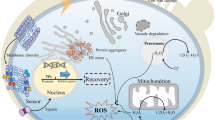
Abstract
Formic acid is a representative small molecule acid in lignocellulosic hydrolysate that can inhibit the growth of Saccharomyces cerevisiae cells during alcohol fermentation. However, the mechanism of formic acid cytotoxicity remains largely unknown. In this study, RNA-Seq technology was used to study the response of S. cerevisiae to formic acid stress at the transcriptional level. Scanning electron microscopy and Fourier transform infrared spectroscopy were conducted to observe the surface morphology of yeast cells. A total of 1504 genes were identified as being differentially expressed, with 797 upregulated and 707 downregulated genes. Transcriptomic analysis showed that most genes related to glycolysis, glycogen synthesis, protein degradation, the cell cycle, the MAPK signaling pathway, and redox regulation were significantly induced under formic acid stress and were involved in protein translation and synthesis amino acid synthesis genes were significantly suppressed. Formic acid stress can induce oxidative stress, inhibit protein biosynthesis, cause cells to undergo autophagy, and activate the intracellular metabolic pathways of energy production. The increase of glycogen and the decrease of energy consumption metabolism may be important in the adaptation of S. cerevisiae to formic acid. In addition, formic acid can also induce sexual reproduction and spore formation. This study through transcriptome analysis has preliminarily reveal the molecular response mechanism of S. cerevisiae to formic acid stress and has provided a basis for further research on methods used to improve the tolerance to cell inhibitors in lignocellulose hydrolysate.








Similar content being viewed by others
References
Abbott DA, Suir E, Duong GH, Hulster ED, Pronk JT, Maris AJA (2009) Catalase overexpression reduces lactic acid-induced oxidative stress in Saccharomyces cerevisiae. Appl Environ Microbiol 75(8):2320–2325. https://doi.org/10.1128/AEM.00009-09
Auesukaree C (2017) Molecular mechanisms of the yeast adaptive response and tolerance to stresses encountered during ethanol fermentation. J Biosci Bioeng 124(2):133–142. https://doi.org/10.1016/j.jbiosc.2017.03.009
Becker E, Liu YC, Lardenois A, Walther T, Horecka J, Stuparevic I, Law MJ, Lavigne R, Evrard B, Demougin P, Riffle M, Strich R, Davis RW, Pineau C, Primig M (2015) Integrated RNA- and protein profiling of fermentation and respiration in diploid budding yeast provides insight into nutrient control of cell growth and development. J Proteomics 119:30–44. https://doi.org/10.1016/j.jprot.2015.01.015
Chen YH, Zhang X, Zhang M, Zhu JY, Wu ZF, Zheng XJ (2018) A transcriptome analysis of the ameliorate effect of Cyclocarya paliurus triterpenoids on ethanol stress in Saccharomyces cerevisiae. World J Microbiol Biotechnol 34(12):182. https://doi.org/10.1007/s11274-018-2561-1
Cheng L, Zhang X, Zheng X, Wu ZF, Weng PF (2019) RNA-seq transcriptomic analysis of green tea polyphenols regulation of differently expressed genes in Saccharomyces cerevisiae under ethanol stress. World J Microbiol Biotechnol 35(4):59. https://doi.org/10.1007/s11274-019-2639-4
Cray JA, Bell AN, Bhaganna P, Mswaka AY, Timson DJ, Hallsworth JE (2013) The biology of habitat dominance; can microbes behave as weeds? Microb Biotechnol 6(5):453–492. https://doi.org/10.1111/1751-7915.12027
