Abstract
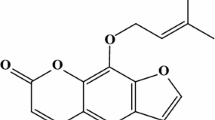
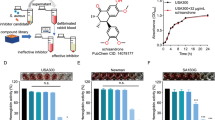
α-Toxin, a pore-forming toxin secreted by most Staphylococcus aureus, plays critical role in the pathogenesis associated with various infectious diseases. The USA300 which is a major international epidemic methicilin-resisrant S. aureus has spread rapidly to multiple countries and become an emerging public health concern. In this study, the in vitro efficacy of Dracorhodin Perochlorate (DP) against USA300 virulence was evaluated. Using susceptibility testing, immunoblots, rabbit blood haemolytic assay and real-time RT-PCR, we observed that the α-toxin production was decreased when USA300 was co-cultured with different sub-inhibitory concentration of DP. Further, the protective effect of DP against USA300-mediated injury of human alveolar epithelial cells (A549) and MH-S cells was evaluated by cytotoxicity assays, and the result revealed that DP, at final concentration of 16 µg/ml, is a potent antagonist for USA300-mediated cell damage. Importantly, those beneficial effects might partially correlate with hla and RNAIII suppression by DP, leading to the inhibition of α-toxin production in culture supernatant. Overall, these results suggest that DP could attenuate the virulence of USA300 by decreasing α-toxin production without inhibiting bacterial growth, and this compound may represent an ideal candidate for the development of anti-virulence agent combating S. aureus infection.




Similar content being viewed by others
References
Abdelhady W et al (2013) Reduced vancomycin susceptibility in an in vitro catheter-related biofilm model correlates with poor therapeutic outcomes in experimental endocarditis due to methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother 57:1447–1454. doi:10.1128/AAC.02073-12
Berube BJ, Bubeck Wardenburg J (2013) Staphylococcus aureus alpha-toxin: nearly a century of intrigue. Toxins 5:1140–1166
Bubeck Wardenburg J, Patel RJ, Schneewind O (2007) Surface proteins and exotoxins are required for the pathogenesis of Staphylococcus aureus pneumonia. Infect Immun 75:1040–1044. doi:10.1128/IAI.01313-06
Chen Y et al (2015) Basis of virulence in a Panton-Valentine leukocidin-negative community-associated methicillin-resistant Staphylococcus aureus strain. J Infect Dis 211:472–480. doi:10.1093/infdis/jiu462
Daly SM et al (2015) omega-Hydroxyemodin limits Staphylococcus aureus quorum sensing-mediated pathogenesis and inflammation. Antimicrob Agents Chemother 59:2223–2235. doi:10.1128/AAC.04564-14
DeLeo FR, Otto M, Kreiswirth BN, Chambers HF (2010) Community-associated meticillin-resistant Staphylococcus aureus. Lancet 375:1557–1568. doi:10.1016/S0140-6736(09)61999-1
Diekema DJ et al (2014) Continued emergence of USA300 methicillin-resistant Staphylococcus aureus in the United States: results from a nationwide surveillance study. Infect Control Hosp Epidemiol 35:285–292. doi:10.1086/675283
Enright MC, Robinson DA, Randle G, Feil EJ, Grundmann H, Spratt BG (2002) The evolutionary history of methicillin-resistant Staphylococcus aureus (MRSA). Proc Natl Acad Sci USA 99:7687–7692. doi:10.1073/pnas.122108599
Foster TJ, Geoghegan JA, Ganesh VK, Hook M (2014) Adhesion, invasion and evasion: the many functions of the surface proteins of Staphylococcus aureus. Nat Rev Microbiol 12:49–62. doi:10.1038/nrmicro3161
Gould IM, David MZ, Esposito S, Garau J, Lina G, Mazzei T, Peters G (2012) New insights into meticillin-resistant Staphylococcus aureus (MRSA) pathogenesis, treatment and resistance. Int J Antimicrob Agents 39:96–104. doi:10.1016/j.ijantimicag.2011.09.028
Hua L et al (2014) Assessment of an anti-alpha-toxin monoclonal antibody for prevention and treatment of Staphylococcus aureus-induced pneumonia. Antimicrob Agents Chemother 58:1108–1117. doi:10.1128/AAC.02190-13
