Abstract
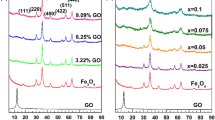
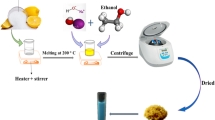
Highly active catalysts remain a vital issue for the Fenton-like degradation of polychlorinated phenols by H2O2. To this question, Cu0-Fe3O4 nanocatalysts functionalized with hydroxyl and carboxyl groups were prepared in this paper, and the Cu0-Fe3O4-H2O2 Fenton-like system was constructed and investigated for the degradation of chlorophenols. The results showed that Cu0 could greatly improve the oxidation activity of the system. 0.04 g Cu0-Fe3O4 composite (Fe/Cu = 3/1 mol/mol) could activate 20 μL H2O2 (30%) degrade near 100% DCP or 2,4,6-TCP or nitrophenol in 100 mL solution in the concentration of 50 mg/L within 1–5 min under initial pH 3.0–8.0, 25 °C. The Cu0-Fe3O4-H2O2 system showed high activity and broad pH applicability which was mainly due to the improved Fe(III)/Fe(II) cycle by Cu0, the reduced redox potential of Fe(III)/Fe(II) by functionalized group, and the adsorption ability of Cu0-Fe3O4 nanocomposites on chlorophenols. The degradation of 2,4-DCP was a combination of oxidative and reductive processes. About 86.6% was oxidized and 13.4% was reduced during the degradation. This study provides a potential method for quickly removing phenolic pollutants.






Similar content being viewed by others
Data availability
The datasets used and analyzed during the current study are available within the article and its supplementary information files or from the corresponding author upon reasonable request.
References
Anang, E., Liu, H., Fan, X., Zhao, D., & Gong, X. (2021). Compositional evolution of nanoscale zero valent iron and 2,4-dichlorophenol during dechlorination by attapulgite supported Fe/Ni nanoparticles. Journal of Hazardous Materials, 412, 125246. https://doi.org/10.1016/j.jhazmat.2021.125246
Bao, T., Damtie, M. M., Wu, K., Wei, X. L., Zhang, Y., Chen, J., Deng, C. X., Jin, J., Yu, Z. M., Wang, L., & Frost, R. L. (2019). Rectorite-supported nano-Fe3O4 composite materials as catalyst for P-chlorophenol degradation: Preparation, characterization, and mechanism. Applied Clay Science, 176, 66–77. https://doi.org/10.1016/j.clay.2019.04.020
Chen, L., Yuan, T., Ni, R., Yue, Q., & Gao, B. (2019). Multivariate optimization of ciprofloxacin removal by polyvinylpyrrolidone stabilized NZVI/Cu bimetallic particles. Chemical Engineering Journal, 365, 183–192. https://doi.org/10.1016/j.cej.2019.02.051
Daud, N. K., Akpan, U. G., & Hameed, B. H. (2012). Decolorization of Sunzol Black DN conc. in aqueous solution by Fenton oxidation process: Effect of system parameters and kinetic study. Desalination and Water Treatment, 37, 1–7. https://doi.org/10.1080/19443994.2012.661246
Deng, H., Li, X., Peng, Q., Wang, X., Chen, J., & Li, Y. (2005). Monodisperse magnetic single-crystal ferrite microspheres. Angewandte Chemie International Edition, 44, 2782–2785. https://doi.org/10.1002/anie.200462551
Deng, J., Yoon, S., Pasturel, M., Bae, S., & Hanna, K. (2022). Interactions between nanoscale zerovalent iron (NZVI) and silver nanoparticles alter the NZVI reactivity in aqueous environments. Chemical Engineering Journal, 450, 138406. https://doi.org/10.1016/j.cej.2022.138406
Ding, Y., Ruan, Y., Zhu, L., & Tang, H. (2017). Efficient oxidative degradation of chlorophenols by using magnetic surface carboxylated Cu0/Fe3O4 nanocomposites in a wide pH range. Journal of Environmental Chemical Engineering, 5, 2681–2690. https://doi.org/10.1016/j.jece.2017.05.021
Dong, G., Ai, Z., & Zhang, L. (2014). Total aerobic destruction of azo contaminants with nanoscale zero-valent copper at neutral pH: Promotion effect of in-situ generated carbon center radicals. Water Research, 66, 22–30. https://doi.org/10.1016/j.watres.2014.08.011
Feng, G., Cheng, P., Yan, W., Boronat, M., Li, X., Su, J.-H., Wang, J., Li, Y., Corma, A., Xu, R., & Yu, J. (2016). Accelerated crystallization of zeolites via hydroxyl free radicals. Science, 351, 1188–1191. https://doi.org/10.1126/science.aaf1559
