Abstract
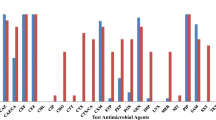
A total of 21 isolates of Vibrio cholerae non-O1 strains were isolated from three wastewater treatment plants in Agadir, Morocco. The isolates were analyzed by biochemical analysis, antibiogram, pulsed-field gel electrophoresis and the MALDI-TOF patterns of their protein masses were compared. Over 67% of isolates were susceptible to antimicrobial agents tested and 14% proved resistant to both trimethoprim-sulfamethoxazole and nalidixic acid. Typing by pulsed-field gel electrophoresis with NotI digestion revealed that the V. cholerae non-O1 strains from Agadir (Morocco) have a lower level of genetic homogeneity, the restriction patterns of whole-chromosomal DNA grouped the V. cholerae O1 and V. alginolyticus strains into a separate cluster from V. metschnikovii and V. cholerae non-O1 isolates. Furthermore, to gain additional analytical accuracy and reliability in the analysis we used dendrogram based on MALDI-TOF spectral patterns generated by the BioTyper 1.1™ software. All m/z signatures of all strains tested indicate that the mass spectral data contained sufficient information to distinguish between strains of V. cholerae.



Similar content being viewed by others
References
Albert M, Anderson L, Varkey JB, Petti CA, Liddle RA, Frothingham R, Woods CW (2004) Non-O1 Vibrio cholerae septicemia: case report, discussion of literature, and relevance to bioterrorism. Diagn Microbiol Infect Dis 49:295–297
Amahmid O, Asmama S, Bouhoum K (2002) Urban wastewater treatment in stabilization pond: occurrence and removal of pathogens. Urban Water 4:255–262
Annick RP, Baron S, Lesne J, Fournier JM, Quilici ML (2006) Specific detection of Vibrio cholerae in marine ecosystem by a colony hybridization assay after culture on selective medium. Hydroecol Appl 15:97–105
Arita M, Takeda T, Honda T, Miwatani T (1986) Purification and characterization of Vibrio cholerae non-O1 heat-stable enterotoxin. Infect Immun 52:45–49
Aström J, Carlander A, Sahlén K, Stenström TA (2006) Fecal indicator and pathogen reduction in vegetation microcosms. Water Air Soil Poll 176:375–387
Baquero F, Blazquez J (1997) Evolution of antibiotic resistance. Trends Ecol Evol 12:482
Bright JJ, Claydon MA, Soufian M, Gordon DB (2002) Rapid typing of bacteria using matrix-assisted laser desorption ionisation time-of-flight mass spectrometry and pattern recognition software. J Microbiol Methods 48:127–138
Burrus V, Marrero J, Waldor MK (2006) The current ICE age: biology and evolution of SXT-related integrating conjugative elements. Plasmid 55:173–183
Chauret C, Springthorpe S, Sattar S (1999) Fate of Cryptosporidium oocysts, Giardia cysts, and microbial indicators during wastewater treatment and anaerobic sludge digestion. Can J Microbiol 45:257–262
Chun J, Huq A, Colwell RR (1999) Analysis of 16S–23S rRNA Intergenic spacer regions of Vibrio cholerae and Vibrio mimicus. Appl Environ Microbiol 65:2202–2208
Claydon MA, Davey SN, Edwards-Jones V, Gordon DB (1996) The rapid identification of intact microorganisms using mass spectrometry. Nat Biotechnol 14:1584–1586
Colwell RR, Kaper J (1977) Vibrio species as bacterial indicators of potential health hazards associated with water. In: Hoadley AW, Dutka BJ (ed) Bacterial indicators/health hazards associated with water. ASTM STP 635, Philadelphia, pp 115–125
Colwell RR, Spira WM (1992) The ecology of Vibrio cholerae. In: Barua D, Greenough WB III (eds) Cholera. Plenum Medical, New York, pp 107–127
Dieckmann R, Strauch E, Alter T (2010) Rapid identification and characterization of Vibrio species using whole-cell MALDI-TOF mass spectrometry. J Appl Microbiol 109:199–211
Dupont MJ, Jouvenot M, Couetdic G, Michel-Briand Y (1985) Development of plasmid-mediated resistance in Vibrio cholerae during treatment with trimethoprim-sulfamethoxazole. Antimicrob Agents Chemother 27:280–281
