Abstract
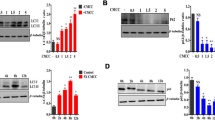
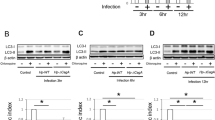
Helicobacter pylori infection is an essential factor in the development of human gastric diseases, but its pathogenic mechanism is still unclear. In this work we have showed that, the LC3II levels were increased and β1-integrin levels were decreased in H. pylori-positive human gastric tissue samples and H. pylori co-cultured GES-1 cells. There was significant upregulation of LC3II levels and downregulation of P62 levels in GES-1 cells after β1-integrin knockdown co-cultured with H. pylori. This indicated that β1-integrin downregulation promoted autophagy in GES-1 cells after H. pylori infection. The cell apoptosis rate and poly ADP-ribose polymerase (PARP) and caspase-3 activities were increased in GES-1 cells pretreated with 3-methyladenine (3-MA ) after H. pylori infection. Furthermore, there was a significant decrease in apoptosis of β1-integrin knockdown GES-1 cells co-cultured with H. pylori; apoptosis was also downregulated in β1-integrin knockdown- and 3-MA-treated GES-1 cells co-cultured with H. pylori. Correspondingly, PARP and caspase-3 activities were decreased in β1-integrin knockdown cells co-cultured with H. pylori and β1-integrin knockdown-3-MA-treated-1 cells with H. pylori infection. Thus, β1-integrin is a novel autophagy and apoptosis regulator during H. pylori infection. However, inhibition of autophagy did not reverse the decrease in apoptosis caused by downregulation of β1-integrin.




Similar content being viewed by others
Data availability
The authors declare data transparency.
Code availability
Not applicable.
References
Barrow-McGee R, Kishi N, Joffre C, Ménard L, Hervieu A, Bakhouche BA, Noval AJ, Mai A, Guzmán C, Robbez-Masson L, Iturrioz X, Hulit J, Brennan CH, Hart IR, Parker PJ, Ivaska J, Kermorgant S (2016) Beta 1-integrin-c-Met cooperation reveals an inside-in survival signalling on autophagy-related endomembranes. Nat Commun 7:1–17. https://doi.org/10.1038/ncomms11942
Blaser MJ (1987) Gastric Campylobacter-like organisms, gastritis, and peptic ulcer disease. Gastroenterology 93:371–383. https://doi.org/10.1016/0016-5085(87)91028-6
Chu YT, Wang YH, Wu JJ, Lei HY (2010) Invasion and multiplication of Helicobacter pylori in gastric epithelial cells and implications for antibiotic resistance. Infect Immun 78:4157–4165. https://doi.org/10.1128/IAI.00524-10
Covacci A, Telford JL, Del Giudice G, Parsonnet J, Rappuoli R (1999) Helicobacter pylori virulence and genetic geography. Science 284:1328–1333. https://doi.org/10.1126/science.284.5418.1328
Cover TL, Blaser MJ (2009) Helicobacter pylori in health and disease. Gastroenterology 136:1863–1873. https://doi.org/10.1053/j.gastro.2009.01.073
Cremer TJ, Amer A, Tridandapani S, Butchar JP (2009) Francisella tularensis regulates autophagy-related host cell signaling pathways. Autophagy 5:125–128. https://doi.org/10.4161/auto.5.1.7305
Datan E, Shirazian A, Benjamin S, Matassov D, Tinari A, Malorni W, Lockshin RA, Garcia-Sastre A, Zakeri Z (2014) mTOR/p70S6K signaling distinguishes routine, maintenance-level autophagy from autophagic cell death during influenza A infection. Virology 5:175–190. https://doi.org/10.1016/j.virol.2014.01.008
Deuretzbacher A, Czymmeck N, Reimer R, Trülzsch K, Gaus K, Hohenberg H, Heesemann J, Aepfelbacher M, Ruckdeschel K (2009) Beta1 integrin-dependent engulfment of Yersinia enterocolitica by macrophages is coupled to the activation of autophagy and suppressed by type III protein secretion. J Immunol 183:5847–5860. https://doi.org/10.4049/jimmunol.0804242
Djavaheri-Mergny M, Maiuri MC, Kroemer G (2010) Cross talk between apoptosis and autophagy by caspase-mediated cleavage of Beclin 1. Oncogene 29:1717–1719. https://doi.org/10.1038/onc.2009.519
