Abstract
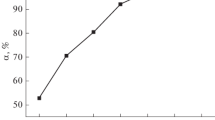
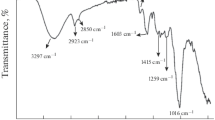
The purpose of this work is to investigate the efficiency of orange peel (OP) in the removal of a pharmaceutical product (Naproxen) from aqueous solutions. Adsorption isotherms were determined experimentally. The results showed that orange peel has an appreciable affinity for Naproxen, with an elimination capacity of 45 mg/g and that the temperature has a very significant effect on the adsorption process. The obtained data were then tested by three models (Langmuir, Freundlich and Temkin) and the Langmuir model provided a good fit with the experimental values. A thermodynamic study proved that the adsorption process is physical, spontaneous and endothermic. In the adsorption kinetics study, the influence of certain parameters, such as pH and the initial concentration of the naproxen solution, was investigated by three models (pseudo-first-order, pseudo-second-order and Elovich kinetics). The results show that the kinetics decrease with the initial naproxen concentration, and that the first-order model describes the adsorption kinetics well.








Similar content being viewed by others
Data Availability
The author declare that the data supporting the finding of this study are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
M. Ahmed, F. Mashkoor, and A. Nasar (2020). Development, characterization, and utilization of magnetized orange peel waste as a novel adsorbent for the confiscation of crystal violet dye from aqueous solution, Groundwater for Sustainable Development., vol. 10, no. May 2019, p. 100322, https://doi.org/10.1016/j.gsd.2019.100322.
Al-Maliky, E. A., Gzar, H. A., & Al-Azawy, M. G. (2021). Determination of Point of Zero Charge (PZC) of Concrete Particles Adsorbents. IOP Conference Series: Materials Science and Engineering, 1184(1), 012004. https://doi.org/10.1088/1757-899x/1184/1/012004
Ausavasukhi, A., Kampoosaen, C., & Kengnok, O. (2016). Adsorption characteristics of Congo red on carbonized leonardite. Journal of Cleaner Production, 134(Part B), 506–514. https://doi.org/10.1016/j.jclepro.2015.10.034
Deborah T. Bankole, A. P. Oluyori, and A. A. Inyinbor (2023). The removal of pharmaceutical pollutants from aqueous solution by Agro-waste, Arabian Journal of Chemistry, vol. Volume 16, no. Issue 5, p. 104699. https://doi.org/10.1016/j.arabjc.2023.104699.
Bieber, S., Snyder, S. A., Dagnino, S., Rauch-Williams, T., & Drewes, J. E. (2018). Management strategies for trace organic chemicals in water – A review of international approaches. Chemosphere, 195, 410–426. https://doi.org/10.1016/j.chemosphere.2017.12.100
Bukhari, A., et al. (2022). Removal of Eosin dye from simulated media onto lemon peel-based low cost biosorbent. Arabian Journal of Chemistry, 15(7), 103873. https://doi.org/10.1016/j.arabjc.2022.103873
Z. Ciğeroğlu, O. K. Özdemir, S. Şahin, and A. Haşimoğlu (2020). Naproxen Adsorption onto Graphene Oxide Nanopowders: Equilibrium, Kinetic, and Thermodynamic Studies, Water, Air, & Soil Pollution, vol. 231, no. 3, https://doi.org/10.1007/s11270-020-04472-7.
B. Czech, M. Kończak, M. Rakowska, and P. Oleszczuk (2021). Engineered biochars from organic wastes for the adsorption of diclofenac, naproxen and triclosan from water systems, Journal of Cleaner Production, vol. 288, https://doi.org/10.1016/j.jclepro.2020.125686.
Dai, Y., et al. (2018). Utilizations of agricultural waste as adsorbent for the removal of contaminants: A review. Chemosphere, 211, 235–253. https://doi.org/10.1016/j.chemosphere.2018.06.179
Darowna, D., Grondzewska, S., Morawski, A. W., & Mozia, S. (2014). Removal of non-steroidal anti-inflammatory drugs from primary and secondary effluents in a photocatalytic membrane reactor. Journal of Chemical Technology and Biotechnology, 89(8), 1265–1273. https://doi.org/10.1002/jctb.4386
De Gisi, S., Lofrano, G., Grassi, M., & Notarnicola, M. (2016). Characteristics and adsorption capacities of low-cost sorbents for wastewater treatment: A review. Sustainable Materials and Technologies, 9, 10–40. https://doi.org/10.1016/j.susmat.2016.06.002
Dey, S., Basha, S. R., Babu, G. V., & Nagendra, T. (2021). Characteristic and biosorption capacities of orange peels biosorbents for removal of ammonia and nitrate from contaminated water. Cleaner Materials, 1(June), 100001. https://doi.org/10.1016/j.clema.2021.100001
do Nascimento, G. E., Duarte, M. M. M. B., Campos, N. F., de Barbosa, C. M. B. M., & da Silva, V. L. (2014). Adsorption of the reactive gray BF-2R dye on orange peel Kinetics and equilibrium studies. Desalin Water Treat., 52(7), 1578–1588. https://doi.org/10.1080/19443994.2013.788457
Fernández, R. L., McDonald, J. A., Khan, S. J., & Le-Clech, P. (2014). Removal of pharmaceuticals and endocrine disrupting chemicals by a submerged membrane photocatalysis reactor (MPR). Separation and Purification Technology, 127, 131–139. https://doi.org/10.1016/j.seppur.2014.02.031
A. Gogoi, P. Mazumder, V. K. Tyagi, G. G. Tushara Chaminda, A. K. An, and M. Kumar (2018). Occurrence and fate of emerging contaminants in water environment: A review, Groundwater for Sustainable Development, vol. 6, pp. 169–180, https://doi.org/10.1016/j.gsd.2017.12.009.
