Abstract
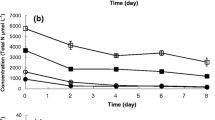
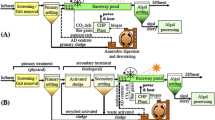
Huge discharge of different organic and inorganic waste compounds into water sources is the prime reason for water pollution. To protect the environment, appropriate biological treatment methods of wastewater with high removal efficacy are needed. To meet this end, indigenously available microbial consortiums were explored for their possible bioremediation efficiency. Cyanobacteria purified from microbial consortium was identified as Desertifilum sp. based on 16 s rRNA gene sequencing, and its biochemical characteristics were determined. High-rate algal pond (HRAP) of 6 m3 volume with dimensions of 3 m × 2 m × 1 m was inoculated @ 0.25% and operated in an open environment at a light intensity of 38,000 to 62,000 lx with a hydraulic retention time (HRT) of 12 days. Results obtained after 12 days showed removal efficiencies of 78.26, 76, 79.55, 4.77, and 58.74% for soluble chemical oxygen demand (sCOD), total chemical oxygen demand (tCOD), biochemical oxygen demand (BOD), nitrates, and total phosphorus, respectively. The results from the study inferred that Desertifilum sp. is a suitable candidate for secondary-stage wastewater treatment without any additional amendment. Moreover, the biochemical composition of the biomass obtained unraveled its potential application in the field of nutraceuticals.




Similar content being viewed by others
Data Availability
The datasets used and analyzed during the current study are available within the article and its supplementary information files or from the corresponding author upon reasonable request.
References
Akao, P. K., Singh, B., Kaur, P., Sor, A., Avni, A., Dhir, A., Verma, S., Kapoor, S., Phutela, U. G., Satpute, S., Sharma, S., Avisar, D., Sandha, K. S., & Mamane, H. (2021). Coupled microalgal–bacterial biofilm for enhanced wastewater treatment without energy investment. Journal of Water Process Engineering, 41, 1–10. https://doi.org/10.1016/j.jwpe.2021.102029
Alibas, I., Yilmaz, A., Alibas, K., Arslan, M., & Koc, C. (2023). The effect of different drying methods and microalgae species on the quality parameters of biodiesel obtained by transesterification technique. Biomass and Bioenergy, 168, 106688. https://doi.org/10.1016/j.biombioe.2022.106688
Al-Jalil, S. A. (2009). COD and BOD reduction of domestic wastewater using activated sludge, sand filters and activated carbon in Saudi Arabia. Biotechnology, 8, 473–477. https://doi.org/10.3923/biotech.2009.473.477
Altschul, S. F., Gish, W., Miller, W., Myers, E. W., & Lipman, D. J. (1990). Basic local alignment search tool. Journal of Molecular Biology, 215, 403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Alvarez, X., Alves, A., Ribeiro, M. P., Lazzari, M., Coutinho, P., & Otero, A. (2021). Biochemical characterization of Nostoc sp. exopolysaccharides and evaluation of potential use in wound healing. Carbohydrate Polymers, 254, 117303. https://doi.org/10.1016/j.carbpol.2020.117303
APHA. (2012). Standards methods for examinations of waste and wastewater (22nd ed.). American Public Health Association.
Ayers, R. S., & Westcot, D. W. (1985). Water quality for agriculture. FAO Irrigation and Drainage, Paper 29, Food and Agriculture Organization, Rome.