Gao, Q., Chen, F., Zhang, J., Hong, G., Ni, J., Wei, X., & Wang, D. (2009). The study of novel Fe3O4@γ-Fe2O3 core/shell nanomaterials with improved properties. Journal of Magnetism and Magnetic Materials, 321, 1052–1057. https://doi.org/10.1016/j.jmmm.2008.10.022
Hu, H., Miao, K., Luo, X., Guo, S., Yuan, X., Pei, F., Qian, H., & Feng, G. (2021). Efficient Fenton-like treatment of high-concentration chlorophenol wastewater catalysed by Cu-Doped SBA-15 mesoporous silica. Journal of Cleaner Production, 318, 128632. https://doi.org/10.1016/j.jclepro.2021.128632
Hu, H., Wang, Z., Pan, L., Zhao, S., & Zhu, S. (2010). Ag-coated Fe3O4@SiO2 three-ply composite microspheres: Synthesis, characterization, and application in detecting melamine with their surface-enhanced Raman scattering. The Journal of Physical Chemistry C, 114, 7738–7742. https://doi.org/10.1021/jp100141c
Jiang, W., Cai, Q., Xu, W., Yang, M., Cai, Y., Dionysiou, D. D., & O’shea, K. E. (2014). Cr(VI) adsorption and reduction by humic acid coated on magnetite. Environmental Science & Technology, 48, 8078–8085. https://doi.org/10.1021/es405804m
Khan, A., Valicsek, Z., & Horváth, O. (2020). Synthesis, characterization and application of iron(II) doped copper ferrites (CuII(x)FeII(1-x)FeIII2O4) as novel heterogeneous photo-fenton catalysts. Nanomaterials, 10, 921.
Li, L., Guo, Q., Lv, B., Zheng, M., Zhan, W., Liu, Y., Xu, W., Wang, R., Zeng, H., & Mao, B. (2022). Surface modified silver/magnetite nanocomposite activating hydrogen peroxide for efficient degradation of chlorophenols. Journal of Colloid and Interface Science, 617, 246–256. https://doi.org/10.1016/j.jcis.2022.02.137
Li, L., Wang, R., Xing, X., Qu, W., Chen, S., & Zhang, Y. (2019). Preparation of porous semi-IPN temperature-sensitive hydrogel-supported nZVI and its application in the reduction of nitrophenol. Journal of Environmental Sciences, 82, 93–102. https://doi.org/10.1016/j.jes.2019.02.024
Lim, J.-W., Mohd Zaid, H. F., Isa, M. H., Oh, W.-D., Adnan, R., Bashir, M. J. K., Kiatkittipong, W., & Wang, D. K. (2018). Shielding immobilized biomass cryogel beads with powdered activated carbon for the simultaneous adsorption and biodegradation of 4-chlorophenol. Journal of Cleaner Production, 205, 828–835. https://doi.org/10.1016/j.jclepro.2018.09.153
Lin, J., Gulbagca, F., Aygun, A., Elhouda Tiri, R. N., Xia, C., Van Le, Q., Gur, T., Sen, F., & Vasseghian, Y. (2022). Phyto-mediated synthesis of nanoparticles and their applications on hydrogen generation on NaBH4, biological activities and photodegradation on azo dyes: Development of machine learning model. Food and Chemical Toxicology, 163, 112972. https://doi.org/10.1016/j.fct.2022.112972
Liu, S., Yu, B., Wang, S., Shen, Y., & Cong, H. (2020). Preparation, surface functionalization and application of Fe3O4 magnetic nanoparticles. Advances in Colloid and Interface Science, 281, 102165. https://doi.org/10.1016/j.cis.2020.102165
Liu, Y., Zhao, Y., & Wang, J. (2021). Fenton/Fenton-like processes with in-situ production of hydrogen peroxide/hydroxyl radical for degradation of emerging contaminants: Advances and prospects. Journal of Hazardous Materials, 404, 124191. https://doi.org/10.1016/j.jhazmat.2020.124191
Liu, Z., Yang, H., Wang, M., Sun, Y., Fei, Z., Chen, S., Luo, R., Hu, L., & Gu, C. (2022). Enhanced reductive debromination of decabromodiphenyl ether by organic-attapulgite supported Fe/Pd nanoparticles: Synergetic effect and mechanism. Journal of Colloid and Interface Science, 613, 337–348. https://doi.org/10.1016/j.jcis.2022.01.064
Luo, G., Jiao, Y., Lv, X., Zhang, X., & Gao, X. (2018). Green synthesis of dihydroxybenzene from phenol with hydrogen peroxide catalyzed by iron modified FSM-16. Research on Chemical Intermediates, 44, 5377–5387. https://doi.org/10.1007/s11164-018-3428-7