Eddabra R, Mimouni R, Meunier O, Moussaoui W, Prévost G, Scheftel JM (2010) MALDI-TOF MS analysis and molecular typing by pulsed field gel electrophoresis of environmental Vibrio isolates. Proceedings of ECCMID 20th, Vienna, Austria
Faruque SM, Chowdhury N, Kamruzzaman M, Dziejman M, Hasibu Rahman M, Sack DA, Balakrish Nair G, Mekalanos JJ (2004) Genetic diversity and virulence potential of environmental Vibrio cholerae population in a cholera-endemic area. PNAS (Proc Natl Acad Sci USA) 101:2123–2128
Filetici E, Bonadonna L, Ciccozzi M, Anastasio MP, FantasiaM Shimada T (1997) Phenotypic and genotypic biotyping of environmental strains of Vibrio cholerae non-O1 isolated in Italy. Appl Environ Microbiol 63:4102–4106
Gerba CP, Smith JE (2005) Sources of pathogenic microorganisms and their fate during land application of wastes. J Environ Qual 34:42–48
Gilbride KA, Lee DY, Beaudette LA (2006) Molecular techniques in wastewater: understanding microbial communities, detecting pathogens, and real-time process control. J Microbiol Meth 66:1–20
Godfree A, Farrell J (2005) Processes for managing pathogens. J Environ Qual 34:105–113
Hazen TH, Martinez RJ, Chen Y, Lafon PC, Garrett NM, Parsons MB, Bopp CA, Sullards MC, Sobecky PA (2009) Rapid identification of Vibrio parahaemolyticus by whole-cell matrix-assisted laser desorption ionization-time of flight mass spectrometry. Appl Environ Microbiol 75:6745–6756
Honisch C, Chen Y, Mortimer C, Arnold Oliver Schmidt C, Van Den Boom D, Cantor CR, Shah HN, Gharbia SE (2007) Automated comparative sequence analysis by base-specific cleavage and mass spectrometry for nucleic acid-based microbial typing. Proc Natl Acad Sci USA 104:10649–10654
Jiang SC, Louis V, Choopun N, Sharma A, Huq A, Colwell RR (2000) Genetic diversity of Vibrio cholerae in Chesapeake Bay determined by amplified fragment length polymorphism fingerprinting. Appl Environ Microbiol 66:140–147
Keys CJ, Dare DJ, Sutton H, Wells G, Lunt M, McKenna T, McDowall M, Shah HN (2004) Compilation of a MALDI-TOF mass spectral database for the rapid screening and characterisation of bacteria implicated in human infectious diseases. Infect Genet Evol 4:221–242
Lay JO Jr (2000) MALDI-TOF mass spectrometry and bacterial taxonomy. Trends Anal Chem 19:507–516
Lesne J, Baleux B, Boussaid A, Hassani L (1991) Dynamics of non-O1 Vibrio cholerae in experimental sewage stabilization ponds under arid mediterranean climate. Water Sci Technol 24:387–390
Levy SB (2001) Antibiotic resistance: consequences of inaction. Clin Infect Dis 33(Suppl 3):S124–S129
Louis VR, Russek-Cohen E, Choopun N, Rivera ING, Gangle B, Jiang SC, Rubin A, Patz JA, Huq A, Colwell RR (2003) Predictability of Vibrio cholerae in Chesapeake Bay. Appl Environ Microbiol 69:2773–2785
Melano R, Petroni A, Garutti A, Saka HA, Mange L, Pasterán F, Rapoport M, Rossi A, Galas M (2002) New Carbenicillin-Hydrolyzing β-Lactamase (CARB-7) from Vibrio cholerae Non-O1, Non-O139 Strains Encoded by the VCR Region of the V. cholerae Genome. Antimicrob Agent Chemother 46:2162–2168
Mellmann A, Bimet F, Bizet C, Borovskaya AD, Drake RR, Eigner U, Fahr AM, He Y, Ilina EN, Kostrzewa M, Maier T, Mancinelli L, Moussaoui W, Prévost G, Putignani L, Seachord CL, Tang YW, Harmsen D (2009) High interlaboratory reproducibility of matrix-assisted laser desorption ionization time of light mass spectrometry-based species identification of non fermenting bacteria. J Clin Microbiol 47:3732–3734
Mezrioui N, Oufdou K (1996) Abundance and antibiotic resistance of non-O1 Vibrio cholerae, strains in domestic wastewater before and after treatment in stabilization ponds in an arid region (Marrakech, Morocco). FEMS Microbiol Ecol 21:277–284
Mezrioui N, Oufdou K, Baleux B (1995) Dynamics of non-O1 Vibrio cholerae and fecal coliforms in experimental stabilization ponds in the arid region of Marrakech, Morocco, and the effect of pH, temperature, and sunlight on their experimental survival. Can J Microbiol 41:489–498