Baweja, S., Aggarwal, R., & Brar, M. (2017). Groundwater depletion in Punjab, India. Encyclopedia of Soil Science (3rd edn., pp. 1–5). https://doi.org/10.1081/E-ESS3-120052901
Bleakley, S., & Hayes, M. (2017). Algal proteins: Extraction, application, and challenges concerning production. Foods, 6, 1–34. https://doi.org/10.3390/foods6050033
Bolatkhan, K., Sadvakasova, A. K., Zayadan, B. K., Kakimova, A. B., Sarsekeyeva, F. K., Kossalbayev, B. D., Bozieva, A. M., Alwasel, S., & Allakhverdiev, S. I. (2020). Prospects for the creation of a waste-free technology for wastewater treatment and utilization of carbon dioxide based on cyanobacteria for biodiesel production. Journal of Biotechnology, 324, 162–170. https://doi.org/10.1016/j.jbiotec.2020.10.010
Cai, T., Park, S. Y., & Li, Y. (2013). Nutrient recovery from wastewater streams by microalgae: Status and prospects. Renewable and Sustainable Energy Reviews, 19, 360–369. https://doi.org/10.1016/j.rser.2012.11.030
Chiu, S. Y., Kao, C. Y., Chen, T. Y., Chang, Y. B., Kuo, C. M., & Lin, C. S. (2015). Cultivation of microalgal Chlorella for biomass and lipid production using wastewater as nutrient resource. Bioresource Technology, 184, 179–189. https://doi.org/10.1016/j.biortech.2014.11.080
Choi, H. J. (2014). Effect of optical panel distance in a photobioreactor for nutrient removal and cultivation of microalgae. World Journal of Microbiology and Biotechnology, 30, 2015–2023. https://doi.org/10.1007/s11274-014-1626-z
Choi, H.-J. (2016). Dairy wastewater treatment using microalgae for potential biodiesel application. Environmental Engineering Research, 21, 393–400. https://doi.org/10.4491/eer.2015.151
da Silva Braga, V., da Silveira Mastrantonio, D. J., Costa, J. A. V., & de Morais, M. G. (2018). Cultivation strategy to stimulate high carbohydrate content in Spirulina biomass. Bioresource Technology, 269, 221–226. https://doi.org/10.1016/j.biortech.2018.08.105
Daneshvar, E., Zarrinmehr, M. J., Koutra, E., Karnaros, M., Farhadian, O., & Bhatnagar, A. (2019). Sequential cultivation of microalgae in raw and recycled dairy wastewater: Microalgal growth, wastewater treatment and biochemical composition. Bioresource Technology, 273, 556–564. https://doi.org/10.1016/j.biortech.2018.11.059
Devi, M. P., Subhash, G. V., & Mohan, S. V. (2012). Heterotrophic cultivation of mixed microalgae for lipid accumulation and wastewater treatment during sequential growth and starvation phases: Effect of nutrient supplementation. Renewable Energy, 43, 276–283. https://doi.org/10.1016/j.renene.2011.11.021
Ding, J., Zhao, F., Cao, Y., Xing, L., Liu, W., Mei, S., & Li, S. (2015). Cultivation of microalgae in dairy farm wastewater without sterilization. International Journal of Phytoremediation, 17, 222–27. https://doi.org/10.1080/15226514.2013.876970
Divya, M., & Santhanam, P. (2019). Bioremediation of wastewater using a novel method of microalgae immobilized on twin-layer recirculation system (TLRS). In P. Santhanam, A. Begum, & P. Pachiappan (eds.), Basic and Applied Phytoplankton Biology. Springer, Singapore. https://doi.org/10.1007/978-981-10-7938-2_8
DuBois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28, 350–356. https://doi.org/10.1038/168167a0
El-Baky, H. H. A., El-Baz, F. K., & El-Baroty, G. S. (2008). Characterization of nutraceutical compounds in blue-green alga Spirulina maxima. Journal of Medicinal Plants Research, 2, 292–300.