Cunha JT, Aguiar QT, Romaní A, Oliveira C, Domingues L (2015) Contribution of PRS3, RPB4 and ZWF1 to the resistance of industrial Saccharomyces cerevisiae CCUG53310 and PE-2 strains to lignocellulosic hydrolysate-derived inhibitors. Biores Technol 191:7–16. https://doi.org/10.1016/j.biortech.2015.05.006
Decesare JM, Stuart DT (2012) Among B-type cyclins only CLB5 and CLB6 promote premeiotic S phase in Saccharomyces cerevisiae. Genetics 190(3):1001–1016. https://doi.org/10.1534/genetics.111.134684
Deng D (2016) Yan N (2016) GLUT, SGLT, and SWEET: structural and mechanistic investigations of the glucose transporters. Protein Sci 25(3):546–558. https://doi.org/10.1002/pro.2858
Dong YC, Hu JJ, Fan LL, Chen QH (2017) RNA-Seq-based transcriptomic and metabolomic analysis reveal stress responses and programmed cell death induced by acetic acid in Saccharomyces cerevisiae. Sci Rep 17(7):42659. https://doi.org/10.1038/srep42659
Dragon F, Gallagher JEG, Compagnone-Post PA, Mitchell BM, Porwancher KA, Wehner KA, Wormsley S, Settlage RE, Shabanowitz J, Osheim Y, Beyer AL, Hunt DF, Baserga SJ (2002) A large nucleolar U3 ribonucleoprotein required for 18S ribosomal RNA biogenesis. Nature 417(6892):967–970. https://doi.org/10.1038/nature00769
Fletcher E, Baetz K (2020) Multi-faceted systems biology approaches present a cellular landscape of phenolic compound inhibition in Saccharomyces cerevisiae. Front Bioeng Biotechnol 8:539902. https://doi.org/10.3389/fbioe.2020.539902
Goldberg AL (2003) Protein degradation and protection against misfolded or damaged proteins. Nature 426(6968):895–899. https://doi.org/10.1038/nature02263
Gowda NKC, Kandasamy G, Froehlich MS, Dohmen RJ, Andréasson C (2013) Hsp70 nucleotide exchange factor Fes1 is essential for ubiquitin-dependent degradation of misfolded cytosolic proteins. Proc Natl Acad Sci USA 110(15):5975–5980. https://doi.org/10.1073/pnas.1216778110
Guldfeldt LU, Arneborg N (1998) Measurement of the effects of acetic acid and extracellular pH on intracellular pH of nonfermenting, individual Saccharomyces cerevisiae cells by fluorescence microscopy. Appl Environ Microbiol 64(2):530–534. https://doi.org/10.1128/AEM.64.2.530-534.1998
Hailey DW, Rambold AS, Satpute-Krishnan P, Mitra K, Sougrat R, Kim PK, Lippincott-Schwartz J (2010) Mitochondria supply membranes for autophagosome biogenesis during starvation. Cell 141(4):656–667. https://doi.org/10.1016/j.cell.2010.04.009
Henriques SF, Mira NP, Sácorreia I (2017) Genome-wide search for candidate genes for yeast robustness improvement against formic acid reveals novel susceptibility (Trk1 and positive regulators) and resistance (Haa1-regulon) determinants. Biotechnol Biofuels 10(1):96. https://doi.org/10.1186/s13068-017-0781-5
Horsey EW, Jakovljevic J, Miles TD, Harnpicharnchai P (2004) Role of the yeast Rrpl protein in the dynamics of pre-ribosome maturation. RNA 10(5):813–827. https://doi.org/10.1261/rna.5255804
Jain A, Lamark T, Sjottem E, Larsen KB, Awuh JA, Øvervatn A, McMahon M, Hayes JD, Johansen T (2010) p62/SQSTMI is a target gene for transcription factor NRF2 and creates a positive feedback loop by inducingantioxidant response element-driven gene transcription. J Biol Chem 285(29):22576–22591. https://doi.org/10.1074/jbc.M110.118976
