Abstract
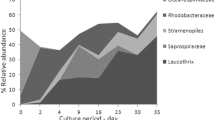
The quantity and composition of the bacterial microbiota in the rearing water, sediment, gills and intestines of tilapia Oreochromis niloticus collected every 2 weeks from Day 30 to Day 120 after stocking for grow-out culture in 6 earthen brackish water ponds in the Philippines were examined. The total heterotrophic aerobic bacterial counts obtained in the water, sediment, gills and intestines of tilapia ranged from 103 to 104 c.f.u. ml−1, 103–105, 105–107 and 104–107 c.f.u. g−1, respectively. In terms of composition, a total of 20 bacterial genera and 31 species were identified with the preponderance of gram-negative bacteria constituting 84 % of all bacterial isolates examined. Aeromonas hydrophila, Bacillus spp., Plesiomonas shigelloides, Shewanella putrefaciens, Pseudomonas fluorescens, Staphylococcus spp. and Vibrio cholerae were the dominant bacteria identified in the gills and intestine of tilapia. These bacteria also dominated in the pond sediment and rearing water, except for the nil isolation of S. putrefaciens and V. cholerae in the water samples examined, indicating that resident bacteria in the pond water and sediment congruently typify the composition of bacterial microbiota in the gills and intestine of tilapia which under stressful conditions may propel the ascendance of disease epizootics.




Similar content being viewed by others
References
Al-Harbi AH, Uddin N (2003) Quantitative and qualitative studies on bacterial flora of hybrid tilapia (Oreochromis niloticus × Oreochromis aureus) cultured in earthen ponds in Saudi Arabia. Aquac Res 34:43–48
Al-Harbi AH, Uddin N (2004) Seasonal variation in the intestinal bacterial flora of hybrid tilapia (Oreochromis niloticus × Oreochromis aureus) cultured in earthen ponds in Saudi Arabia. Aquaculture 229:37–44
Al-Harbi AH, Uddin N (2005) Bacterial diversity of tilapia (Oreochromis niloticus) cultured in brackish water in Saudi Arabia. Aquaculture 250:566–572
Aly SM, Abd-El-Rahman AM, John G, Mohamed MF (2008) Characterization of some bacteria isolated from Oreochromis niloticus and their potential use as probiotics. Aquaculture 277:1–6
Apun-Molina JP, Santamaria-Miranda AP, Luna-Gonzalez A, Martinez-Dias SF, Rojas-Contreras M (2009) Effect of potential probiotic bacteria on growth and survival of tilapia Oreochromis niloticus L., cultured in the laboratory under high density and suboptimum temperature. Aquac Res 40:887–894
Austin B, Austin DA (1993) Bacterial fish pathogens, disease in farmed and wild fish, 2nd edn. Ellis Horwood, Chichester, England
Baliao DD, Dosado NS (2011) Tilapia cage farming in freshwater reservoir using artificial diets during dry and wet seasons. Ph Agri Sci 94:38–45
Bisharat N, Raz R (1996) Vibrio infection in Israel due to changes in fish marketing. Lancet 348:1586
Bonner JR, Coker AS, Berryman CR, Pollock HM (1983) Spectrum of Vibrio infections in a Gulf Coast community. Ann Intern Med 99:464–469
Chen YS, Liu YC, Yen MY, Wang JH, Wann SR, Cheng DL (1997) Skin and soft-tissue manifestations of Shewanella putrefaciens infection. Clin Infect Dis 25:225–229
Chowdhury MBR, Muniruzzaman M, Uddin MN (1989) Study on the intestinal bacterial flora of tilapia Oreochromis niloticus. Bangladesh J Aquacult 11:65–70
Dan M, Gutman R, Biro A (1992) Peritonitis caused by Pseudomonas putrefaciens in patients undergoing continuous ambulatory peritoneal dialysis. Clin Infect Dis 14:359–360
Dey MM, Paraguas FJ, Bimbao GB, Regaspi PB (2000) Technical efficiency of tilapia grow out pond operations in the Philippines. Aquacult Econ Manag 4:33–47
Gram L (1992) Evaluation of the bacteriological quality of seafood. Int J Food Microbiol 16:25–39
Hlady WG, Klontz KC (1996) The epidemiology of Vibrio infections in Florida, 1981–1993. J Infect Dis 173:1176–1183