El-Khattouti A, Selimovic D, Haikel Y, Hassan M (2013) Crosstalk between apoptosis and autophagy: molecular mechanisms and therapeutic strategies in cancer. J Cell Death 6:37–55. https://doi.org/10.4137/JCD.S11034
Fischbach W, Malfertheiner P (2018) Helicobacter pylori infection. Dtsch Arztebl Int 115:429–436. https://doi.org/10.3238/arztebl.2018.0429
Hathroubi S, Servetas SL, Windham I, Merrell DS, Ottemann KM (2018) Helicobacter pylori biofilm formation and its potential role in pathogenesis. Microbiol Mol Biol Rev 82:e00001-18. https://doi.org/10.1128/MMBR.00001-18
Hou W, Han J, Lu C, Goldstein LA, Rabinowich H (2010) Autophagic degradation of active caspase-8: a crosstalk mechanism between autophagy and apoptosis. Autophagy 6:891–900. https://doi.org/10.4161/auto.6.7.13038
Hunt RH, Camilleri M, Crowe SE, El-Omar EM, Fox JG, Kuipers EJ, Malfertheiner P, McColl KE, Pritchard DM, Rugge M, Sonnenberg A, Sugano K, Tack J (2015) The stomach in health and disease. Gut 64:1650–1668. https://doi.org/10.1136/gutjnl-2014-307595
Jiménez-Soto LF, Kutter S, Sewald X, Ertl C, Weiss E, Kapp U, Rohde M, Pirch T, Jung K, Retta SF, Terradot L, Fischer W, Haas R (2009) Helicobacter pylori type IV secretion apparatus exploits beta1 integrin in a novel RGD-independent manner. PLoS Pathog 5:1–14. https://doi.org/10.1371/journal.ppat.1000684
Kamm KE, Stull JT (2001) Dedicated myosin light chain kinases with diverse cellular functions. J Biol Chem 276:4527–4530. https://doi.org/10.1074/jbc.R000028200
Kayath CA, Hussey S, El hajjami N, Nagra K, Philpott D, Allaoui A (2010) Escape of intracellular Shigella from autophagy requires binding to cholesterol through the type III effector, IcsB. Microbes Infect 12:956–966. https://doi.org/10.1016/j.micinf.2010.06.006
Klionsky DJ, Emr SD (2000) Autophagy as a regulated pathway of cellular degradation. Science 290:1717–1721. https://doi.org/10.1126/science.290.5497.1717
Lamb A, Chen LF (2013) Role of the Helicobacter pylori-induced inflammatory response in the development of gastric cancer. J Cell Biochem 114:491–497. https://doi.org/10.1002/jcb.24389
Lv X, Pu X, Qin G, Zhu T, Lin H (2014) The roles of autophagy in development and stress responses in Arabidopsis thaliana. Apoptosis 19:905–921. https://doi.org/10.1007/s10495-014-0981-4
Maiuri MC, Zalckvar E, Kimchi A, Kroemer G (2007) Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol 8:741–752. https://doi.org/10.1038/nrm2239
Marshall BJ, Warren JR (1984) Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet 1:1311–1315. https://doi.org/10.1016/s0140-6736(84)91816-6
Merino E, Flores-Encarnación M, Aguilar-Gutiérrez GR (2017) Functional interaction and structural characteristics of unique components of Helicobacter pylori T4SS. FEBS J 284:3540–3549. https://doi.org/10.1111/febs.14092
Miller LM, Pritchard JM, Macdonald SJF, Jamieson C, Watson AJB (2017) Emergence of Small-Molecule Non-RGD-Mimetic Inhibitors for RGD Integrins. J Med Chem 60:3241–3251. https://doi.org/10.1021/acs.jmedchem.6b01711
Nakagawa I, Amano A, Mizushima N, Yamamoto A, Yamaguchi H, Kamimoto T, Nara A, Funao J, Nakata M, Tsuda K, Hamada S, Yoshimori T (2004) Autophagy defends cells against invading group A Streptococcus. Science 306:1037–1040. https://doi.org/10.1126/science.1103966
Oral O, Akkoc Y, Bayraktar O, Gozuacik D (2016) Physiological and pathological significance of the molecular cross-talk between autophagy and apoptosis. Histol Histopathol 31:479–498. https://doi.org/10.14670/HH-11-714
Ouimet M, Koster S, Sakowski E, Ramkhelawon B, van Solingen C, Oldebeken S, Karunakaran D, Portal-Celhay C, Sheedy FJ, Ray TD, Cecchini K, Zamore PD, Rayner KJ, Marcel YL, Philips JA, Moore KJ (2016) Mycobacterium tuberculosis induces the miR-33 locus to reprogram autophagy and host lipid metabolism. Nat Immunol 17:677–686. https://doi.org/10.1038/ni.3434