Machado, L. M. M., Lütke, S. F., Perondi, D., Godinho, M., Oliveira, M. L. S., Collazzo, G. C., & Dotto, G. L. (2020). Treatment of effluents containing 2-chlorophenol by adsorption onto chemically and physically activated biochars. Journal of Environmental Chemical Engineering, 8, 104473. https://doi.org/10.1016/j.jece.2020.104473
Pavithra, K. G., & Jaikumar, V. J. (2019). Removal of colorants from wastewater: A review on sources and treatment strategies. Journal of Industrial and Engineering Chemistry, 75, 1–19. https://doi.org/10.1016/j.jiec.2019.02.011
Satapanajaru, T., Anurakpongsatorn, P., Pengthamkeerati, P., & Boparai, H. (2008). Remediation of atrazine-contaminated soil and water by nano zerovalent iron. Water, Air, and Soil Pollution, 192, 349–359. https://doi.org/10.1007/s11270-008-9661-8
Seckin, H., Tiri, R. N. E., Meydan, I., Aygun, A., Gunduz, M. K., & Sen, F. (2022). An environmental approach for the photodegradation of toxic pollutants from wastewater using Pt–Pd nanoparticles: Antioxidant, antibacterial and lipid peroxidation inhibition applications. Environmental Research, 208, 112708. https://doi.org/10.1016/j.envres.2022.112708
Suflita, J. M., & Miller, G. D. (1985). Microbial metabolism of chlorophenolic compounds in ground water aquifers. Environmental Toxicology and Chemistry, 4, 751–758. https://doi.org/10.1002/etc.5620040606
Sun, C., Yang, S.-T., Gao, Z., Yang, S., Yilihamu, A., Ma, Q., Zhao, R.-S., & Xue, F. (2019). Fe3O4/TiO2/reduced graphene oxide composites as highly efficient Fenton-like catalyst for the decoloration of methylene blue. Materials Chemistry and Physics, 223, 751–757. https://doi.org/10.1016/j.matchemphys.2018.11.056
Sun, Y., Liu, Z., Fei, Z., Li, C., Chun, Y., & Zhang, A. (2019). Synergistic effect and degradation mechanism on Fe-Ni/CNTs for removal of 2,4-dichlorophenol in aqueous solution. Environmental Science and Pollution Research, 26, 8768–8778. https://doi.org/10.1007/s11356-019-04394-w
Tian, Z., Li, J., Zhu, G., Lu, J., Wang, Y., Shi, Z., & Xu, C. (2016). Facile synthesis of highly conductive sulfur-doped reduced graphene oxide sheets. Physical Chemistry Chemical Physics, 18, 1125–1130. https://doi.org/10.1039/C5CP05475C
Wang, J., Wang, X., Peng, Q., & Li, Y. (2004). Synthesis and characterization of bismuth single-crystalline nanowires and nanospheres. Inorganic Chemistry, 43, 7552–7556. https://doi.org/10.1021/ic049129q
Wang, M., Fang, G., Liu, P., Zhou, D., Ma, C., Zhang, D., & Zhan, J. (2016). Fe3O4@β-CD nanocomposite as heterogeneous Fenton-like catalyst for enhanced degradation of 4-chlorophenol (4-CP). Applied Catalysis B: Environmental, 188, 113–122. https://doi.org/10.1016/j.apcatb.2016.01.071
Wang, Y., & Xia, Y. (2004). Bottom-up and top-down approaches to the synthesis of monodispersed spherical colloids of low melting-point metals. Nano Letters, 4, 2047–2050. https://doi.org/10.1021/nl048689j
Wang, Z., Chen, G., Wang, X., Yang, G., Liu, Y., & Zhang, C. (2021). Performance of L-Cu&Mn-nZVFe@B nanomaterial on nitrate selective reduction under UV irradiation and persulfate activation in the presence of oxalic acid. Journal of Hazardous Materials, 401, 123378. https://doi.org/10.1016/j.jhazmat.2020.123378
Wang, Z., Zhai, S., Lv, J., Qi, H., Zheng, W., Zhai, B., & An, Q. (2015). Versatile hierarchical Cu/Fe3O4 nanocatalysts for efficient degradation of organic dyes prepared by a facile, controllable hydrothermal method. RSC Advances, 5, 74575–74584. https://doi.org/10.1039/C5RA16027H
Wilson, D., & Langell, M. A. (2014). XPS analysis of oleylamine/oleic acid capped Fe3O4 nanoparticles as a function of temperature. Applied Surface Science, 303, 6–13. https://doi.org/10.1016/j.apsusc.2014.02.006
Xing, L., Liu, H., Giesy, J. P., & Yu, H. (2012). pH-dependent aquatic criteria for 2,4-dichlorophenol, 2,4,6-trichlorophenol and pentachlorophenol. Science of the Total Environment, 441, 125–131. https://doi.org/10.1016/j.scitotenv.2012.09.060