Guiza, S. (2017). Biosorption of heavy metal from aqueous solution using cellulosic waste orange peel. Ecological Engineering, 99, 134–140. https://doi.org/10.1016/j.ecoleng.2016.11.043
A. Gunay Gurer, K. Aktas, M. Ozkaleli Akcetin, A. Erdem Unsar, and M. Asilturk (2021). Adsorption Isotherms, Thermodynamics, and Kinetic Modeling of Methylene Blue onto Novel Carbonaceous Adsorbent Derived from Bitter Orange Peels, Water, Air, & Soil Pollution, vol. 232, no. 4, https://doi.org/10.1007/s11270-021-05090-7.
Hammani, H., El Achaby, M., El Harfi, K., & El Mhammedi, M. A. (2020). Optimization and characterization of bio-oiland biochar production from date stone pyrolysis using Box-Behnken experimental design. Comptes Rendus Chim., 23, 589–606.
Hasan, Z., Choi, E. J., & Jhung, S. H. (2013). Adsorption of naproxen and clofibric acid over a metal–organic framework MIL-101 functionalized with acidic and basic groups. Chemical Engineering Journal, 219, 537–544. https://doi.org/10.1016/j.cej.2013.01.002
Hübner, U., Keller, S., & Jekel, M. (2013). Evaluation of the prediction of trace organic compound removal during ozonation of secondary effluents using tracer substances and second order rate kinetics. Water Research, 47(17), 6467–6474. https://doi.org/10.1016/j.watres.2013.08.025
Janyasuthiwong, S., Phiri, S. M., Kijjanapanich, P., Rene, E. R., Esposito, G., & Lens, P. N. L. (2015). Copper, lead and zinc removal from metal-contaminated wastewater by adsorption onto agricultural wastes. Environmental Technology (United Kingdom), 36(24), 3071–3083. https://doi.org/10.1080/09593330.2015.1053537
Jmai, S., Guiza, S., Jellali, S., Bagane, M., & Jeguirim, M. (2022). Competitive bio-sorption of basic dyes onto petiole palm tree wastes in single and binary systems. Comptes Rendus Chimie, 15, 1–12. https://doi.org/10.1016/j.crci.2015.07.016
Khazri, H., Ghorbel-Abid, I., Kalfat, R., & Trabelsi-Ayadi, M. (2017). Removal of ibuprofen, naproxen and carbamazepine in aqueous solution onto natural clay: Equilibrium, kinetics, and thermodynamic study. Applied Water Science, 7(6), 3031–3040. https://doi.org/10.1007/s13201-016-0414-3
Kim, E., et al. (2016). Sorptive removal of selected emerging contaminants using biochar in aqueous solution. Journal of Industrial and Engineering Chemistry, 36, 364–371. https://doi.org/10.1016/j.jiec.2016.03.004
Kyzas, G. Z., Fu, J., Lazaridis, N. K., Bikiaris, D. N., & Matis, K. A. (2015). New approaches on the removal of pharmaceuticals from wastewaters with adsorbent materials. Journal of Molecular Liquids, 209, 87–93. https://doi.org/10.1016/j.molliq.2015.05.025
López-Cázares, M. I., Isaacs-Páez, E. D., Ascacio-Valdés, J., Aguilar-González, C. N., Rangel-Mendez, J. R., & Chazaro-Ruiz, L. F. (2020). Electro-assisted naproxen adsorption followed by its electrodegradation and simultaneous electroreactivation of the activated carbon electrode. Separation and Purification Technology, 258(November), 2021. https://doi.org/10.1016/j.seppur.2020.118030
M. A. Mahmoud (2014). Adsorption of Cadmium onto Orange Peels: Isotherms, Kinetics, and Thermodynamics, Journal of Chromatography & Separation Techniques, vol. 05, no. 05, https://doi.org/10.4172/2157-7064.1000238.