Fito, J., & Alemu, K. (2019). Microalgae–bacteria consortium treatment technology for municipal wastewater management. Nanotechnology for Environmental Engineering, 4, 1–9. https://doi.org/10.1007/s41204-018-0050-2
Gonçalves, A. L., Pires, J. C., & Simões, M. (2017). A review on the use of microalgal consortia for wastewater treatment. Algal Research, 24, 403–415. https://doi.org/10.1016/j.algal.2016.11.008
Gonçalves, C. F., Menegol, T., & Rech, R. (2019). Biochemical composition of green microalgae Pseudoneochloris marina grown under different temperature and light conditions. Biocatalysis, 18, 1–10. https://doi.org/10.1016/j.bcab.2019.101032
Grubišić, M., Šantek, B., Zorić, Z., Čošić, Z., Vrana, I., Gašparović, B., Čož-Rakovac, R., & Šantek, M. I. (2022). Bioprospecting of microalgae isolated from the Adriatic Sea: Characterization of biomass, pigment, lipid and fatty acid composition, and antioxidant and antimicrobial activity. Molecules, 27, 1248. https://doi.org/10.3390/molecules27041248
Hadiyanto, C. M., & Soestrisnanto, D. (2013). Phytoremediation of palm oil mill effluent (POME) by using aquatic plants and microalgae for biomass production. Journal of Environmental Science and Technology, 6, 79–90. https://doi.org/10.5829/idosi.ije.2014.27.12c.02
Hamida, R. S., Abdelmeguid, N. E., Ali, M. A., Bin-Meferij, M. M., & Khalil, M. I. (2020). Synthesis of silver nanoparticles using a novel cyanobacteria Desertifilum sp. extract: Their antibacterial and cytotoxicity effects. International Journal of Nanomedicine, 15, 49–63. https://doi.org/10.2147/IJN.S238575
Harun, M. R., Singh, M., Forde, G., & Danquah, M. K. (2010). Bioprocess engineering of microalgae to produce a variety of consumer products. Renewable and Sustainable Energy Reviews, 14(3), 1037–1047. https://doi.org/10.1016/j.rser.2009.11.004
Hashem, S. M., El-Lahot, A., Helal, A. M., & Massoud, M. I. (2021). Evaluation of the phytochemicals and nutritional characteristics of some microalgae grown in Egypt as healthy food supplements. Egyptian Journal of Food Science, 49, 173–185. https://doi.org/10.21608/ejfs.2021.66232.1100
Hotos, G., Avramidou, D., Mastropetros, S. G., Tsigkou, K., Kouvara, K., Makridis, P., & Kornaros, M. (2023). Isolation, identification, and chemical composition analysis of nine microalgal and cyanobacterial species isolated in lagoons of Western Greece. Algal Research, 69, 102935. https://doi.org/10.1016/j.algal.2022.102935
Hu, B., Min, M., Zhou, W., Li, Y., Mohr, M., Cheng, Y., Lei, H., Liu, Y., Lin, X., Chen, P., & Ruan, R. (2012). Influence of exogenous CO2 on biomass and lipid accumulation of microalgae Auxenochlorella protothecoides cultivated in concentrated municipal wastewater. Applied Biochemistry and Biotechnology, 166, 1661–1673. https://doi.org/10.1007/s12010-012-9566-2
Huo, S., Kong, M., Zhu, F., Qian, J., Huang, D., Chen, P., & Ruan, R. (2020). Co-culture of Chlorella and wastewater-borne bacteria in vinegar production wastewater: Enhancement of nutrients removal and influence of algal biomass generation. Algal Research, 45, 101744. https://doi.org/10.1016/j.algal.2019.101744
Jerez-Martel, I., García-Poza, S., Rodríguez-Martel, G., Rico, M., Afonso-Olivares, C., & Gómez-Pinchetti, J. (2017). Phenolic profile and antioxidant activity of crude extracts from microalgae and cyanobacteria strains. Journal of Food Quality, 4, 1–8. https://doi.org/10.1155/2017/2924508
Khazi, M. I., Li, C., Liaqat, F., Malec, P., Li, J., & Fu, P. (2021). Acclimation and characterization of marine cyanobacterial strains Euryhalinema and Desertifilum for C-phycocyanin production. Frontiers in Bioengineering and Biotechnology, 9, 1–13. https://doi.org/10.3389/fbioe.2021.752024
Khemka, A., & Saraf, M. (2017). Strategic enhancement of Desertifilum tharense MSAK01 on dairy wastewater: An integrated approach for remediation and biomass production. 7, 2779–27785.Applied Water Science, 7:1–7. https://doi.org/10.1007/s13201-017-0525-5
Khemka, A., & Saraf, M. (2017a). Strategic enhancement of Desertifilum tharense MSAK01 on dairy wastewater: An integrated approach for remediation and biomass production. Applied Water Science, 7, 2779–2785. https://doi.org/10.1007/s13201-017-0525-5
Kotteswari, M., Murugesan, S., & Ranjith Kumar, R. (2012). Phycoremediation of dairy effluent by using the microalgae Nostoc sp. International Journal of Environmental Research and Development, 2, 35–43.
