Abstract
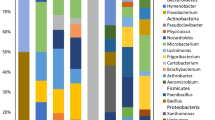
The aim of this study was to clarify effects of soil and climatic conditions on community structure of sweet potato bacterial endophytes by applying locked nucleic acid oligonucleotide-PCR clamping technique and metagenomic analysis. For this purpose, the soil samples in three locations were transferred each other and sweet potato nursery plants from the same farm were cultivated for ca. 3 months. After removal of plastid, mitochondria and undefined sequences, the averaged numbers of retained sequences and operational taxonomic units per sample were 20,891 and 846, respectively. Proteobacteria (85.0%), Bacteroidetes (6.6%) and Actinobacteria (6.3%) were the three most dominant phyla, accounting for 97.9% of the reads, and γ-Proteobacteria (66.3%) being the most abundant. Top 10 genera represented 81.2% of the overall reads in which Pseudomonas (31.9–45.0%) being the most predominant. The overall endophytic bacterial communities were similar among the samples which indicated that the soil and the climatic conditions did not considerably affect the entire endophytic community. The original endophytic bacterial community might be kept during the cultivation period.





Similar content being viewed by others
References
Amann RI, Ludwig W, Schleifer KH (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59:143–169
Amplicon PCR, Clean-Up PCR, Index PCR (2013) 16S metagenomic sequencing library preparation, pp 1–28
Andreote FD, De Araújo WL, De Azevedo JL et al (2009) Endophytic colonization of potato (Solanum tuberosum L.) by a novel competent bacterial endophyte, Pseudomonas putida strain P9, and its effect on associated bacterial communities. Appl Environ Microbiol 75:3396–3406
Duffy BK, Défago G (1999) Environmental factors modulating antibiotic and siderophore biosynthesis by Pseudomonas fluorescens biocontrol strains. Appl Environ Microbiol 65:2429–2438
Gaiero JR, McCall CA, Thompson KA et al (2013) Inside the root microbiome: bacterial root endophytes and plant growth promotion. Am J Bot 100:1738–1750
Garbeva P, Van Overbeek LS, Van Vuurde JWL et al (2001) Analysis of endophytic bacterial communities of potato by plating and denaturing gradient gel electrophoresis (DGGE) of 16S rDNA based PCR fragments. Microb Ecol 41:369–383
Gottel NR, Castro HF, Kerley M et al (2011) Distinct microbial communities within the endosphere and rhizosphere of Populus deltoides roots across contrasting soil types. Appl Environ Microbiol 77:5934–5944
Huong NL, Itoh K, Suyama K (2007) Diversity of 2,4-dichlorophenoxyacetic acid (2,4-d) and 2,4,5-trichlorophenoxyacetic acid (2,4,5-T)-degrading bacteria in Vietnamese soils. Microbes Environ 22:243–256
Ikenaga M, Sakai M (2014) Application of Locked Nucleic Acid (LNA) oligonucleotide–PCR clamping technique to selectively PCR amplify the SSU rRNA genes of bacteria in investigating the plant-associated community structures. Microbes Environ 29:286–295
Ikenaga M, Tabuchi M, Oyama T et al (2015) Development of LNA oligonucleotide-PCR clamping technique in investigating the community structures of plant-associated bacteria. Biosci Biotechnol Biochem 79:1556–1566
Jackson CR, Randolph KC, Osborn SL et al (2013) Culture dependent and independent analysis of bacterial communities associated with commercial salad leaf vegetables. BMC Microbiol 13:1–12
Jaric M, Segal J, Silva-Herzog E et al (2013) Better primer design for metagenomics applications by increasing taxonomic distinguishability. BMC Proc 7:1–10
Khan Z, Doty SL (2009) Characterization of bacterial endophytes of sweet potato plants. Plant Soil 322:197–207
Klindworth A, Pruesse E, Schweer T et al (2013) Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res 41:1–11
Magnani GS, Cruz LM, Weber H et al (2013) Culture-independent analysis of endophytic bacterial communities associated with Brazilian sugarcane. Genet Mol Res 12:4549–4558
Marques JM, da Silva TF, Vollu RE et al (2015) Bacterial endophytes of sweet potato tuberous roots affected by the plant genotype and growth stage. Appl Soil Ecol 96:273–281
Miller CM, Miller RV, Garton-Kenny D et al (1998) Ecomycins, unique antimycotics from Pseudomonas viridiflava. J Appl Microbiol 84:937–944
Moronta-Barrios F, Gionechetti F, Pallavicini A et al (2018) Bacterial microbiota of rice roots: 16S-based taxonomic profiling of endophytic and rhizospheric diversity, endophytes isolation and simplified endophytic community. Microorganisms 6:14
