of indole-3-acetic acid production and phosphate solubilization by rhizobacterial strains isolated from Acacia cyanophylla root nodules and their effects on its plant growth. J Genet Eng Biotechnol 18:1–12. https://doi.org/10.1186/s43141-020-00090-2
Lebrazi S, Fikri-Benbrahim K (2022) Potential of tree legumes in agroforestry systems and soil conservation. In: Advances in legumes for sustainable intensification. Academic Press, New York, pp 461–482
Lebrazi S, Chraibi M, Fadil M, et al (2018) Phenotypic, genotypic and symbiotic characterization of rhizobial isolates nodulating Acacia sp. in Morocco. J Pure Appl Microbiol 12:249–263. https://doi.org/10.22207/JPAM.12.1.30
Maâtallah J, Berraho EB, Muñoz S et al (2002) Phenotypic and molecular characterization of chickpea rhizobia isolated from different areas of Morocco. J Appl Microbiol 93:531–540. https://doi.org/10.1046/J.1365-2672.2002.01718.X
Mhamdi R, Mrabet M, Laguerre G et al (2005) Colonization of Phaseolus vulgaris nodules by Agrobacterium-like strains. Can J Microbiol. https://doi.org/10.1139/w04-120
Missbah El Idrissi M, Bouhnik O, ElFaik S et al (2021) Characterization of Bradyrhizobium spp. nodulating Lupinus cosentinii and L. luteus microsymbionts in Morocco. Front Agron 3:17. https://doi.org/10.3389/FAGRO.2021.661295/BIBTEX
Mohamed SH, Smouni A, Neyra M et al (2000) Phenotypic characteristics of root-nodulating bacteria isolated from Acacia spp. grown in Libya. Plant Soil 224:171–183. https://doi.org/10.1023/A:1004838218642
Moschetti G, Peluso AL, Protopapa A et al (2005) Use of nodulation pattern, stress tolerance, nodC gene amplification, RAPD-PCR and RFLP–16S rDNA analysis to discriminate genotypes of Rhizobium leguminosarum biovar viciae. Syst Appl Microbiol 28:619–631. https://doi.org/10.1016/J.SYAPM.2005.03.009
Moulin L, Béna G, Boivin-Masson C, Stȩpkowski T (2004) Phylogenetic analyses of symbiotic nodulation genes support vertical and lateral gene co-transfer within the Bradyrhizobium genus. Mol Phylogenet Evol 30:720–732. https://doi.org/10.1016/S1055-7903(03)00255-0
Mrabet M, Mnasri B, Ben RS et al (2006) Agrobacterium strains isolated from root nodules of common bean specifically reduce nodulation by Rhizobium gallicum. FEMS Microbiol Ecol 56:304–309. https://doi.org/10.1111/J.1574-6941.2006.00069.X
Muindi MM, Muthini M, Njeru EM, Maingi J (2017) Symbiotic efficiency and genetic characterization of rhizobia and non rhizobial endophytes associated with cowpea grown in semi-arid tropics of Kenya. Heliyon e06867. https://doi.org/10.1016/j.heliyon.2021.e06867
Nelson DW, Sommers LE (1973) Determination of total nitrogen in plant material. Agron J 65:109–112. https://doi.org/10.2134/agronj1973.00021962006500010033x
Odee DW, Haukka K, McInroy SG et al (2002) Genetic and symbiotic characterization of rhizobia isolated from tree and herbaceous legumes grown in soils from ecologically diverse sites in Kenya. Soil Biol Biochem 34:801–811. https://doi.org/10.1016/S0038-0717(02)00009-3
Patel K, Banjare U, Kumari A, et al (2020) Temperature tolerant Rhizobium leguminosorum bv. viciae strains with plant growth promotion traits. J Pure Appl Microbiol 14:2603–2609. https://doi.org/10.22207/JPAM.14.4.36
Requena N, Perez-Solis E, Azcón-Aguilar C et al (2001) Management of indigenous plant-microbe symbioses aids restoration of desertified ecosystems. Appl Environ Microbiol 67:495–498. https://doi.org/10.1128/AEM.67.2.495-498.2001/ASSET/2B9559B2-882D-4FFA-9FA8-5B70879ECF61/ASSETS/GRAPHIC/AM0211115003.JPEG
Ribeiro-Barros AI, Silva MJ, Moura I et al (2018) The potential of tree and shrub legumes in agroforestry systems. In: Amanullah K, Fahad S (eds) Nitrogen in agriculture—updates. InTechOpen, London, pp 223–239