Kube, M., Jefferson, B., Fan, L., & Roddick, F. (2018). The impact of wastewater characteristics, algal species selection and immobilisation on simultaneous nitrogen and phosphorus removal. Algal Research, 31, 478–488. https://doi.org/10.1016/j.algal.2018.01.009
Liang, K., Zhang, Q., Gu, M., & Cong, W. (2013). Effect of phosphorus on lipid accumulation in freshwater microalgae Chlorella sp. Journal of Applied Phycology, 25, 311–318. https://doi.org/10.1007/s10811-012-9865-6
López-Rodríguez, A., Mayorga, J., Flaig, D., Fuentes, G., Cotabarren, J., Obregón, W. D., & Gómez, P. I. (2021). Comparison of two strains of the edible cyanobacteria Arthrospira: Biochemical characterization and antioxidant properties. Food Bioscience, 42, 101144. https://doi.org/10.1016/j.fbio.2021.101144
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with folin-phenol reagent. Journal of Biological Chemistry, 193, 265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Lv, J., Feng, J., Liu, Q., & Xie, S. (2017). Microalgal cultivation in secondary effluent: Recent developments and future work. International Journal of Molecular Sciences, 18, 79–94. https://doi.org/10.3390/ijms18010079
Mahapatra, D. M., Chanakya, H. N., & Ramachandra, T. V. (2013). Treatment efficacy of algae-based sewage treatment plants. Environmental Monitoring and Assessment, 185, 7145–7164. https://doi.org/10.1007/s10661-013-3090-x
Malaviya, P., & Rathore, V. S. (2001). A correlation study on some physicochemical quality parameters of pulp and paper mill effluents. Pollution Research, 20, 465–470.
Mata, T. M., Martins, A. A., & Caetano, N. S. (2010). Microalgae for biodiesel production and other applications. Renewable and Sustainable Energy Reviews, 14, 217–232. https://doi.org/10.1016/j.rser.2009.07.020
Maza-Márquez, P., González-Martínez, A., Martínez-Toledo, M. V., Fenice, M., Lasserrot, A., & González-López, J. (2017). Biotreatment of industrial olive washing water by synergetic association of microalgal-bacterial consortia in a photobioreactor. Environmental Science and Pollution Research, 24, 527–538. https://doi.org/10.1007/s11356-016-7753-3
Mishra, S. K., Suh, W. I., Farooq, W., Moon, M., Shrivastav, A., Park-Min, S., & Yang, J. W. (2014). Rapid quantification of microalgal lipids in aqueous medium by a simple colorimetric method. Bioresource Technology, 155, 330–333. https://doi.org/10.1016/j.biortech.2013.12.077
Moondra, N., Jariwala, N. D., & Christian, R. A. (2020). Microalgal-bacterial consortia: An alluring and novel approach for domestic wastewater treatment. Waste Conversion and Management, 4, 51–56. https://doi.org/10.26480/wcm.01.2020.51.56
Morales, M., Sanchez, L., & Revah, S. (2018). The impact of environmental factors on carbon dioxide fixation by microalgae. FEMS Microbiology Letters, 365, 1–11. https://doi.org/10.1093/femsle/fnx262
Moreira, A. S. P., Gonçalves, G., Conde, T. A., Couto, D., Melo, T., Maia, I. B., Pereira, H., Silva, J., Domingues, M. R., & Nunes, C. (2022). Chrysotila pseudoroscoffensis as a source of high-value polar lipids with antioxidant activity: A lipidomic approach. Algal Research, 66, 102756. https://doi.org/10.1016/j.algal.2022.102756
Nagappan, S., Bhosale, R., Duc Nguyen, D., Pugazhendhi, A., Tsai, P.-C., Chang, S. W., Ponnusamy, V. K., & Kumar, G. (2020). Nitrogen-fixing cyanobacteria as a potential resource for efficient biodiesel production. Fuel, 279, 118440. https://doi.org/10.1016/j.fuel.2020.118440
