Abstract
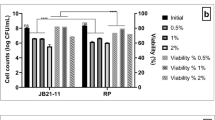
The study aimed at assessing the potential influences of two polyphenolic compounds on growth and some recently proven probiotic attributes of Streptococcus thermophilus CHCC 3534. The strain was tolerant to 0.8% gallic acid and 0.3% catechin. Gallic acid and catechin adapted cells were moderately to highly sensitive to most antibiotics, and to low pH (2.0) compared to the parental cells. Gallic acid adapted cells survived pHs 3.0 and 4.0, and were rapid β-galactosidase producers. Catechin adapted cells demonstrated marked tolerance to 0.4% oxgall, and possessed low hydrophobicity to organic solvents tested. They also produced large capsules of 5.0 μm in diameter due to high content of glucose and galactose monomers as determined by gas chromatography mass spectroscopy. Matrix-assisted laser-desorption time-of-flight mass spectrometry analysis detected small peptide subunits of masses 656 Da in bacteriocins of adapted cells that significantly inhibited the growth of Escherichia coli O157:H7, Pseudomonas aeroginosa, and Listeria monocytogenes. The data demonstrates that selection of S.thermophilus 3534 strain in dairy products and formulations supplemented with polyphenolics may present a nutritional benefit as a promising probiotic starter candidate as well as a potential source of dietary antioxidants.







Similar content being viewed by others
References
Ahn YJ, Lee C-O, Kwon J-H, Ahn J-W, Park J-H (1998) Growth-inhibitory effects of Galla Rhois-derived tannins on intestinal bacteria. J Appl Microbiol 84:439–443
Akyptis A, Kalantzopoulos G, Huis in’t Veld JH, Ten Brink B (1998) Purification and characerization of thermophilin T, a novel bacteriocin produced by Streptococcus thermophilus ACA-DC 0040. J Appl Microbiol 84:568–576
Alberto MR, Farías ME, María C, Manca de Nadra MC (2001) Effect of gallic acid and catechin on Lactobacillus hilgardii 5w on growth and metabolism of organic compounds. J Agric Food Chem 49:4359–4363
Alberto MR, Gόmez-Cordovés C, Manca de Nadra MC (2004) Metabolism of gallic acid and catechin by Lactobacillus hilgardii from wine. J Agric Food Chem 52:6465–6469
Alvarez-Jubete L, Wijngaard H, Arendt EK, Gallagher E (2010) Polyphenol composition and in vitro antioxidant activity of amaranth, quinoa buckwheat and wheat as affected by sprouting and baking. Food Chem 119:770–778
Awaisheh SS, Haddadin MSY, Robinson RK (2005) Incorporation of selected nutraceutical and probiotic bacteria into a fermented milk. Int Dairy J 15:1184–1190
Bently R, Sweely CC, Makita M, Wells WW (1963) Gas chromatography of sugars and other polyhydroxy compounds. Biochem Biophys Res Commun 1:14–18
Bodet C, Grenier D, Chandad F, Ofek I, Steinberg D, Weiss EI (2008) Potential oral health benefits of cranberry. Crit Rev Food Sci Nutr 48:672–680
Borneman WS, Akin DE, Van Eseltine WP (1986) Effect of phenolic monomers on ruminal bacteria. Appl Environ Microbiol 52:1331–1339
Campos FM, Couto JA, Hogg TA (2003) Influence of phenolic acids on growth and inactivation of Oenococcus oeni and Lactobacillus hilgardii. J Appl Microbiol 94:167–174
Campos FM, Couto JA, Figueiredo AR, Tóth IV, Rangel AO, Hogg TA (2009) Cell membrane damage induced by phenolic acids on wine lactic acid bacteria. Int J Food Microbiol 135:144–151
Clinical and Laboratory Standards Institute (CLSI) (2007) Performance standards for antimicrobial disk susceptibility tests. 27 (1)
Daeschel MA, McGuire J, Al-Makhkafi H (1992) Antimicrobial activities of nisin adsorbed onto hydrophilic and hydrophobic silicon surfaces. J Food Prot 55:731–735
De Ambrosini MV, Gonzalez S, De Ruiz Holgando AP, Oliver G (1998) Study of morphology of cell walls of some strains of lactic acid bacteria and related species. J Food Prot 61:557–562
Degl’Innocenti E, Guidi L, Pardossi A, Tognoni F (2005) Biochemical study of leaf browning in minimally processed leaves of lettuce (Lactuca sativa L. var. acephala). J Agric Food Chem 53:9980–9984
Dick AJ, Bearne SL (1988) Inhibition of β-galactosidase of apple by flavonoids and other polyphenols. J Food Biochem 12:97–108
Duda-Chodak A, Tarko T, Statek M (2008) The effects of antioxidants on Lactobacillus casei cultures. Acta Sci Pol Technol Aliment 7:39–51
Faber EJ, Zoon P, Kamerling JP, Vliegenthart JFG (1998) The exopolysaccharides produced by S. thermophilus Rs and Sts have the same repeating unit but differ in viscosity of their milk cultures. Carbohydr Res 310:269–276