Kelliher CM, Foster MW, Motta FC, Deckard A, Soderblom EJ, Moseley MA, Haase SB (2018) Layers of regulation of cell-cycle gene expression in the budding yeast Saccharomyces cerevisiae. Mol Biol Cell 29(22):2644–2655. https://doi.org/10.1091/mbc.E18-04-0255
Kieliszek M, Dourou M (2021) Effect of selenium on the growth and lipid accumulation of Yarrowia lipolytica yeast. Biol Trace Elem Res 199(4):1611–1622. https://doi.org/10.1007/s12011-020-02266-w
Kieliszek M, Błażejak S, Bzducha-Wróbel A, Kot AM (2019) Effect of selenium on lipid and amino acid metabolism in yeast cells. Biol Trace Elem Res 187(1):316–327. https://doi.org/10.1007/s12011-018-1342-x
Kornblatt MJ, Richard JR, Mattie S, Zakaib J, Dayanandan S, Hanic-Joyce PJ, Joyce PBM (2013) The saccharomyces cerevisiae, enolase-related regions encode proteins that are active enolases. Yeast 30(2):55–69. https://doi.org/10.1002/yea.2940
Kosa G, Shapaval V, Kohler A, Zimmermann B (2017) FTIR spectroscopy as a unified method for simultaneous analysis of intra- and extracellular metabolites in high-throughput screening of microbial bioprocesses. Microb Cell Fact 16(1):195. https://doi.org/10.1186/s12934-017-0817-3
Kot AM, Błażejak S, Kieliszek M, Gientka I, Bryś J, Reczek L, Pobiega K (2019) Effect of exogenous stress factors on the biosynthesis of carotenoids and lipids by Rhodotorula yeast strains in media containing agroindustrial waste. World J Microbiol Biotechnol 35(10):1–10. https://doi.org/10.1007/s11274-019-2732-8
Krause SA, Cundell MJ, Poon PP (2012) Functional specialisation of yeast Rho1 GTP exchange factors. J Cell Sci 125(11):2721–2731. https://doi.org/10.1242/jcs.100685
Lazar Z, Neuveglise C, Rossignol T, Devillers H, Morin N, Robak M, Nicaud JM, Crutz-Le Coq AM (2017) Characterization of hexose transporters in Yarrowia lipolytica reveals new groups of Sugar Porters involved in yeast growth. Fungal Genet Biol 100:1–12. https://doi.org/10.1016/j.fgb.2017.01.001
Lee SE, Park BS, Yoon JJ (2010) Proteomic evaluation of cellular responses of Saccharomyces cerevisiae to formic acid stress. Mycobiology 38(4):302–309. https://doi.org/10.4489/MYCO.2010.38.4.302
Levin DE (2005) Cell wall integrity signaling in Saccharomyces cerevisiae. Microbiol Mol Biol Rev 69(2):262–291. https://doi.org/10.1128/MMBR.69.2.262-291.2005
Li Y, Wen TT, Xue C, Han Q, Wang Y, Hong JL, Zhou XM, Jiang HJ (2013) RGO LBL modified biomimetic electrochemical sensor for detection of sildenafil in herbal sexual healthproducts. Biosens Bioelectron 42:287–292. https://doi.org/10.1016/j.bios.2012.10.056
Li DC, Wang DH, Wei GY (2017) Efficient co-production of S-adenosylmethionine and glutathione by Candida utilis: effect ofdissolved oxygen on enzyme activity and energy supply. J Chem Technol Biotechnol 92(8):2150–2158. https://doi.org/10.1002/jctb.5226
Li X, Cen N, Liu L, Chen Y, Yang X, Yu K, Guo JL, Liao XP, Shi B (2020) Collagen peptide provides Saccharomyces cerevisiae with robust stress tolerance for enhanced bioethanol production. ACS Appl Mater Interfaces 12(48):53879–53890. https://doi.org/10.1021/acsami.0c18919
Lovato FL, Teixeira da Rocha JB, Dalla Corte CL (2017) Diphenyl diselenide protects against methylmercury-induced toxicity in Saccharomyces cerevisiae via the Yap1 transcription factor. Chem Res Toxicol 30(5):1134–1144. https://doi.org/10.1021/acs.chemrestox.6b00449