Rincón-Rosales R, Lloret L, Ponce E, Martínez-Romero E (2009) Rhizobia with different symbiotic efficiencies nodulate Acaciella angustissima in Mexico, including Sinorhizobium chiapanecum sp. nov. which has common symbiotic genes with Sinorhizobium mexicanum. FEMS Microbiol Ecol 67:103–117. https://doi.org/10.1111/j.1574-6941.2008.00590.x
Sharma A, Bandamaravuri KB, Sharma A et al (2017) Phenotypic and molecular assessment of chickpea rhizobia from different chickpea cultivars of India. 3 Biotech. https://doi.org/10.1007/S13205-017-0952-X
Somasegaran P, Hoben HJ (1994) Quantifying the growth of rhizobia. In: Handbook for rhizobia. Springer, New York. https://doi.org/10.1007/978-1-4613-8375-8_5
Suman J, Rakshit A, Ogireddy SD et al (2022) Microbiome as a key player in sustainable agriculture and human health. Front Soil Sci. https://doi.org/10.3389/FSOIL.2022.821589/FULL
Surange S, Wollum AG, Kumar N, Shekhar Nautiyal C (1997) Characterization of Rhizobium from root nodules of leguminous trees growing in alkaline soils. Can J Microbiol 43:891–894. https://doi.org/10.1139/M97-130
Taoufiq K, Faghire M, Tahrouch S et al (2018) Screening and molecular identification of endophytic bacteria isolated from legumes nodules and roots cultivated in Acacia rhizosphere soils collected in an arid region, Tata-Akka in south of Morocco. Indian J Nat Sci 9:14910–14919
Vincent J. (1970) A manual for practical study of root nodule bacteria. In: IBP Hand book No. 15. Blackwell, Oxford
Wang LL, Wang ET, Liu J et al (2006) Endophytic occupation of root nodules and roots of Melilotus dentatus by Agrobacterium tumefaciens. Microb Ecol 52:436–443. https://doi.org/10.1007/S00248-006-9116-Y/METRICS
Young JM, Kuykendall LD, Martínez-Romero E et al (2003) Classification and nomenclature of Agrobacterium and Rhizobium—a reply to Farrand et al. (2003). Int J Syst Evol Microbiol 53:1689–1695. https://doi.org/10.1099/IJS.0.02762-0/CITE/REFWORKS
Youseif SH, Abd El-Megeed FH, Ageez A et al (2014) Phenotypic characteristics and genetic diversity of rhizobia nodulating soybean in Egyptian soils. Eur J Soil Biol 60:34–43. https://doi.org/10.1016/J.EJSOBI.2013.10.008
Zahran HH (1999) Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol Mol Biol Rev 63:968–989. https://doi.org/10.1128/MMBR.63.4.968-989.1999
Zahran HH, Fattah A, Yasser MM et al (2012) Diversity and environmental stress responses of rhizobial bacteria from Egyptian grain legumes. Aust J Basic Appl Sci 6:571–583
Zakhia F, Jeder H, Willems A et al (2006) Diverse bacteria associated with root nodules of spontaneous legumes in Tunisia and first report for nifH-like gene within the Genera Microbacterium and Starkeya. Microb Ecol 513(51):375–393. https://doi.org/10.1007/S00248-006-9025-0
Zerhari K, Aurag J, Khbaya B et al (2000) Phenotypic characteristics of rhizobia isolates nodulating acacia species in the arid and Saharan regions of Morocco. Lett Appl Microbiol 30:351–357. https://doi.org/10.1046/J.1472-765X.2000.00730.X
Zhang X, Harper R, Karsisto M, Lindstrom K (1991) Diversity of Rhizobium bacteria isolated from the root nodules of leguminous trees. Int J Syst Bacteriol 41:104–113. https://doi.org/10.1099/00207713-41-1-104/CITE/REFWORKS
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
LS conducted the main experiments, designed the work, and wrote the manuscript. FM analyzed the data.CM participated in some experiments. FBK directed the work. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests and this work did not receive funding from any organization or institution.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lebrazi, S., Fadil, M., Chraibi, M. et al. Phenotypic, molecular, and symbiotic characterization of the rhizobial symbionts isolated from Acacia saligna grown in different regions in Morocco: a multivariate approach. World J Microbiol Biotechnol 39, 343 (2023). https://doi.org/10.1007/s11274-023-03775-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-023-03775-1