Gilliland SE, Staley TE, Bush LJ (1984) Importance of bile tolerance of Lactobacillus acidophilus used as dietary adjunct. J Dairy Sci 67:3045–3051
Goel G, Puniya AK, Aguilar CN, Singh K (2005) Interaction of gut microflora with tannins in feeds. Naturwissenschaften 92:497–503
Guarner F, Perdigon G, Corthier G, Salminen S, Koletzko B, Morelli L (2005) Should yogurt cultures be considered probiotic? Br J Nutr 93:783–786
Guzmán-Lόpez O, Loera O, Parada JL, Castillo-Morales A, Martínez-Ramírez C, Augur C, Gaime-Perraud I, Saucedo-Castañeda G (2009) Microcultures of lactic acid bacteria: characterization and selection of strains, optimization of nutrients and gallic acid concentration. J Ind Microbiol Biotechnol 36:11–20
Hassan AN, Frank JF, Shalabi SI (2001) Factors affecting capsule size production by lactic acid bacteria used as dairy starter cultures. Int J Food Microbiol 64:199–203
Hata T, Tanaka R, Ohmomo S (2010) Isolation and characterization of plantaricin ASM1: a new bacteriocin produced by Lactobacillus plantarum. Int J Food Microbiol 137:94–99
Howell AB (2002) Cranberry proanthocyanidins and the maintenance of urinary tract health. Crit Rev Food Sci Nutr 42:273–278
Iyer R, Tomar SK, Kapila S, Mani J, Singh R (2010) Probiotic properties of folate producing Streptococcus thermophilus strains. Food Res Int 43:103–110
Jiménez-Díaz R, Rios-Sánchez RM, Desmazeaud M, Ruiz-Barba JL, Piard JC (1993) Plantaricins S and T, two new bacteriocins produced by Lactobacillus plantarum LPCO10 isolated from a green olive fermentation. Appl Environ Microbiol 59:1416–1424
Johnsen L, Fimland G, Nissen-Meyer J (2005) The C-terminal domain of pediocin-like antimicrobial peptides (class IIa bacteriocins) is involved in specific recognition of the C-terminal part of cognate immunity proteins and in determining the antimicrobial spectrum. J Biol Chem 280:9243–9250
Khalil RK (2004) Production of exopolysaccharides by lactic acid bacteria and possible applications in some dairy products. Dissertation, Alexandria University
Khalil RK (2009) Evidence for probiotic potential of a capsular-producing Streptococcus thermophilus CHCC 3534 strain. Pol J Microbiol 58:49–55
Knoll C, Divol B, du Toit M (2008) Influence of phenolic compounds on activity of nisin and pediocin PA-1. Am J Enol Vitic 59:418–421
Konuklar G, Gunasekaran S (2002) Rennet-induced milk coagulation by continuous steady shear stress. J Colloid Interface Sci 250:149–158
Koren E, Kohen R, Ovadia H, Ginsburg I (2009) Bacteria coated by polyphenols acquire potent oxidant-scavenging capacities. Exp Biol Med 234:940–951
Krause DO, Smith WJM, Brooker JD, McSweeney CS (2005) Tolerance mechanisms of streptococci to hydrolysable and condensed tannins. Anim Feed Sci Technol 121:59–75
Kristensen D, Jensen PY, Madsen F, Birdi KS (1997) Rheology and surface tension of selected processed dairy fluids: influence of temperature. J Dairy Sci 80:2282–2290
Landete JM, Rodríguez H, de las Rivas B, Muñoz R (2007) High-added-value antioxidants obtained from the degradation of wine phenolics by Lactobacillus plantarum. J Food Prot 70:2670–2675
Lorca G, Torino MI, Font de Valdez G, Ljungh AA (2002) Lactobacilli express cell surface proteins which mediate binding of immobilized collagen and fibronectin. FEMS Microbiol Lett 206:31–37
Maeta K, Nomura W, Takatsume Y, Izawa S, Inoue Y (2007) Green tea polyphenols function as prooxidants to activate oxidative-stress-responsive transcription factors in yeasts. Appl Environ Microbiol 73:572–580
Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L (2004) Polyphenols: food sources and bioavailability. Am J Clin Nutr 79:727–747
Mazauric JP, Salmon JM (2005) Interactions between yeast lees and wine polyphenols during simulation of wine aging: I. Analysis of remnant polyphenolic compounds in the resulting wines. J Agric Food Chem 53:5647–5653
Merkle RK, Poppe I (1994) Carbohydrate composition analysis of glycoconjugates by gas-liquid chromatography/mass spectroscopy. Methods Enzymol 230:1–15
Mertens-Talcott SU, Jilma-Stohlawetz P, Rios J, Hingorani L, Derendorf H (2006) Absorption, metabolism, and antioxidant effects of pomegranate (Punica granatum L.) polyphenols after ingestion of a standardized extract in healthy human volunteers. J Agric Food Chem 54:8956–8961
O’Donovan L, Brooker JD (2001) Effect of hydrolysable and condensed tannins on growth, morphology and metabolism of Streptococcus gallolyticus (S. caprinus) and Streptococcus bovis. Microbiol 147:1025–1033