Ma M, Liu ZL (2010) Comparative transcriptome profiling analyses during the lag phase uncover YAP1, PDR1, PDR3, RPN4, and HSF1 as key regulatory genes in genomic adaptation to the lignocellulose derived inhibitor HMF for Saccharomyces cerevisiae. BMC Genomics 11(1):660. https://doi.org/10.1186/1471-2164-11-660
Mailloux RJ, Mcbride SL, Harper ME (2013) Unearthing the secrets of mitochondrial ROS and glutathione in bioenergetics. Trends Biochem Sci 38(12):592–602. https://doi.org/10.1016/j.tibs.2013.09.001
Martani F, Fossati T, Posteri R, Signori L, Porro D, Branduardi P (2014) Different response to acetic acid stress in Saccharomyces cerevisiae wild-type and l-ascorbic acid-producing strains. Yeast 30(9):365–378. https://doi.org/10.1002/yea.2969
Martin C, Alriksson B, Sjöde A, Nilvebrant NO, Jönsson LJ (2007) Dilute sulfuric acid pretreatment of agricultural and agro-industrial residues for ethanol production. Appl Biochem Biotechnol 137(1):339–352. https://doi.org/10.1007/s12010-007-9063-1
Matsushika A, Sawayama S (2012) Characterization of a recombinant flocculent Saccharomyces cerevisiae strain that co-ferments glucose and xylose: II. Influence of pH and acetic acid on ethanol production. Appl Biochem Biotechnol 168(8):2094–2104. https://doi.org/10.1007/s12010-012-9920-4
Meijer A, Codogno P (2011) Autophagy: regulation by energy sensing. Curr Biol 21(6):227–229. https://doi.org/10.1016/j.cub.2011.02.007
Mira NP, Palma M, Guerreiro JF, Sá-Correia I (2010) Genome-wide identification of Saccharomyces cerevisiae genes required for tolerance to acetic acid. Microb Cell Fact 9(1):79. https://doi.org/10.1186/1475-2859-9-79
Mitsui R, Yamada R, Matsumoto T, Yoshihara S, Tokumoto H, Ogino H (2020) Construction of lactic acid-tolerant Saccharomyces cerevisiae by using CRISPR-Cas-mediated genome evolution for efficient d-lactic acid production. Appl Microbiol Biotechnol 104(21):9147–9158. https://doi.org/10.1007/s00253-020-10906-3
Mizushima N (2007) Autophagy: process and function. Genes Dev 21(22):2861. https://doi.org/10.1101/gad.1599207
Nomura N, Verdon G, Kang HJ, Shimamura T, Nomura Y, Sonoda Y, Hussien SA, Qureshi AA, Coincon M (2015) Structure and mechanism of the mammalian fructose transporter GLUTS. Nature 526(7573):397–401. https://doi.org/10.1038/nature14909
Özcan S, Johnston M (1999) Function and regulation of yeast hexose transporters. Microbiol Mol Biol Rev 63(3):554–569. https://doi.org/10.1128/MMBR.63.3.554-569.1999
Palmqvist E, Hahn-Hägerdal B (2000) Fermentation of lignocellulosic hydrolysates. II: Inhibitors and mechanisms of inhibition. Bioresour Technol 74(1):25–33. https://doi.org/10.1016/S0960-8524(99)00161-3
Pérez-Torrado R, Gimeno-alcañiz JV, Matallana E (2002) Wine yeast strains engineered for glycogen overproduction display enhanced viability under glucose deprivation conditions. Appl Environ Microbiol 68(7):3339–3344. https://doi.org/10.1128/AEM.68.7.3339-3344.2002
Piotrowska M, Masek A (2015) Saccharomyces cerevisiae cell wall components as tools for ochratoxin A decontamination. Toxins 7(4):1151–1162. https://doi.org/10.3390/toxins7041151
Piper PW, Ortiz-Calderon C, Holyoak C, Coote P, Cole M (1997) Hsp30, the integral plasma membrane heat shock protein of Saccharomyces cerevisiae, is a stress-inducible regulator of plasma membrane H(+)-ATPase. Cell Stress Chaperones 2(1):12–24. https://doi.org/10.1379/1466-1268(1997)002%3c0012:htipmh%3e2.3.co;2
