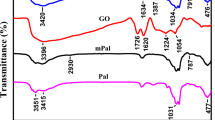
Abstract
Functionalized graphene oxide was an important candidate for removing heavy metal pollution. However, its difficult separation, low mechanical strength, and secondary pollution were restricted. In our study, polyvinyl alcohol/functionalized graphene oxide macroporous composite spheres (PVA/GO-SH) were prepared by wrapping sulfhydryl-functionalized graphene oxide (GO-SH) in polyvinyl alcohol (PVA) to solve the above problems. The experiments showed that the removal rates of Cu2+ and Pb2+ by 1 g L−1 PVA/GO-SH were 86.59% and 94.7%, and the equilibrium adsorption capacities were 84.42 mg g−1 and 218.62 mg g−1, respectively. The isothermal equation calculation showed that the adsorption was monomolecular layer adsorption. Kinetic and thermodynamic results showed that adsorption follows a second-order reaction kinetic model, and the more high temperature, the more favorable the adsorption. At a flow rate of 2.0 mL min−1, 80 mg L−1 of Cu2+ solution, and 100 mg L−1 of Pb2+ solution could be adsorbed entirely, with volumes of 1960 mL and 2480 mL, respectively. After five adsorption–desorption cycles, the adsorption rate achieved more than 75% of the initial adsorption rate. The composite sphere maintained an excellent spherical shape without damage, which had good stability. In conclusion, PVA/GO-SH has potential practical value.











Similar content being viewed by others
Data Availability
Data sharing is not applicable as no new data were generated or analyzed during this study.
References
Aklil, A., Mouflih, M., & Sebti, S. (2004). Removal of heavy metal ions from water by using calcined phosphate as a new adsorbent. Journal of Hazardous Materials, 112(3), 183–190. https://doi.org/10.1016/j.jhazmat.2004.05.018
Atia, A. A., Donia, A. M., & Elwakeel, K. Z. (2005). Adsorption behaviour of non-transition metal ions on a synthetic chelating resin bearing iminoacetate functions. Separation and Purification Technology, 43(1), 43–48. https://doi.org/10.1016/j.seppur.2004.09.012
Azad, H., Mohsennia, M., Cheng, C., & Amini, A. (2021). Facile fabrication of PVB-PVA blend polymer nanocomposite for simultaneous removal of heavy metal ions from aqueous solutions: Kinetic, equilibrium, reusability and adsorption mechanism. Journal of Environmental Chemical Engineering, 9(5), 106214. https://doi.org/10.1016/j.jece.2021.106214
Benettayeb, A., Morsli, A., Elwakeel, K. Z., Hamza, M. F., & Guibal, E. (2021). Recovery of heavy metal ions using magnetic glycine-modified chitosan—Application to aqueous solutions and tailing leachate. Applied Sciences, 11(18), 8377. https://doi.org/10.3390/app11188377
Bilal, M., Ihsanullah, I., Younas, M., & Ul Hassan Shah, M. (2021). Recent advances in applications of low-cost adsorbents for the removal of heavy metals from water: A critical review. Separation and Purification Technology, 278, 119510. https://doi.org/10.1016/j.seppur.2021.119510
Chen, D., Zhang, H., Yang, K., & Wang, H. (2016). Functionalization of 4-aminothiophenol and 3-aminopropyltriethoxysilane with graphene oxide for potential dye and copper removal. Journal of Hazardous Materials, 310, 179–187. https://doi.org/10.1016/j.jhazmat.2016.02.040