Xu, J., Tang, J., Baig, S. A., Lv, X., & Xu, X. (2013). Enhanced dechlorination of 2,4-dichlorophenol by Pd/FeFe3O4 nanocomposites. Journal of Hazardous Materials, 244, 628–636. https://doi.org/10.1016/j.jhazmat.2012.10.051
Xu, L., & Wang, J. (2012). Magnetic nanoscaled Fe3O4/CeO2 composite as an efficient fenton-like heterogeneous catalyst for degradation of 4-chlorophenol. Environmental Science & Technology, 46, 10145–10153. https://doi.org/10.1021/es300303f
Xu, W., Hu, X., Lou, Y., Jiang, X., Shi, K., Tong, Y., Xu, X., Shen, C., Hu, B., & Lou, L. (2020). Effects of environmental factors on the removal of heavy metals by sulfide-modified nanoscale zerovalent iron. Environmental Research, 187, 109662. https://doi.org/10.1016/j.envres.2020.109662
Xue, X., Hanna, K., Despas, C., Wu, F., & Deng, N. (2009). Effect of chelating agent on the oxidation rate of PCP in the magnetite/H2O2 system at neutral pH. Journal of Molecular Catalysis A: Chemical, 311, 29–35. https://doi.org/10.1016/j.molcata.2009.06.016
Yang, Z., Gong, X.-B., Peng, L., Yang, D., & Liu, Y. (2018). Zn0-CNTs-Fe3O4 catalytic in situ generation of H2O2 for heterogeneous Fenton degradation of 4-chlorophenol. Chemosphere, 208, 665–673. https://doi.org/10.1016/j.chemosphere.2018.06.016
Yifeng, D., Chao, L., Yudan, S., Famei, W., Yang, L., Qiang, L., Tao, S., & Paul, K. C. (2020). Single-polarization photonic crystal fiber filter composed of elliptical gold films. Optical Engineering, 59, 076103. https://doi.org/10.1117/1.OE.59.7.076103
Yin, Y., Liu, W., Huo, N., & Yang, S. (2017). High rate capability and long cycle stability of Fe2O3/MgFe2O4 anode material synthesized by gel-cast processing. Chemical Engineering Journal, 307, 999–1007. https://doi.org/10.1016/j.cej.2016.09.025
You, J., Guo, Y., Guo, R., & Liu, X. (2019). A review of visible light-active photocatalysts for water disinfection: Features and prospects. Chemical Engineering Journal, 373, 624–641. https://doi.org/10.1016/j.cej.2019.05.071
Zheng, K., Song, Y., Wang, X., Li, X., Mao, X., & Wang, D. (2019). Understanding the electrode reaction process of dechlorination of 2,4-dichlorophenol over Ni/Fe nanoparticles: Effect of pH and 2,4-dichlorophenol concentration. Journal of Environmental Sciences, 84, 13–20. https://doi.org/10.1016/j.jes.2019.01.012
Zhou, R., Lu, S., Su, Y., Li, T., Ma, T., & Ren, H. (2020). Hierarchically fusiform CuO microstructures decorated with Fe3O4 nanoparticles as novel persulfate activators for 4-aminobenzenesulfonic acid degradation in aqueous solutions. Journal of Alloys and Compounds, 815, 152394. https://doi.org/10.1016/j.jallcom.2019.152394
Funding
This work was supported by the National Natural Science Foundation of China [Grant number 51508233] and the Jiangsu Collaborative Innovation Center of Technology and Material of Water Treatment, Suzhou University of Science and Technology.
Author information
Authors and Affiliations
Contributions
Lixia Li: the conception of the study; Jiajian Xiao: experiment, analysis, and manuscript preparation; Yuan Lu and Xinyu Yao: experiment; Wang Zhan: drawing of the graph; Qinkai Guo: data analysis; Songmei Wang: tests
Corresponding author
Ethics declarations
Ethics Approval
This work does not contain any studies with human participants or animals.
Consent to Participate
All authors provided informed consent to participate in this research study.
Consent for Publication
The authors transfer to the journal the publication rights and guarantee that this contribution is original. All the authors mutually agree to its submission.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 1765 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xiao, J., Lu, Y., Zhan, W. et al. Highly Efficient Functionalized Cu0-Fe3O4-H2O2 Fenton-Like System for the Degradation of Chlorophenols and Its Mechanism. Water Air Soil Pollut 234, 596 (2023). https://doi.org/10.1007/s11270-023-06601-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-023-06601-4