Khodaverdian V et al (2013) Discovery of antivirulence agents against methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother 57:3645–3652. doi:10.1128/AAC.00269-13
Kobayashi SD et al (2011) Comparative analysis of USA300 virulence determinants in a rabbit model of skin and soft tissue infection. J Infect Dis 204:937–941. doi:10.1093/infdis/jir441
Mediavilla JR, Chen L, Mathema B, Kreiswirth BN (2012) Global epidemiology of community-associated methicillin resistant Staphylococcus aureus (CA-MRSA). Curr Opin Microbiol 15:588–595. doi:10.1016/j.mib.2012.08.003
Rigby KM, DeLeo FR (2012) Neutrophils in innate host defense against Staphylococcus aureus infections. Semin Immunopathol 34:237–259. doi:10.1007/s00281-011-0295-3
Seybold U et al (2006) Emergence of community-associated methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of health care-associated blood stream infections. Clin Infect Dis 42:647–656. doi:10.1086/499815
Skov R, Christiansen K, Dancer SJ, Daum RS, Dryden M, Huang YC, Lowy FD (2012) Update on the prevention and control of community-acquired meticillin-resistant Staphylococcus aureus (CA-MRSA). Int J Antimicrob Agents 39:193–200. doi:10.1016/j.ijantimicag.2011.09.029
Stefani S, Chung DR, Lindsay JA, Friedrich AW, Kearns AM, Westh H, Mackenzie FM (2012) Meticillin-resistant Staphylococcus aureus (MRSA): global epidemiology and harmonisation of typing methods. Int J Antimicrob Agents 39:273–282. doi:10.1016/j.ijantimicag.2011.09.030
Strommenger B et al (2014) Evolution of methicillin-resistant Staphylococcus aureus towards increasing resistance. J Antimicrob Chemother 69:616–622. doi:10.1093/jac/dkt413
Sully EK et al (2014) Selective chemical inhibition of agr quorum sensing in Staphylococcus aureus promotes host defense with minimal impact on resistance. PLoS Pathog 10:e1004174. doi:10.1371/journal.ppat.1004174
Tavares A et al (2014) Insights into alpha-hemolysin (Hla) evolution and expression among Staphylococcus aureus clones with hospital and community origin. PLoS One 9:e98634. doi:10.1371/journal.pone.0098634
van Hal SJ, Fowler VG Jr (2013) Is it time to replace vancomycin in the treatment of methicillin-resistant Staphylococcus aureus infections? Clin Infect Dis 56:1779–1788. doi:10.1093/cid/cit178
Watkins RR, David MZ, Salata RA (2012) Current concepts on the virulence mechanisms of meticillin-resistant Staphylococcus aureus. J Med Microbiol 61:1179–1193. doi:10.1099/jmm.0.043513-0
Xia M, Wang D, Wang M, Tashiro S, Onodera S, Minami M, Ikejima T (2004) Dracorhodin perchlorate induces apoptosis via activation of caspases and generation of reactive oxygen species. J Pharmacol Sci 95:273–283
Yu JH et al (2013) Dracorhodin perchlorate induced human breast cancer MCF-7 apoptosis through mitochondrial pathways. Int J Med Sci 10:1149–1156. doi:10.7150/ijms.6275
Yu G, Kuo D, Shoham M, Viswanathan R (2014) Combinatorial synthesis and in vitro evaluation of a biaryl hydroxyketone library as antivirulence agents against MRSA. ACS Comb Sci 16:85–91. doi:10.1021/co400142t
Zhou X et al (2015) Phloretin derived from apple can reduce alpha-hemolysin expression in methicillin-resistant Staphylococcus aureus USA300. World J Microbiol Biotechnol 31:1259–1265. doi:10.1007/s11274-015-1879-1
Acknowledgments
This work was supported by the National Basic Research Program of China (Grant 2013CB127205) and the National Nature Science Foundation of China (Grant 31130053). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animals rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Liu, Y., Shi, D., Guo, Y. et al. Dracorhodin Perochlorate attenuates Staphylococcus aureus USA300 virulence by decreasing α-toxin expression. World J Microbiol Biotechnol 33, 17 (2017). https://doi.org/10.1007/s11274-016-2129-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-016-2129-x