Donga, C., Mishra, S. B., Abd-El-Aziz, A. S., Ndlovu, L. N., Mishra, A. K., & Kuvarega, A. T. (2022). (3-Aminopropyl) Triethoxysilane (APTES) functionalized magnetic nanosilica graphene oxide (MGO) nanocomposite for the comparative adsorption of the heavy metal [Pb(II), Cd(II) and Ni(II)] ions from aqueous solution. Journal of Inorganic and Organometallic Polymers and Materials, 32(6), 2235–2248. https://doi.org/10.1007/s10904-022-02287-z
Elgarahy, A. M., Elwakeel, K. Z., Mohammad, S. H., & Elshoubaky, G. A. (2021). A critical review of biosorption of dyes, heavy metals and metalloids from wastewater as an efficient and green process. Cleaner Engineering and Technology, 4, 100209. https://doi.org/10.1016/j.clet.2021.100209
Elgarahy, A. M., Elwakeel, K. Z., Akhdhar, A., & Hamza, M. F. (2021a). Recent advances in greenly synthesized nanoengineered materials for water/wastewater remediation: an overview. Nanotechnology for Environmental Engineering, 6(1), 1–24. https://doi.org/10.1007/s41204-021-00104-5
El-Korashy, S. A., Elwakeel, K. Z., & El-Hafeiz, A. A. (2016). Fabrication of bentonite/thiourea-formaldehyde composite material for Pb(II), Mn(VII) and Cr(VI) sorption: A combined basic study and industrial application. Journal of Cleaner Production, 137, 40–50. https://doi.org/10.1016/j.jclepro.2016.07.073
Elwakeel, K. Z. (2010). Environmental application of chitosan resins for the treatment of water and wastewater: A review. Journal of Dispersion Science and Technology, 31(3), 273–288. https://doi.org/10.1080/01932690903167178
Elwakeel, K. Z., El-Bindary, A. A., & Kouta, E. Y. (2017). Retention of copper, cadmium and lead from water by Na-Y-Zeolite confined in methyl methacrylate shell. Journal of Environmental Chemical Engineering, 5(4), 3698–3710. https://doi.org/10.1016/j.jece.2017.06.049
Elwakeel, K. Z., Aly, M. H., El-Howety, M. A., El-Fadaly, E., & Al-Said, A. (2018). Synthesis of Chitosan@activated carbon beads with abundant amino groups for capture of Cu(II) and Cd(II) from aqueous solutions. Journal of Polymers and the Environment, 26(9), 3590–3602. https://doi.org/10.1007/s10924-018-1243-2
Elwakeel, K. Z., El-Bindary, A. A., Kouta, E. Y., & Guibal, E. (2018). Functionalization of polyacrylonitrile/Na-Y-zeolite composite with amidoxime groups for the sorption of Cu(II), Cd(II) and Pb(II) metal ions. Chemical Engineering Journal, 332, 727–736. https://doi.org/10.1016/j.cej.2017.09.091
Elwakeel, K. Z., Elgarahy, A. M., Khan, Z. A., Almughamisi, M. S., & Al-Bogami, A. S. (2020). Perspectives regarding metal/mineral-incorporating materials for water purification: With special focus on Cr(vi) removal. Materials Advances, 1(6), 1546–1574. https://doi.org/10.1039/D0MA00153H
Guo, C., Wang, Y., Wang, F., & Wang, Y. (2021). Adsorption performance of amino functionalized magnetic molecular sieve adsorbent for effective removal of lead ion from aqueous solution. Nanomaterials, 11(9), 2353. https://doi.org/10.3390/nano11092353
He, K., Chen, Y., Tang, Z., & Hu, Y. (2016). Removal of heavy metal ions from aqueous solution by zeolite synthesized from fly ash. Environmental Science and Pollution Research, 23(3), 2778–2788. https://doi.org/10.1007/s11356-015-5422-6
Hui, B., Zhang, Y., & Ye, L. (2015). Structure of PVA/gelatin hydrogel beads and adsorption mechanism for advanced Pb(II) removal. Journal of Industrial and Engineering Chemistry, 21, 868–876. https://doi.org/10.1016/j.jiec.2014.04.025