Mimouni R, Eddabra R, Benzine L, Aarab D, Cherradi MA, Moukrim A (2006) Qualité Microbiologique et Physico-chimique des Eaux Usées de Ben Sergao Epurées par Infiltration Percolation après Quinze Ans de Fonctionnement de la Station (Sud-Ouest, Maroc). Proceedings of 3rd international conference on water resource Mediter Bassin, Liban
Morris JG Jr, Drusano GL, Tenney JH (1985) In vitro susceptibility of pathogenic Vibrio species to norfloxacin and six other antimicrobial agents. Antimicrob Agents Chemother 28:442–445
Morris JG (1990) Non-O group 1 Vibrio cholerae: a look at the epidemiology of an occasional pathogen. Epidemiol Rev 12:179–191
Moussaoui W, Bouakaze C, Prevost G (2009) Applications de la spectrographie de masse MALDI TOF à l’identification bactérienne. Bull Soc Fr Microbiol 24:293–302
Ryzhov V, Fenselau C (2001) Characterization of the protein subset desorbed by MALDI from whole bacterial cells. Anal Chem 73:746–750
Sauer S, Freiwald A, Maier T, Kube M, Reinhardt R, Kostrzewa M, Geider K (2008) Classification and identification of bacteria by mass spectrometry and computational analysis. PloS ONE 3(7):e2843. doi:10.1371/journal.pone.0002843
Sciortino CV, Johnson JA, Hamad A (1996) Vitek system antimicrobial susceptibility testing of O1, O139, and non-O1 Vibrio cholerae. J Clin Microbiol 34:897–900
Shannon KE, Lee DY, Trevors JT, Beaudette LA (2007) Application of real-time quantitative PCR for the detection of selected bacterial pathogens during municipal wastewater treatment. Sci Total Environ 382:121–129
Sharma C, Thungapathra M, Ghosh A, Mukhopadhyay AK, Basu A, Mitra R, Basu I, Bhattacharya SK, Shimada T, Ramamurthy T, Takeda T, Yamasaki S, Takeda Y, Balakrish Nair G (1998) Molecular analysis of non-O1, non-O139 Vibrio cholerae associated with an unusual upsurge in the incidence of cholera-like disease in Calcutta, India. J Clin Microbiol 36:756–763
Singh DV, Matte MH, Matte GR, Jiang S, Sabeena F, Shukla BN, Sanyal SC, Huq A, Colwell RR (2001) Molecular analysis of Vibrio cholerae O1, O139, non-O1, and non-O139 strains: clonal relationships between clinical and environmental isolates. Appl Environ Microbiol 67:910–921
Soussy CJ (2005) Comité de l’Antibiogramme de la Société Française de Microbiologie. http://www.sfm.asso.fr/
Suh MJ, Limbach PA (2004) Investigation of methods suitable for the matrix- assisted laser desorption/ionization mass spectrometric analysis of proteins from ribonucleoprotein complexes. Eur J Mass Spectrom 10:89–99
Sur D, Dutta S, Sarkar BL, Manna B, Bhattacharya MK, Datta KK, Saha A, Dutta B, Pazhani GP, Choudhuri AR, Bhattacharya SK (2007) Occurrence, significance & molecular epidemiology of cholera outbreaks in West Bengal. Indian J Med Res 125:772–776
Tenover FC, Arbeit RD, Goering RV, Mickelsen PA, Murray BE, Persing DH, Swaminathan B (1995) Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol 33:2233–2239
Thungapathra M, Amita Sinha KK, Chaudhuri SR, Garg P, Ramamurthy T, Nair GB, Ghosh A (2002) Occurrence of antibiotic resistance gene cassettes aac(6’)-Ib, dfrA5, dfrA12 and ereA2 in Class I Integrons in Non-O1, Non-O139 vibrio cholerae strains in India. Antimicrob Agent Chemother 46:2948–2955
Xydas S, Lange CS, Phil D, Neimark HC (1996) Effect of methylation on the electrophoretic mobility of chromosomal DNA in pulsed-field agarose gels. Appl Theor Electrophor 6:43–47
Acknowledgments
We thank Dr A. Van Dorsselear (UMR-7178), Pr B. Ludes (EA-4438) for access to MALDI-TOF facilities, and Institut de Bactériologie for Vitek II facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eddabra, R., Moussaoui, W., Prévost, G. et al. Occurrence of Vibrio cholerae non-O1 in three wastewater treatment plants in Agadir (Morocco). World J Microbiol Biotechnol 27, 1099–1108 (2011). https://doi.org/10.1007/s11274-010-0556-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-010-0556-7