Abstract
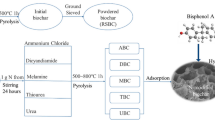
In this study, N-doped straw biochar(N-BC) was prepared using urea for the removal of Pb2+, Cu2+, and Cr6+ from water. SEM, FT-IR, XRD, and XPS characterization analyses demonstrated the successful loading of N onto the biochar. The adsorption performance of the N-doped biochar was evaluated by orthogonal and single-factor experiments. The results showed that the magnitude of the effect on the adsorption of the three heavy metals was consistent with initial pH > dosing > pyrolysis temperature. In addition, the optimum initial pH for the adsorption of Pb2+, Cu2+, and Cr6+ was 5, 6, and 2, respectively. Compared to the single biochar, the adsorption of Pb2+, Cu2+, and Cr6+ by N-doped biochar increased from 6.33, 4.25, and 3.78 mg/g to 30.2, 16.28, and 14.53 mg/g. The adsorption of Pb2+, Cu2+, and Cr6+ followed quasi-secondary kinetics and the Langmuir model. Mechanistic studies showed that the adsorption mechanism of N-doped biochar for Cu2+ and Pb2+ involved surface adsorption, cation-π bonding, and complexation, while that for Cr6+ included electrostatic attraction and reduction. The reusability and excellent adsorption properties of the surface N-doped biochar from cycling experiments indicate that biochar is a good material for the removal of heavy metals from water.











Similar content being viewed by others
Data Availability
All relevant data are included in the paper or its Supplementary data.
References
Alhooshani, K. (2020). Application of porous membrane bag enclosed alkaline treated Y-Zeolite for removal of heavy metal ions from water. Microchemical Journal, 152, 104289. https://doi.org/10.1016/j.microc.2019.104289
Almasian, A., Giahi, M., Chizari Fard, Gh., Dehdast, S. A., & Maleknia, L. (2018). Removal of heavy metal ions by modified PAN/PANI-nylon core-shell nanofibers membrane: filtration performance, antifouling and regeneration behavior. Chemical Engineering Journal, 351, 1166–1178. https://doi.org/10.1016/j.cej.2018.06.127
Awual, Md. R., Rahman, I. M. M., Yaita, T., Khaleque, Md. A., & Ferdows, M. (2014). pH dependent Cu(II) and Pd(II) ions detection and removal from aqueous media by an efficient mesoporous adsorbent. Chemical Engineering Journal, 236, 100–109. https://doi.org/10.1016/j.cej.2013.09.083
Bahadir, T., Şimşek, İ, Tulun, Ş, & Çelebi, H. (2023). Use of different food wastes as green biosorbent: isotherm, kinetic, and thermodynamic studies of Pb2+. Environmental Science and Pollution Research, 30(46), 103324–103338. https://doi.org/10.1007/s11356-023-29745-6
Çelebi, H. (2020). Recovery of detox tea wastes: usage as a lignocellulosic adsorbent in Cr6+ adsorption. Journal of Environmental Chemical Engineering, 8(5), 104310. https://doi.org/10.1016/j.jece.2020.104310
Cheng, S., Liu, Y., Xing, B., Qin, X., Zhang, C., & Xia, H. (2021). Lead and cadmium clean removal from wastewater by sustainable biochar derived from poplar saw dust. Journal of Cleaner Production, 314, 128074. https://doi.org/10.1016/j.jclepro.2021.128074
Chin, J. F., Heng, Z. W., Teoh, H. C., Chong, W. C., & Pang, Y. L. (2022). Recent development of magnetic biochar crosslinked chitosan on heavy metal removal from wastewater – modification, application and mechanism. Chemosphere, 291, 133035. https://doi.org/10.1016/j.chemosphere.2021.133035
Crini, G., Peindy, H. N., Gimbert, F., & Robert, C. (2007). Removal of C.I. Basic Green 4 (Malachite Green) from aqueous solutions by adsorption using cyclodextrin-based adsorbent: kinetic and equilibrium studies. Separation and Purification Technology, 53(1), 97–110. https://doi.org/10.1016/j.seppur.2006.06.018
Dinh, V. C., Hou, C.-H., & Dao, T. N. (2022). O, N-doped porous biochar by air oxidation for enhancing heavy metal removal: the role of O. N Functional Groups. Chemosphere, 293, 133622. https://doi.org/10.1016/j.chemosphere.2022.133622
Gao, L.-Y., Deng, J.-H., Huang, G.-F., Li, K., Cai, K.-Z., Liu, Y., & Huang, F. (2019). Relative distribution of Cd2+ adsorption mechanisms on biochars derived from rice straw and sewage sludge. Bioresource Technology, 272, 114–122. https://doi.org/10.1016/j.biortech.2018.09.138