Önal, Y., Akmil-Başar, C., & Sarici-Özdemir, Ç. (2007). Elucidation of the naproxen sodium adsorption onto activated carbon prepared from waste apricot: Kinetic, equilibrium and thermodynamic characterization. Journal of Hazardous Materials, 148(3), 727–734. https://doi.org/10.1016/j.jhazmat.2007.03.037
Pandiarajan, A., Kamaraj, R., Vasudevan, S., & Vasudevan, S. (2018). OPAC (orange peel activated carbon) derived from waste orange peel for the adsorption of chlorophenoxyacetic acid herbicides from water: Adsorption isotherm, kinetic modelling and thermodynamic studies. Bioresource Technology, 261(April), 329–341. https://doi.org/10.1016/j.biortech.2018.04.005
P. D. Pathak, S. A. Mandavgane, and B. D. Kulkarni (2017). Fruit peel waste: Characterization and its potential uses, Current Science, vol. 113, no. 3, pp. 444–454, https://doi.org/10.18520/cs/v113/i03/444-454.
Rafati, L., Hassan, M., Abbas, A., Mokhtari, M., & Hossein, A. (2016). Modeling of adsorption kinetic and equilibrium isotherms of naproxen onto functionalized nano-clay composite adsorbent. Journal of Molecular Liquids, 224, 832–841. https://doi.org/10.1016/j.molliq.2016.10.059
Reungoat, J., Macova, M., Escher, B. I., Carswell, S., Mueller, J. F., & Keller, J. (2010). Removal of micropollutants and reduction of biological activity in a full scale reclamation plant using ozonation and activated carbon filtration. Water Research, 44(2), 625–637. https://doi.org/10.1016/j.watres.2009.09.048
Saba, B., Christy, A. D., & Jabeen, M. (2016). Kinetic and Enzymatic Decolorization of Industrial Dyes Utilizing Plant-Based Biosorbents: A Review. Environmental Engineering Science, 33(9), 601–614. https://doi.org/10.1089/ees.2016.0038
Saruchi, & Kumar, V. (2019). Adsorption kinetics and isotherms for the removal of rhodamine B dye and Pb +2 ions from aqueous solutions by a hybrid ion-exchanger. Arabian Journal of Chemistry, 12(3), 316–329. https://doi.org/10.1016/j.arabjc.2016.11.009
Sirés, I., Brillas, E., Oturan, M. A., Rodrigo, M. A., & Panizza, M. (2014). Electrochemical advanced oxidation processes: Today and tomorrow. A review. Environmental Science and Pollution Research, 21(14), 8336–8367. https://doi.org/10.1007/s11356-014-2783-1
Urtiaga, A. M., Pérez, G., Ibáñez, R., & Ortiz, I. (2013). Removal of pharmaceuticals from a WWTP secondary effluent by ultrafiltration/reverse osmosis followed by electrochemical oxidation of the RO concentrate. Desalination, 331, 26–34. https://doi.org/10.1016/j.desal.2013.10.010
Yi, Z. J., Yao, J., Kuang, Y. F., Chen, H. L., Wang, F., & Yuan, Z. M. (2015). Removal of Pb(II) by adsorption onto Chinese walnut shell activated carbon. Water Science and Technology, 72(6), 983–989. https://doi.org/10.2166/wst.2015.305
Zhang, L., Wang, Y., Jin, S. W., Lu, Q. Z., & Ji, J. (2017). Adsorption isotherm, kinetic and mechanism of expanded graphite for sulfadiazine antibiotics removal from aqueous solutions. Environ. Technol. (united Kingdom), 38(20), 2629–2638. https://doi.org/10.1080/09593330.2016.1272637
W. Zhang et al. (2022). Sorbent Properties of Orange Peel-Based Biochar for Different Pollutants in Water, Processes, vol. 10, no. 5, https://doi.org/10.3390/pr10050856.
Zhou, Y., Zhang, L., & Cheng, Z. (2015). Removal of organic pollutants from aqueous solution using agricultural wastes: A review. Journal of Molecular Liquids, 212, 739–762. https://doi.org/10.1016/j.molliq.2015.10.023
Acknowledgements
The authors received support from the National Engineering School of Gabes / ENIG- University of Gabes, Tunisia. Special thanks to Mr Karim Hamdy (Culture Bridge International, Corvallis, Oregon USA) for many helpful style corrections.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jmai, S., Jmai, L., Guiza, S. et al. Removal of Naproxen from Aqueous Solutions using Eco-Friendly Bio-Adsorbent Prepared from Orange Peels. Water Air Soil Pollut 235, 321 (2024). https://doi.org/10.1007/s11270-024-07101-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-024-07101-9