Nagarajan, D., Chen, C. Y., Ariyadasa, T. U., Lee, D. J., & Chang, J. S. (2022). Macroalgal biomass as a potential resource for lactic acid fermentation. Chemosphere, 309, 136694. https://doi.org/10.1016/j.chemosphere.2022.136694
Nübel, U., Garcia-Pichel, F., & Muyzer, G. (1997). PCR primers to amplify 16S rRNA genes from cyanobacteria. Applied and Environmental Microbiology, 63, 3327–3332. https://doi.org/10.1128/aem.63.8.3327-3332.1997
Olguín, E. J. (2003). Phycoremediation: Key issues for cost-effective nutrient removal processes. Biotechnological Advances, 22, 81–91. https://doi.org/10.1016/S0734-9750(03)00130-7
Patil, L., & Kaliwal, B. B. (2019). Microalga Scenedesmus bajacalifornicus BBKLP-07, a new source of bioactive compounds with in vitro pharmacological applications. Bioprocess and Biosystems Engineering. https://doi.org/10.1007/s00449-019-02099-5
Perendeci, N. A., Yılmaz, V., Tastan, B. E., Gökgöl, S., Fardinpoor, M., Namlı, A., & Steyer, J. P. (2019). Correlations between biochemical composition and biogas production during anaerobic digestion of microalgae and cyanobacteria isolated from different sources of Turkey. Bioresource Technology, 281, 209–216. https://doi.org/10.1016/j.biortech.2019.02.086
Prieto, P., Pineda, M., & Aguilar, M. (1999). Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Analytical Biochemistry, 269, 337–341. https://doi.org/10.1006/abio.1999.4019
Ramanan, R., Kim, B., Cho, D., Oh, H., & Kim, H. (2016). Algae bacteria interactions: Evolution, ecology and emerging applications. Biotechnology Advances, 34, 1–29. https://doi.org/10.1016/j.biotechadv.2015.12.003
Reitan, K. I., Øie, G., Jørgensen, H., & Wang, X. (2021). Chemical composition of selected marine microalgae, with emphasis on lipid and carbohydrate production for potential use as feed resources. Journal of Applied Phycology, 33, 3831–3842. https://doi.org/10.1007/s10811-021-02586-x
Rodolfi, L., Zittelli, G. C., Bassi, N., Padovani, G., Biondi, N., Bonini, G., & Tredici, M. R. (2009). Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in low-cost photobioreactor. Biotechnology and Bioengineering, 102, 100–112. https://doi.org/10.1002/bit.22033
Ryckebosch, E., Bruneel, C., Termote-Verhalle, R., Goiris, K., Muylaert, K., & Foubert, I. (2014). Nutritional evaluation of microalgae oils rich in omega-3 long chain polyunsaturated fatty acids as an alternative for fish oil. Food Chemistry, 160, 399–400. https://doi.org/10.1016/j.foodchem.2014.03.087
Sarfraz, R., Taneez, M., Sardar, S., Danish, L., & Hameed, A. (2021). Evaluation of Desmodesmus subspicatus for the treatment of wastewater. International Journal of Environmental and Analytical Chemistry, 103, 1–12. https://doi.org/10.1080/03067319.2021.1910681
Shrimali, M., & Singh, K. P. (2001). New methods of nitrate removal from water. Environmental Pollution, 112, 351–359. https://doi.org/10.1016/s0269-7491(00)00147-0
Sidhu, R. S., Vatta, K., & Dhaliwal, H. S. (2010). Conservation agriculture in Punjab: Economic implication of technologies and practices. Indian Journal of Agricultural Economics, 65, 413–427.
Singh, A. L. (2013). Nitrate and phosphate contamination in water and possible remedial measures. In Environmental Problems and Plant; Essay; Environmental Science, Department of Botany, Banaras Hindu University: Varanasi (pp. 44–51), India.