Herath, A., Salehi, M., & Jansone-Popova, S. (2022). Production of polyacrylonitrile/ionic covalent organic framework hybrid nanofibers for effective removal of chromium(VI) from water. Journal of Hazardous Materials, 427, 128167. https://doi.org/10.1016/j.jhazmat.2021.128167
Hokkanen, S., Bhatnagar, A., Repo, E., Lou, S., & Sillanpää, M. (2016). Calcium hydroxyapatite microfibrillated cellulose composite as a potential adsorbent for the removal of Cr(VI) from aqueous solution. Chemical Engineering Journal, 283, 445–452. https://doi.org/10.1016/j.cej.2015.07.035
Kayranli, B., Gok, O., Yilmaz, T., Gok, G., Celebi, H., Seckin, I. Y., & Mesutoglu, O. C. (2022). Low-cost organic adsorbent usage for removing Ni2+ and Pb2+ from aqueous solution and adsorption mechanisms. International Journal of Environmental Science and Technology, 19(5), 3547–3564. https://doi.org/10.1007/s13762-021-03653-z
Lei, C., Wang, C., Chen, W., He, M., & Huang, B. (2020). Polyaniline@magnetic chitosan nanomaterials for highly efficient simultaneous adsorption and in-situ chemical reduction of hexavalent chromium: removal efficacy and mechanisms. Science of the Total Environment, 733, 139316. https://doi.org/10.1016/j.scitotenv.2020.139316
Liu, C., & Zhang, H.-X. (2022). Modified-biochar adsorbents (MBAs) for heavy-metal ions adsorption: a critical review. Journal of Environmental Chemical Engineering, 10(2), 107393. https://doi.org/10.1016/j.jece.2022.107393
Liu, D., Yang, L., Wu, J., & Li, B. (2022). Molten salt shielded preparation of rice straw biochars doped by copper sulfide for elemental mercury capture. Journal of the Energy Institute, 102, 176–183. https://doi.org/10.1016/j.joei.2022.03.005
Lv, D., Zhou, J., Cao, Z., Xu, J., Liu, Y., Li, Y., et al. (2019). Mechanism and influence factors of chromium(VI) removal by sulfide-modified nanoscale zerovalent iron. Chemosphere, 224, 306–315. https://doi.org/10.1016/j.chemosphere.2019.02.109
Ma, Q., Teng, W., Sun, Y., Chen, Y., Xue, Y., Chen, X., et al. (2022). Multi-component removal of Pb(II), Cd(II), and As(V) over core-shell structured nanoscale zero-valent iron@mesoporous hydrated silica. Science of the Total Environment, 827, 154329. https://doi.org/10.1016/j.scitotenv.2022.154329
Mahfoudhi, N., & Boufi, S. (2017). Nanocellulose as a novel nanostructured adsorbent for environmental remediation: a review. Cellulose, 24(3), 1171–1197. https://doi.org/10.1007/s10570-017-1194-0
Mandal, S., Calderon, J., Marpu, S. B., Omary, M. A., & Shi, S. Q. (2021). Mesoporous activated carbon as a green adsorbent for the removal of heavy metals and Congo red: characterization, adsorption kinetics, and isotherm studies. Journal of Contaminant Hydrology, 243, 103869. https://doi.org/10.1016/j.jconhyd.2021.103869
Meng, X., & Hu, R. (2021). Nitrogen/phosphorus enriched biochar with enhanced porosity activated by guanidine phosphate for efficient passivation of Pb(II), Cu(II) and Cd(II). Journal of Molecular Liquids, 323, 115071. https://doi.org/10.1016/j.molliq.2020.115071
Nizam, N. U. M., Hanafiah, M. M., Mahmoudi, E., Mohammad, A. W., & Oyekanmi, A. A. (2022). Effective adsorptive removal of dyes and heavy metal using graphene oxide based Pre-treated with NaOH / H2SO4 rubber seed shells synthetic graphite Precursor: Equilibrium Isotherm, kinetics and thermodynamic studies. Separation and Purification Technology, 289, 120730. https://doi.org/10.1016/j.seppur.2022.120730
Oh, W.-D., Lisak, G., Webster, R. D., Liang, Y.-N., Veksha, A., Giannis, A., et al. (2018). Insights into the thermolytic transformation of lignocellulosic biomass waste to redox-active carbocatalyst: durability of surface active sites. Applied Catalysis B: Environmental, 233, 120–129. https://doi.org/10.1016/j.apcatb.2018.03.106
Qi, W., Wang, Y., Ji, M., Zhao, Y., & Zhang, Z. (2015). Highly efficient adsorption of Cr(VI) by sakura leaves from aqueous solution. Chemistry Letters, 44(5), 697–699. https://doi.org/10.1246/cl.150035
Qiu, B., Tao, X., Wang, H., Li, W., Ding, X., & Chu, H. (2021). Biochar as a low-cost adsorbent for aqueous heavy metal removal: a review. Journal of Analytical and Applied Pyrolysis, 155, 105081. https://doi.org/10.1016/j.jaap.2021.105081