Qin L, Wang Z, Tao L, Wang Y (2010) ER stress negatively regulates AKT/TSC/mTOR pathway to enhance autophagy. Autophagy 6(2):239–247. https://doi.org/10.4161/auto.6.2.11062
Rogowska A, Pomastowski P, Złoch M, Railean-Plugaru V, Król A, Rafińska K, Szultka-Młyńska M, Buszewski B (2018) The influence of different pH on the electrophoretic behaviour of Saccharomyces cerevisiae modified by calcium ions. Sci Rep 8(1):1–10. https://doi.org/10.1038/s41598-018-25024-4
Rosenzweig R, Nillegoda NB, Mayer MP, Bukau B (2019) The Hsp70 chaperone network. Nat Rev Mol Cell Biol 20(11):665–680. https://doi.org/10.1038/s41580-019-0133-3
Salman A, Shufan E, Lapidot I (2019) Distinction between mixed genus bacteria using infrared spectroscopy and multivariate analysis. Vib Spectrosc 100:6–13. https://doi.org/10.1016/j.vibspec.2018.10.009
Schröder M, Chang JS, Kaufman RJ (2000) The unfolded protein response represses nitrogen-starvation induced developmental differentiation in yeast. Genes Dev 14(23):2962–2975. https://doi.org/10.1101/gad.852300
Shapaval V, Brandenburg J, Blomqvist J, Tafintseva V, Passoth V, Sandgren M, Kohler A (2019) Biochemical profiling, prediction of total lipid content and fatty acid profile in oleaginous yeasts by FTIR spectroscopy. Biotechnol Biofuels 12(1):140–151. https://doi.org/10.1186/s13068-019-1481-0
Stanley D, Chambers PJ, Stanley GA, Borneman A, Fraser S (2010) Transcriptional changes associated with ethanol tolerance in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 88(1):231–239. https://doi.org/10.1007/s00253-010-2760-7
Suzuki T, Sugiyama M, Wakazono K, Kaneko Y, Harashima S (2012) Lactic-acid stress causes vacuolar fragmentation and impairs intracellular amino-acid homeostasis in Saccharomyces cerevisiae. J Biosci Bioeng 113(4):421–430. https://doi.org/10.1016/j.jbiosc.2011.11.010
Szymanska-Chargot M, Zdunek A (2013) Use of FT-IR spectra and PCA to the bulk characterization of cell wall residues of fruits and vegetables along a fraction process. Food Biophys 8(1):29–42. https://doi.org/10.1007/s11483-012-9279-7
Tal MC, Sasai M, Lee HK, Yordy B, Shadel GS, Iwasaki A (2009) Absence of autophagy results in reactive oxygen species-dependent amplification of RLR signaling. Proc Natl Acad Sci USA 106(8):2770–2775. https://doi.org/10.1073/pnas.0807694106
Tedesco V, Roquet RF, DeMis J, Chiamulera C, Monfils MH (2014) Extinction, applied after retrieval of auditory fear memory, selectively increases zinc-finger protein 268 andphosphorylated ribosomal protein S6 expression inprefrontal cortex and lateral amygdala. Neurobiol Learn Mem 115:78–85. https://doi.org/10.1016/j.nlm.2014.08.015
Van BdH, Abee T, Tempelaars M, Bron PA, Kleerebezem M, Marco ML (2011) Short- and long-term adaptation to ethanol stress and its cross-protective consequences in Lactobacillus plantarum. Appl Environ Microbiol 77(15):5247–5256. https://doi.org/10.1128/AEM.00515-11
Vandermeulen MD, Cullen PJ (2020) New aspects of invasive growth regulation identified by functional profiling of MAPK pathway targets in Saccharomyces cerevisiae. Genetics 216(1):95–116. https://doi.org/10.1534/genetics.120.303369
Wallacesafinas V, Gorwagrauslund MF (2013) Adaptive evolution of an industrial strain of Saccharomyces cerevisiae for combined tolerance to inhibitors and temperature. Biotechnol Biofuels 6(1):151. https://doi.org/10.1186/1754-6834-6-151