Singh, K. P., Singh, V. K., Malik, A., & Basant, N. (2006). Distribution of nitrogen species in groundwater aquifers of an industrial area in alluvial Indo-Gangetic plains: A case study. Environmental Geochemistry and Health, 28, 473–485. https://doi.org/10.1007/s10653-006-9053-1
Singh, P., & Kumar, D. (2022). Biomass and lipid production potential of cyanobacteria and microalgae isolated from the diverse habitats of Garhwal Himalaya, Uttarakhand. India. Biomass and Bioenergy, 162, 106469. https://doi.org/10.1016/j.biombioe.2022.106469
Song, B. H., Lee, D. H., Kim, B. C., Ku, S. H., Park, E. J., Kwon, I. H., Kim, K. H., & Kim, K. J. (2014). Photodynamic therapy using chlorophyll-a in the treatment of acne vulgaris: A randomized, single-blind, split face study. Journal of American Academy of Dermatology, 71, 764–771. https://doi.org/10.1016/j.jaad.2014.05.047
Su, Y., Mennerich, A., & Urban, B. (2011). Municipal wastewater treatment and biomass accumulation with a wastewater-born and settleable algal-bacterial culture. Water Research, 45, 3351–3358. https://doi.org/10.1016/j.watres.2011.03.046
Taga, M. S., Miller, E. E., & Pratt, D. E. (1984). Chia seeds as a source of natural lipid antioxidants. Journal of American Oil Chemists’ Society, 61, 928–931. https://doi.org/10.1007/BF02542169
Thomas, N. V., & Kim, S. K. (2011). Potential pharmacological applications of polyphenolic derivatives from marine brown algae. Environmental Toxicology and Pharmacology, 32, 325–335. https://doi.org/10.1016/j.etap.2011.09.004
Tyagi, T. (2017). Phytochemical screening of active metabolites present in Eichhornia crassipes (Mart.) Solms and Pistia Stratiotes (L.): Role in ethanomedicine. Asian Journal of Pharmaceutical Education and Research, 6, 40–56.
Ukwubile, C. A., Ahmed, A., Katsayal, U. A., Ya’u, J., & Mejida, S. (2019). GC–MS analysis of bioactive compounds from Melastomastrum capitatum (Vahl) Fern. leaf methanol extract: An anticancer plant. Scientific African, 3, 1–8. https://doi.org/10.1016/j.sciaf.2019.e00059
Vignaud, J., Loiseau, C., Herault, J., Mayer, C., Come, M., Martin, I., & Ulmann, I. (2023). Microalgae produce antioxidant molecules with potential preventive effects on mitochondrial functions and skeletal muscular oxidative stress. Antioxidants, 12, 1050. https://doi.org/10.3390/antiox12051050
Voltolina, D., Gómez-Villa, H., & Correa, G. (2005). Nitrogen removal and recycling by Scenedesmus obliquus in semicontinuous cultures using artificial wastewater and a simulated light and temperature cycle. Bioresource Technology, 96, 359–362. https://doi.org/10.1016/j.biortech.2004.04.004
Wilhelm, C., & Jakob, T. (2011). From photons to biomass and biofuels: Evaluation of different strategies for the improvement of algal biotechnology based on comparative energy balances. Applied Microbiology and Biotechnology, 92, 909–919. https://doi.org/10.1007/s00253-011-3627-2
Zhang, M., Gu, J., & Liu, Y. (2019). Engineering feasibility, economic viability and environmental sustainability of energy recovery from nitrous oxide in biological wastewater treatment plant. Bioresource Technology, 282, 514–519. https://doi.org/10.1016/j.biortech.2019.03.040
Zhu, L. D., Li, Z. H., & Hiltunen, E. (2016). Strategies for lipid production improvement in microalgae as a biodiesel feedstock. Biomed Research International, 2016, 1–8. https://doi.org/10.1155/2016/8792548
Funding
Funding was provided by the collaborative project of TIET-TAU Center of Excellence for Food Security (T2CEFS) under the Collaborative Project with PAU, “Enhanced Treatment of Wastewater using a Synergy of Microalgae and Microorganisms—Without Energy Investment and Biofuel Production”.
Author information
Authors and Affiliations
Contributions
BS: experimentation. AD: conceptualization and validation. NS: manuscript writing and data analysis. SGS, GSK, HM: review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, B., Sharma, N., Sharma, S.G. et al. Application of Microbial Consortium to the Pilot-Scale Treatment of Primary Treated Sewage Wastewater. Water Air Soil Pollut 235, 132 (2024). https://doi.org/10.1007/s11270-024-06953-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-024-06953-5