Idris, A., Zain, N. A. M., & Suhaimi, M. S. (2008). Immobilization of Baker’s yeast invertase in PVA–Alginate matrix using innovative immobilization technique. Process Biochemistry, 43(4), 331–338. https://doi.org/10.1016/j.procbio.2007.12.008
Ji, W., Feng, N., Zhao, P., Zhang, S., Zhang, S., Lan, L., Huang, H., Li, K., Sun, Y., Li, Y., & Ma, Y. (2020). Synthesis of single-phase zeolite a by coal gasification fine slag from Ningdong and its application as a high-efficiency adsorbent for Cu2+ and Pb2+ in Simulated Waste Water. ChemEngineering, 4(4), 65. https://doi.org/10.3390/chemengineering4040065
Jia, Y., Zhang, Y., Wang, R., Fan, F., & Xu, Q. (2012). Studies on adsorptions of metallic ions in water by zirconium glyphosate (ZrGP): Behaviors and mechanisms. Applied Surface Science, 258(7), 2551–2561. https://doi.org/10.1016/j.apsusc.2011.10.091
Jokar, M., Mirghaffari, N., Soleimani, M., & Jabbari, M. (2019). Preparation and characterization of novel bio ion exchanger from medicinal herb waste (chicory) for the removal of Pb2+ and Cd2+ from aqueous solutions. Journal of Water Process Engineering, 28, 88–99. https://doi.org/10.1016/j.jwpe.2019.01.007
Kelly-Vargas, K., Cerro-Lopez, M., Reyna-Tellez, S., Bandala, E. R., & Sanchez-Salas, J. L. (2012). Biosorption of heavy metals in polluted water, using different waste fruit cortex. Physics and Chemistry of the Earth, Parts A/B/C, 37–39, 26–29. https://doi.org/10.1016/j.pce.2011.03.006
Kim, N., Park, M., & Park, D. (2015). A new efficient forest biowaste as biosorbent for removal of cationic heavy metals. Bioresource Technology, 175, 629–632. https://doi.org/10.1016/j.biortech.2014.10.092
Li, X., Wang, Z., Li, Q., Ma, J., & Zhu, M. (2015). Preparation, characterization, and application of mesoporous silica-grafted graphene oxide for highly selective lead adsorption. Chemical Engineering Journal, 273, 630–637. https://doi.org/10.1016/j.cej.2015.03.104
Li, Y., Peng, L., & Li, W. (2020). Adsorption behaviors on trace Pb2+ from water of biochar adsorbents from konjac starch. Adsorption Science & Technology, 38(9–10), 344–356. https://doi.org/10.1177/0263617420948699
Lima, E. C., Hosseini-Bandegharaei, A., Moreno-Piraján, J. C., & Anastopoulos, I. (2019). A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t Hoof equation for calculation of thermodynamic parameters of adsorption. Journal of Molecular Liquids, 273, 425–434. https://doi.org/10.1016/j.molliq.2018.10.048
Liu, Z., Kiessling, F., & Gätjens, J. (2010). Advanced nanomaterials in multimodal imaging: Design, functionalization, and biomedical applications. Journal of Nanomaterials, 2010, 1–15. https://doi.org/10.1155/2010/894303
Mi, X., Huang, G., Xie, W., Wang, W., Liu, Y., & Gao, J. (2012). Preparation of graphene oxide aerogel and its adsorption for Cu2+ ions. Carbon, 50(13), 4856–4864. https://doi.org/10.1016/j.carbon.2012.06.013
Mishra, P. C., & Patel, R. K. (2009). Removal of lead and zinc ions from water by low cost adsorbents. Journal of Hazardous Materials, 168(1), 319–325. https://doi.org/10.1016/j.jhazmat.2009.02.026
