Abstract
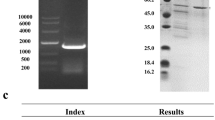
To produce aglycone isoflavones from soy flour, the β-glucosidase A gene (bglA) of Thermotoga maritima was overexpressed in Escherichia coli BL21-CodonPlus (DE3)-RIL. The K m and V max values of the purified BglA for pNPG were 0.43 mM and 323.6 U mg−1, respectively, and those for salicin were 9.0 mM and 183.2 U mg−1, respectively. The biochemical and kinetic characteristics of his-tagged BglA were found to be similar to those of BglA, except for the temperature stability and specific activity. Production of aglycone isoflavones from soy flour by BglA was examined by HPLC. For 3 h at 80°C, all the isoflavone glycosides approximated to the complete conversion into aglycone isoflavones, over seven times higher than that obtained from soy flour without BglA.




Similar content being viewed by others
References
Ausili A, Cobucci-Ponzano B, Di Lauro B, D’Avino R, Scirè A, Rossi M, Tanfani F, Moracci M (2006) Structural basis of the destabilization produced by an amino-terminal tag in the β-glycosidase from the hyperthermophilic archeon Sulfolobus solfataricus. Biochimie 88:807–817
Chuankhayan P, Hua Y, Svasti J, Sakdarat S, Sullivan PA, Ketudat-Cairns JR (2005) Purification of an isoflavonoid 7-O-β-apiosyl-glucoside β-glycosidase and its substrates from Dalbergia nigrescens Kurz. Phytochemistry 66:1880–1889
Claire V, Gregory JZ (2001) Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol Mol Biol Rev 65:1–43
Cotter A, Cashman KD (2003) Genistein appears to prevent early postmenopausal bone loss as effectively as hormone replacement therapy. Nutr Rev 61:346–351
Day AJ, DuPont MS, Ridley S, Rhodes M, Rhodes MJ, Morgan MR, Williamson G (1998) Deglycosylation of flavonoid and isoflavonoid glycosides by human small intestine and liver β-l-glucosidase activity. FEBS Lett 436:71–75
Gabelsberger J, Liebl W, Schleifer KH (1993) Purification and properties of recombinant β-glucosidase of the hyperthermophilic bacterium Thermotoga maritima. Appl Microbiol Biotechnol 40:44–52
Goyal K, Selvakumar P, Hayashi K (2001) Characterization of a thermostable β-glucosidase (BglB) from Thermotoga maritima showing transglycosylation activity. J Mol Cat B: Enzymatic 15:45–53
Huber R, Langworthy TA, Konig H, Thomm M, Woese CR, Sleytr UB, Stetter KO (1986) Thermotoga maritima sp. nov. represents a new genus of unique extremely thermophilic eubacteria growing up 90°C. Arch Microbiol 144:324–333
Izumi T, Piskula MK, Osawa S, Obata A, Tobe K, Saito M, Kataoka S, Kubota Y, Kikuchi M (2000) Soy isoflavone aglycones are absorbed faster and in higher amounts than their glucosides in humans. J Nutr 130:1695–1699
Kawakami Y, Tsurugasaki W, Nakamura S, Osada K (2005) Comparison of regulative functions between dietary soy isoflavones aglycone and glucoside on lipid metabolism in rats fed cholesterol. J Nutr Biochem 16:205–212
Kuo LC, Cheng WY, Wu RY, Huang CJ, Lee KT (2006) Hydrolysis of black soybean isoflavone glycosides by Bacillus subtilis natto. Appl Microbiol Biotechnol 73:314–320
Lamartiniere CA (2000) Protection against breast cancer with genistein: a component of soy. Am J Clin Nutr 71(6):1705–1707
Liu Y, Zhou SN, Chen ZS, Zhong YC, Liu YH (2004) Purification and characterization of an isoflavone-conjugates-hydrolyzing β-glucosidase from endophytic bacterium. J Agric Food Chem 52:1940–1944
Mathias K, Baraem I, Carlos MC, Kirby DH (2006) Heat and pH effects on the conjugated forms of genistin and daidzin isoflavones. J Agric Food Chem 54:7495–7502
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugars. Analyt Chem 31:426–428
Phimonphan C, Thipwarin R, Waraporn T, Narumol M, Prachumporn TK, Pornphimon M, Jisnuson S, Jensen ON, James RKC (2007a) Functional and structural differences between isoflavonoid β-glycosidases from Dalbergia sp. Arch Biochemistry Biophysics 468:205–216
Phimonphan C, Thipwarin R, Jisnuson S, James RKC (2007b) Hydrolysis of soybean isoflavonoid glycosides by Dalbergia β-glucosidases. J Agric Food Chem 55:2407–2412
Ravindranath MH, Muthugounder S, Presser N, Viswanathan S (2004) Anticancer therapeutic potential of soy isoflavone, genistein. Adv Exp Med Biol 546:121–1653
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Setchell KD, Brown NM, Zimmer-Nechemias L, Brashear WT, Wolfe BE, Kirschner AS, Heubi JE (2002) Evidence for lack of absorption of soy isoflavone glycosides in humans, supporting the crucial role of intestinal metabolism for bioavailability. Am J Clin Nutr 76:447–453
Suzuki H, Watanabe R, Fukushima Y, Fujita N, Noguchi A, Yokoyama R, Nishitani K, Nishino T, Nakayama T (2006) An isoflavone conjugate-hydrolyzing β-glucosidase from the roots of soybean (Glycine max) seedlings. J Biol Chem 281(40):30251–30259
Weaver CM, Cheong JM (2005) Soy isoflavones and bone health: the relationship is still unclear. J Nutr 135:1243–1247
Wolfgang L, Josef G, Karl-Heinz S (1994) Comparative amino acid sequence analysis of Thermotoga maritima beta-glucosidase (BglA) deduced from the nucleotide sequence of the gene indicates distant relationship between p-glucosidases of the BGA family and other families of beta-l, 4-glycosyl hydrolases. Mol Gen Genet 242:111–115
Xu X, Hariss KS, Wang HJ, Murphy PA, Hendrich S (1995) Bioavailability of soybean isoflavones depends upon gut microflora in women. J Nutr 125:2307–2315
Xue YM, Wu AL, Zeng HY, Shao WL (2006) High-level expression of an α-l-arabinofuranosidase from Thermotoga maritima in Escherichia coli for the production of xylobiose from xylan. Biotechnology Lett 28:351–356
Yang SQ, Jiang ZQ, Yan QJ, Zhu HF (2008) Characterization of a thermostable extracellular β-glucosidase with activities of exoglucanase and transglycosylation from Paecilomyces thermophila. J Agric Food Chem 56:602–608
Acknowledgments
This was work project BK2006220 supported by NSF of Jiangsu Province of China and project 05KJB180059 supported by NSF of higher school of Jiangsu Province of China, and was supported by a grant from The Nanjing Normal University (04104XGQ2B59).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xue, Y., Song, X. & Yu, J. Overexpression of β-glucosidase from Thermotoga maritima for the production of highly purified aglycone isoflavones from soy flour. World J Microbiol Biotechnol 25, 2165–2172 (2009). https://doi.org/10.1007/s11274-009-0121-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-009-0121-4