Holmes B, Lapage SP, Malnick H (1975) Strains of Pseudomonas putrefaciens from clinical material. J Clin Pathol 28:149–155
Holt JG, Krieg NR, Sneath PHA, Williams ST (1994) Bergey’s Manual of Determinative Bacteriology, 9th ed. Williams and Wilkins, Baltimore, USA
Jørgensen BR, Huss HH (1989) Growth and activity of Shewanella putrefaciens isolated from spoiling fish. Int J Food Microbiol 9:51–62
Kirov SM (2001) Aeromonas and Plesiomonas species. In: Doyle MP, Beuchat LP, Montville TJ (eds) Food microbiology: fundamentals and frontiers, 2nd edn. American Society for Microbiology Press, Washington
Lu S, Levin RE (2010) Shewanella in a tilapia fish farm. J Fish Sci Com 4:159–170
Mahmud Z, Wright A, Mandal SC, Dai J, Jones MK, Hasan M, Rashid MH, Islam MS, Johnson JA, Gulig PA, Morris JG Jr, Ali A (2010) Genetic characterization of Vibrio vulnificus strains from tilapia aquaculture in Bangladesh. Appl Environ Microbiol 76:4890–4895
Maugeri TL, Caccamo D, Gugliandolo C (2000) Potentially pathogenic vibrios in brackish waters and mussels. J Appl Microbiol 89:261–266
Mohanty BR, Sahoo PK (2007) Edwardsiellosis in fish: a brief review. J Biosci 32:1331–1344
Morris JG Jr (2003) Cholera and other types of vibriosis: a story of human pandemics and oysters on the half shell. Food Saf 37:272–280
Oliver JD (2005) Wound infections caused by Vibrio vulnificus and other marine bacteria. Epidemiol Infect 133:383–391
Pagani L, Lang A, Vedovelli C, Moling O, Rimenti G, Pristera R, Mian P (2003) Soft-tissue infection and bacteremia caused by Shewanella putrifaciens. J Clin Microbiol 41:2240–2241
Pakingking R Jr, Takano R, Nishizawa T, Mori K, Iida Y, Arimoto M, Muroga K (2003) Experimental coinfection with aquabirnavirus and viral hemorrhagic septicemia virus (VHSV), Edwardsiella tarda or Streptococcus iniae in Japanese flounder Paralichthys olivaceus. Fish Pathol 38:15–21
Rheinheimer G (1985) Aquatic microbiology, 3rd edn. University of Kiel, West Germany, Wiley, Chichester, New York, Brisbane, Toronto
Romana-Eguia MRR, Eguia RV (1999) Growth of five Asian red tilapia strains in saline environments. Aquaculture 173:161–170
Romana-Eguia MRR, Ikeda M, Basiao ZU, Taniguchi N (2004) Genetic diversity in farmed Asian Nile and red hybrid tilapia stocks evaluated from microsatellite and mitochondrial DNA analysis. Aquaculture 236:131–150
Sakata T, Uno K, Kakimoto D (1984) Dominant bacteria of the aerobic microflora in tilapia intestine. Bull Jpn Soc Sci Fish 50:489–493
Sugita H, Ishida Y, Deguchi Y, Kadota H (1982) Aerobic microflora attached to the wall surface in the gastrointestine of Tilapia nilotica. Bull Coll Agric Vet Med Nih Univ 39:302–306
Yambot AV (1998) Isolation of Aeromonas hydrophila from Oreochromis niloticus during fish disease outbreaks in the Philippines. Asian Fish Sci 10:347–354
Yohe S, Fishbain JT, Andrew M (1997) Shewanella putrefaciens abscess of the lower extremity. J Clin Microbiol 35:33–36
Zaidenstein R, Sadik C, Lerner L, Valinsky L, Kopelowitz J, Yishai R, Agmon V, Persons M, Bopp C, Weinberger M (2008) Clinical characteristics and molecular subtyping of V. vulnificus illnesses, Israel. Emerg Infect Dis 14:1875–1882
Acknowledgments
This study was funded by the Department of Science and Technology-National Research Council of the Philippines (DOST-NRCP) (NRCP Project No. E-225) and partly by SEAFDEC AQD (Study Code: FH02-F2013-T). We would like to thank Mr. Eric Ledesma and the laboratory staff of NPPMCI for the invaluable assistance during our sampling.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pakingking, R., Palma, P. & Usero, R. Quantitative and qualitative analyses of the bacterial microbiota of tilapia (Oreochromis niloticus) cultured in earthen ponds in the Philippines. World J Microbiol Biotechnol 31, 265–275 (2015). https://doi.org/10.1007/s11274-014-1758-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-014-1758-1