Oliver JD (2010) Recent findings on the viable but nonculturable state in pathogenic bacteria. FEMS Microbiol Rev 34:415–425
Oteino N, Lally RD, Kiwanuka S et al (2015) Plant growth promotion induced by phosphate solubilizing endophytic Pseudomonas isolates. Front Microbiol 6:1–9
Pei C, Mi C, Sun L et al (2017) Diversity of endophytic bacteria of Dendrobium officinale based on culture-dependent and culture-independent methods. Biotechnol Biotechnol Equip 31:112–119
Pereira P, Ibáñez F, Rosenblueth M et al (2011) Analysis of the bacterial diversity associated with the roots of maize (Zea mays L.) through culture-dependent and culture-independent methods. ISRN Ecol 2011:1–10
Puri RR, Dangi SR, Dhungana SA et al (2018a) Diversity and plant growth promoting ability of culturable endophytic bacteria in Nepalese sweet potato. Adv Microbiol 8:734–761
Puri RR, Adachi F, Omichi M et al (2018b) Culture-dependent analysis of endophytic bacterial community of sweet potato (Ipomoea batatas) in different soils and climates. J Adv Microbiol 13:1–12
Rediers H, Bonnecarrère V, Rainey PB et al (2003) Development and application of a dapB-based in vivo expression technology system to study colonization of rice by the endophytic nitrogen-fixing bacterium Pseudomonas stutzeri A15. Appl Environ Microbiol 69:6864–6874
Ryan RP, Germaine K, Franks A et al (2008) Bacterial endophytes: recent developments and applications. FEMS Microbiol Lett 278:1–9
Sato T, Matsumoto K, Okumura T et al (2008) Isolation of lactate-utilizing butyrate-producing bacteria from human feces and in vivo administration of Anaerostipes caccae strain L2 and galacto-oligosaccharides in a rat model. FEMS Microbiol Ecol 66:528–536
Shen SY, Fulthorpe R (2015) Seasonal variation of bacterial endophytes in urban trees. Front Microbiol 6:1–13
Shi Y, Yang H, Zhang T et al (2014) Illumina-based analysis of endophytic bacterial diversity and space-time dynamics in sugar beet on the north slope of Tianshan mountain. Appl Microbiol Biotechnol 98:6375–6385
Soil Survey Staff (1999) Soil taxonomy—a basic system of soil classification for making and interpreting soil surveys. USDA Agriculture Handbook No. 436. U.S. Government Publishing Office, Washington DC, pp 1–754
Subramanian P, Mageswari A, Kim K et al (2015) Psychrotolerant endophytic Pseudomonas sp. strains OB155 and OS261 induced chilling resistance in tomato plants (Solanum lycopersicum Mill.) by activation of their antioxidant capacity. Mol Plant Microbe Interact 28:1073–1081
Taghavi S, Garafola C, Monchy S et al (2009) Genome survey and characterization of endophytic bacteria exhibiting a beneficial effect on growth and development of poplar trees. Appl Environ Microbiol 75:748–757
Tian B, Zhang C, Ye Y et al (2017) Beneficial traits of bacterial endophytes belonging to the core communities of the tomato root microbiome. Agric Ecosyst Environ 247:149–156
Torsvik V, Salte K, Sørheim R R et al (1990) Comparison of phenotypic diversity and DNA hererogeneity in a population of soil bacteria. Appl Environ Microbiol 56:776–781
Weisburg WG, Barns SM, Pelletier DA et al (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Xia Y, DeBolt S, Dreyer J et al (2015) Characterization of culturable bacterial endophytes and their capacity to promote plant growth from plants grown using organic or conventional practices. Front Plant Sci 6:1–10
Yang R, Liu P, Ye W (2017) Illumina-based analysis of endophytic bacterial diversity of tree peony (Paeonia Sect. Moutan) roots and leaves. Braz J Microbiol 48:695–705
Acknowledgements
This study was supported in part by a Grant-in-Aid for Scientific Research (B) [16KT0032] from the Japan Society for the Promotion of Science (JSPS). We are thankful to Professor Makoto Ikenaga, Faculty of Agriculture, Kagoshima University for providing the information of mitochondrial primer sequence. We are also grateful to Laboratory of soil and ecological engineering, Shimane University for soil analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Puri, R.R., Adachi, F., Omichi, M. et al. Metagenomic study of endophytic bacterial community of sweet potato (Ipomoea batatas) cultivated in different soil and climatic conditions. World J Microbiol Biotechnol 35, 176 (2019). https://doi.org/10.1007/s11274-019-2754-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-019-2754-2