Py BF, Lipinski MM, Yuan J (2007) Autophagy limits Listeria monocytogenes intracellular growth in the early phase of primary infection. Autophagy 3:117–125. https://doi.org/10.4161/auto.3618
Qin Z, Yang Y, Wang H, Luo J, Huang X, You J, Wang B, Li M (2016) Role of autophagy and apoptosis in the postinfluenza bacterial pneumonia. Biomed Res Int 1:1–10. https://doi.org/10.1155/2016/3801026
Savoldi A, Carrara E, Graham DY, Conti M, Tacconelli E (2018) Prevalence of antibiotic resistance in Helicobacter pylori: a systematic review and meta-analysis in world health organization regions. Gastroenterology 155:1372–1382. https://doi.org/10.1053/j.gastro.2018.07.007
Siao D, Somsouk M (2014) Helicobacter pylori: evidence-based review with a focus on immigrant populations. J Gen Intern Med 29:520–528. https://doi.org/10.1007/s11606-013-2630-y
Song S, Tan J, Miao Y, Li M, Zhang Q (2017) Crosstalk of autophagy and apoptosis: Involvement of the dual role of autophagy under ER stress. J Cell Physiol 232:2977–2984. https://doi.org/10.1002/jcp.25785
Tang B, Li N, Gu J, Zhuang Y, Li Q, Wang HG, Fang Y, Yu B, Zhang JY, Xie QH, Chen L, Jiang XJ, Xiao B, Zou QM, Mao XH (2012) Compromised autophagy by MIR30B benefits the intracellular survival of Helicobacter pylori. Autophagy 8:1045–1057. https://doi.org/10.4161/auto.20159
Wang F, Meng W, Wang B, Qiao L (2014) Helicobacter pylori-induced gastric inflammation and gastric cancer. Cancer Lett 345:196–202. https://doi.org/10.1016/j.canlet.2013.08.016
Warren JR, Marshall BJ (1983) Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet 1:1273–1275
Xie Z, Klionsky DJ (2007) Autophagosome formation: core machinery and adaptations. Nat Cell Biol 9:1102–1109. https://doi.org/10.1038/ncb1007-1102
Zhang LQ, Zhao GZ, Xu XY, Fang J, Chen JM, Li JW, Gao XJ, Hao LJ, Chen YZ (2015) Integrin-β1 regulates chondrocyte proliferation and apoptosis through the upregulation of GIT1 expression. Int J Mol Med 35:1074–1080. https://doi.org/10.3892/ijmm.2015.2114
Zhao H, Chen H, Xiaoyin M, Yang G, Hu Y, Xie K, Yu Y (2019) Autophagy activation improves lung injury and inflammation in sepsis. Inflammation 42:426–439. https://doi.org/10.1007/s10753-018-00952-5
Acknowledgements
All authors thank the Medical Research Center of Binzhou Medical University for providing large-scale instrument use platforms, and the Department of Pathogen Biology, School of Basic Medical Sciences, Binzhou Medical University for providing experimental venues.
Funding
This work was supported by the National Natural Science Foundation of China under Grant [Grant Nos. 82172560 and 81771709], Shandong Provincial Education Department under Grant [Grant No. 2019KJK012] and Development Project of Medicine and Health Science Technology of Shandong Province under Grant [Grant No. 2019WS311].
Author information
Authors and Affiliations
Contributions
BL: conceived and designed study; QR: performed research, analyzed data; YD: wrote the paper; RZ: performed data collection and management; JL: contributed to revision of the manuscript; XT: contributed to revision of the manuscript; LG: contributed new methods or models; YZ: conceived of or designed study; contributed new methods/models. All authors have read and approved to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The clinical gastric tissue samples used in this study were from the outpatients in the gastroscope room of the Department of Gastroenterology of Rongcheng hospital, Weihai, Shandong Province, and voluntarily participated in sample collection when the patients are informed.
Consent for publication
Yes.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, B., Rong, Q., Du, Y. et al. Regulation of β1-integrin in autophagy and apoptosis of gastric epithelial cells infected with Helicobacter pylori. World J Microbiol Biotechnol 38, 12 (2022). https://doi.org/10.1007/s11274-021-03199-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03199-9