Wang T, Zheng X, Wang X, Li X, Shen YH (2017) Different biosorption mechanisms of Uranium(VI) by live and heat-killed Saccharomyces cerevisiae under environmentally relevant conditions. J Environ Radioact 167:92–99. https://doi.org/10.1016/j.jenvrad.2016.11.018
Weber H, Ppolen T, Heuveling J, Wendisch VF, Hengge R (2005) Genome-wide analysis of the general stress response network in Escherichia coli: sigma(S)-dependent genes, promoters, and sigma factor selectivity. J Bacteriol 187(5):1591–1603. https://doi.org/10.1128/JB.187.5.1591-1603.2005
Xia ZC, Zhou XL, Li JY, Li L, Ma Y, Wu Y, Huang Z, Li XR, Xu PX, Xue M (2019) Multiple-omics techniques reveal the role of glycerophospholipid metabolic pathway in the response of Saccharomyces cerevisiae against hypoxic stress. Front Microbiol 10:1398. https://doi.org/10.3389/fmicb.2019.01398
Xuan YH, Hu YB, Chen LQ, Sosso D, Ducat DC, Hou BH, Frommer WB (2013) Functional role of oligomerization for bacterial and plant SWEET sugar transporter family. Proc Natl Acad Sci USA 110(39):E3685–E3694. https://doi.org/10.1073/pnas.1311244110
Yang SH, Franden MA, Yang Q, Chou YC, Zhang M, Pienkos PT (2020) Identification of inhibitors in lignocellulosic slurries and determination of their effect on hydrocarbon-producing microorganisms. Front Bioeng Biotechnol 6:23. https://doi.org/10.3389/fbioe.2018.00023
Yang Y, Ren WB, Xu HN, Cheng L, Dapaah MF, He Rh, Ma H (2021) Incorporating Transcriptomic-Metabolomic analysis reveal the effect of ultrasound on ethanol production in Saccharomyces cerevisiae. Ultrason Sonochem 79:105791. https://doi.org/10.1016/j.ultsonch.2021.105791
Ye W, Zhang W, Liu T, Tan GH, Li HH, Huang ZL (2016) Improvement of ethanol production in Saccharomyces cerevisiae by high-efficient disruption of the ADH2 gene using a novel recombinant TALEN vector. Front Microbiol 11(7):1067. https://doi.org/10.3389/fmicb.2016.01067
Yu L, Qi M, Sheff MA, Elion EA (2008) Counteractive control of polarized morphogenesis duringmating by mitogen-activated protein kinase Fus3 and Gl cyclin-dependent kinase. Mol Biol Cell 19(4):1739–1752. https://doi.org/10.1091/mbc.e07-08-0757
Yun HR, Yong HJ, Kim J, Shin Y, Kim SS, Choi TG (2020) Roles of autophagy in oxidative stress. Int J Mol Sci 21(9):3289. https://doi.org/10.3390/ijms21093289
Zhang Q, Zhao H, Zhang G, He K, Yang Z, Jin Y (2012) Transcriptome analysis of Saccharomyces cerevisiae at the late stage of very high gravity (VHG) fermentation. Afr J Biotechnol 11:9641–9648. https://doi.org/10.5897/AJB12.268
Zhang JG, Chen XM, Zhou J, Luo XG (2020) Uranium biosorption mechanism model of protonated Saccharomyces cerevisiae. J Hazard Mater 385:121588. https://doi.org/10.1016/j.jhazmat.2019.121588
Zimdars S, Schrage L, Sommer S, Schieber A, Weber F (2019) Influence of glutathione on yeast fermentation efficiency under copper stress. J Agric Food Chem 67(39):10913–10920. https://doi.org/10.1021/acs.jafc.9b03519
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 31660250), Natural Science Foundation of Guangxi (Grant No. 2018GXNSFAA050116). We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zeng, L., Huang, J., Feng, P. et al. Transcriptomic analysis of formic acid stress response in Saccharomyces cerevisiae. World J Microbiol Biotechnol 38, 34 (2022). https://doi.org/10.1007/s11274-021-03222-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03222-z