Pandey KB, Rizvi SI (2009) Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Long 2:270–279
Park DJ, Oh S, Ku KH, Mok C, Hunkim S, Imm J (2005) Characteristics of yogurt-like products prepared from the combination of skim milk and soymilk containing saccharified-rice solution. Int J Food Sci Nutr 56:23–34
Patras A, Brunton NP, Da Pieve S, Butler F (2009) Impact of high pressure processing on total antioxidant activity, phenolic, ascorbic acid, anthocyanin content and colour of strawberry and blackberry purées. Inn Food Sci Emerg Technol 10:308–313
Peng Y, Serra M, Horne DS, Lucey JA (2009) Effect of fortification with various types of milk proteins on the rheological properties and permeability of nonfat set yogurt. J Food Sci 74:C666–C673
Puértolas E, Saldaña G, Condόn S, Άlverez I, Raso L (2009) A comparison of the effect of macerating enzymes and pulsed electric fields technology on phenolic content and color of red wine. J Food Sci 74:C647–C652
Puupponen-Pimiä R, Nohynek L, Alakomi H, Oksman-Caldentey K (2005) The action of berry phenolics against human intestinal pathogens. BioFactors 23:243–251
Ranadheera RDCS, Baines SK, Adams MC (2010) Importance of food in probiotic efficacy. Food Res Int 43:1–7
Reguant C, Bordons A, Arola L, Rozés N (2000) Influence of phenolic compounds on the physiology of Oenococcus oeni from wine. J Appl Microbiol 88:1065–1071
Rizkalla SW, Luo J, Kabir M, Chevalier A, Pacher N, Slama G (2000) Chronic consumption of fresh but not heated yogurt improves breath-hydrogen status and short-chain fatty acid profiles: a controlled study in healthy men with or without lactose maldigestion. Am J Clin Nutr 72:1474–1479
Rodríguez H, Curiel JA, Landete JM, de las Rivas B, de Felipe FL, Gómez-Cordovés C, Mancheño JM, Muñoz R (2009) Food phenolics and lactic acid bacteria. Int J Food Microbiol 132:79–90
Rozès N, Peres C (1998) Effects of phenolic compounds on the growth and the fatty acid composition of Lactobacillus plantarum. Appl Microbiol Biotechnol 49:108–111
Ruggeri S, Straniero R, Pacifico S, Aguzzi A, Virgili F (2008) French marine bark extract pycnogenol as a possible enrichment ingredient for yoghurt. J Dairy Sci 91:4484–4491
Salvucci E, Saavedra L, Sesma F (2007) Short peptides derived from the NH2-terminus of subclass IIa bacteriocin enterocin CRL35 show antimicrobial activity. J Antimicrob Chemoth 59:1102–1108
Scalbert A, Manach C, Morand C, Rémésy C, Jiménez L (2005) Dietary polyphenols and the prevention of diseases. Crit Rev Food Sci Nutr 45:287–306
Selma MV, Espin JC, Tomás-Barberán FA (2009) Interaction between phenolics and gut microbiota: role in human health. J Agric Food Chem 57:6485–6501
Shelburne CE, An FY, Dholpe V, Ramamoorthy A, Lopatin DE, Lantz MS (2007) The spectrum of antimicrobial activity of the bacteriocin subtilosin A. J Antimicrob Chemoth 59:297–300
Tiwari BK, Valdramidis VP, O’Donnel C, Muthukumarappan K, Bourke P, Cullen PJ (2009) Application of natural antimicrobials for food preservation. J Agric Food Chem 57:5987–6000
Ultee A, Kets EPW, Alberda M, Hoekstra FA, Smid EJ (2000) Adaptation of the food-borne pathogen Bacillus cereus to carvacrol. Arch Microbiol 174:233–238
Vinderola CG, Reinheimer JA (2003) Lactic acid starter and probiotic bacteria: a comparative “in vitro” study of probiotic characteristics and biological barrier resistance. Food Res Int 36:895–904
Zaidi-Yahiaoui R, Zaidi F, Bessai AA (2008) Influence of gallic and tannic acids on enzymatic activity and growth of Pectobacterium chrysanthemi (Dickeya chrysanthemi bv. chrysanthemi). Afr J Biotechnol 7:482–486
Acknowledgments
This work was conducted in the frame of two post-doctorate scholarships awarded to the author by the Binational Fulbright Commission in Egypt and the US.-Egypt Board on Scientific Cooperation in the period December 2008-April 2010. The author is grateful to Prof. Joseph Frank (Food Science and Technology Department, College of Agricultural and Environmental Sciences, University of Georgia, Athens GA, USA) for provision of bench space, materials and supplies, and for his critical reviewing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khalil, R.K.S. Influence of gallic acid and catechin polyphenols on probiotic properties of Streptococcus thermophilus CHCC 3534 strain. World J Microbiol Biotechnol 26, 2069–2079 (2010). https://doi.org/10.1007/s11274-010-0393-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-010-0393-8