Wang, Q., Yu, J., Chen, X., Du, D., Wu, R., Qu, G., et al. (2019). Non-thermal plasma oxidation of Cu(II)-EDTA and simultaneous Cu(II) elimination by chemical precipitation. Journal of Environmental Management, 248, 109237. https://doi.org/10.1016/j.jenvman.2019.07.008
Wang, S., Xiao, D., Zheng, X., Zheng, L., Yang, Y., Zhang, H., et al. (2021). Halloysite and coconut shell biochar magnetic composites for the sorption of Pb(II) in wastewater: synthesis, characterization and mechanism investigation. Journal of Environmental Chemical Engineering, 9(6), 106865. https://doi.org/10.1016/j.jece.2021.106865
Yang, F., Jiang, Y., Dai, M., Hou, X., & Peng, C. (2022). Active biochar-supported iron oxides for Cr(VI) removal from groundwater: kinetics, stability and the key role of FeO in electron-transfer mechanism. Journal of Hazardous Materials, 424, 127542. https://doi.org/10.1016/j.jhazmat.2021.127542
Yu, W., Lian, F., Cui, G., & Liu, Z. (2018). N-doping effectively enhances the adsorption capacity of biochar for heavy metal ions from aqueous solution. Chemosphere, 193, 8–16. https://doi.org/10.1016/j.chemosphere.2017.10.134
Zahedifar, M., Seyedi, N., Shafiei, S., & Basij, M. (2021). Surface-modified magnetic biochar: highly efficient adsorbents for removal of Pb(ΙΙ) and Cd(ΙΙ). Materials Chemistry and Physics, 271, 124860. https://doi.org/10.1016/j.matchemphys.2021.124860
Zamora-Ledezma, C., Negrete-Bolagay, D., Figueroa, F., Zamora-Ledezma, E., Ni, M., Alexis, F., & Guerrero, V. H. (2021). Heavy metal water pollution: a fresh look about hazards, novel and conventional remediation methods. Environmental Technology & Innovation, 22, 101504. https://doi.org/10.1016/j.eti.2021.101504
Zhang, J., Chen, S., Zhang, H., & Wang, X. (2017). Removal behaviors and mechanisms of hexavalent chromium from aqueous solution by cephalosporin residue and derived chars. Bioresource Technology, 238, 484–491. https://doi.org/10.1016/j.biortech.2017.04.081
Zhang, H., Xiao, R., Li, R., Ali, A., Chen, A., & Zhang, Z. (2020). Enhanced aqueous Cr(VI) removal using chitosan-modified magnetic biochars derived from bamboo residues. Chemosphere, 261, 127694. https://doi.org/10.1016/j.chemosphere.2020.127694
Zhao, N., Zhao, C., Liu, K., Zhang, W., Tsang, D. C. W., Yang, Z., et al. (2021). Experimental and DFT investigation on N-functionalized biochars for enhanced removal of Cr(VI). Environmental Pollution, 291, 118244. https://doi.org/10.1016/j.envpol.2021.118244
Zhou, X., Liu, Y., Zhou, J., Guo, J., Ren, J., & Zhou, F. (2018). Efficient removal of lead from aqueous solution by urea-functionalized magnetic biochar: preparation, characterization and mechanism study. Journal of the Taiwan Institute of Chemical Engineers, 91, 457–467. https://doi.org/10.1016/j.jtice.2018.04.018
Zhou, Y., He, Y., Xiang, Y., Meng, S., Liu, X., Yu, J., et al. (2019). Single and simultaneous adsorption of pefloxacin and Cu(II) ions from aqueous solutions by oxidized multiwalled carbon nanotube. Science of the Total Environment, 646, 29–36. https://doi.org/10.1016/j.scitotenv.2018.07.267
Acknowledgements
This work was supported by the National Natural Science Foundation of China under grants (No.21477050), and the International Scientific and Technological Cooperation in Changzhou (CZ20140017). Besides, the author would like to thank the Analysis and Testing Center of Changzhou University[http://atc.cczu.edu.cn/main.htm] for their help in FT-IR, BET, and XRD testing and Shiyanjia Lab [www.shiyanjia.com] for related testing works on XPS.
Author information
Authors and Affiliations
Contributions
Hongli Guo: Conceptualization, Methodology, Formal analysis, Writing—original draft. Jinmei Xu: Visualization, Formal analysis. Qiuya Zhang: Visualization, Formal analysis. Sihai Zhou: Data curation, Visualization. Jiabao Song: Investigation, Visualization. Liping Wang: Resources, Writing – review & editing, Supervision, Project administration.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, H., Xu, J., Zhang, Q. et al. Study on the Performance of N-Doped Biochar for Removing Pb2+, Cu2+, and Cr6+from Water. Water Air Soil Pollut 235, 130 (2024). https://doi.org/10.1007/s11270-024-06947-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-024-06947-3