Renu, N. A., Agarwal, M., & Singh, K. (2017). Methodologies for removal of heavy metal ions from wastewater: An overview. Interdisciplinary Environmental Review, 18(2), 124. https://doi.org/10.1504/IER.2017.087915
Singh, S., Patel, P., Shahi, V. K., & Chudasama, U. (2011). Pb2+ selective and highly cross-linked zirconium phosphonate membrane by sol–gel in aqueous media for electrochemical applications. Desalination, 276(1–3), 175–183. https://doi.org/10.1016/j.desal.2011.03.048
Sönmezay, A., Öncel, M. S., & Bektaş, N. (2012). Adsorption of lead and cadmium ions from aqueous solutions using manganoxide minerals. Transactions of Nonferrous Metals Society of China, 22(12), 3131–3139. https://doi.org/10.1016/S1003-6326(12)61765-8
Tantis, I., Psarras, G. C., & Tasis, D. (2012). Functionalized graphene-poly(vinyl alcohol) nanocomposites: Physical and dielectric properties. Express Polymer Letters, 6(4), 283–292. https://doi.org/10.3144/expresspolymlett.2012.31
Wang, S., Zhu, Z. H., Coomes, A., Haghseresht, F., & Lu, G. Q. (2005). The physical and surface chemical characteristics of activated carbons and the adsorption of methylene blue from wastewater. Journal of Colloid and Interface Science, 284(2), 440–446. https://doi.org/10.1016/j.jcis.2004.10.050
Wang, H., Gao, B., Wang, S., Fang, J., Xue, Y., & Yang, K. (2015). Removal of Pb(II), Cu(II), and Cd(II) from aqueous solutions by biochar derived from KMnO4 treated hickory wood. Bioresource Technology, 197, 356–362. https://doi.org/10.1016/j.biortech.2015.08.132
Wang, S., Gao, B., Li, Y., Mosa, A., Zimmerman, A. R., Ma, L. Q., Harris, W. G., & Migliaccio, K. W. (2015). Manganese oxide-modified biochars: Preparation, characterization, and sorption of arsenate and lead. Bioresource Technology, 181, 13–17. https://doi.org/10.1016/j.biortech.2015.01.044
Wang, Y., Su, Y., Fang, W., Zhang, Y., Li, X., Zhang, G., & Sun, W. (2020). SnO2/SnS2 nanocomposite anchored on nitrogen-doped RGO for improved photocatalytic reduction of aqueous Cr(VI). Powder Technology, 363, 337–348. https://doi.org/10.1016/j.powtec.2020.01.009
Yakout, A. A., Shaker, M. A., Elwakeel, K. Z., & Alshitari, W. (2019). Lauryl sulfate@magnetic graphene oxide nanosorbent for fast methylene blue recovery from aqueous solutions. Journal of Dispersion Science and Technology, 40(5), 707–715. https://doi.org/10.1080/01932691.2018.1477604
Yao, L., Zhang, Y. C., Li, J., & Chen, Y. (2014). Photocatalytic properties of SnS2/SnO2 nanocomposite prepared by thermal oxidation of SnS2 nanoparticles in air. Separation and Purification Technology, 122, 1–5. https://doi.org/10.1016/j.seppur.2013.10.038
Yari, M., Rajabi, M., Moradi, O., Yari, A., Asif, M., Agarwal, S., & Gupta, V. K. (2015). Kinetics of the adsorption of Pb(II) ions from aqueous solutions by graphene oxide and thiol functionalized graphene oxide. Journal of Molecular Liquids, 209, 50–57. https://doi.org/10.1016/j.molliq.2015.05.022
Zhang, H., Chang, Q., Jiang, Y., Li, H., & Yang, Y. (2018). Synthesis of KMnO4-treated magnetic graphene oxide nanocomposite (Fe3O4@GO/MnOx) and its application for removing of Cu2+ ions from aqueous solution. Nanotechnology, 29(13), 135706. https://doi.org/10.1088/1361-6528/aaaa2f
Zhang, F., Shen, L., Li, J., Zhang, Y., Wang, G., & Zhu, A. (2021). Room temperature photocatalytic deposition of Au nanoparticles on SnS2 nanoplates for enhanced photocatalysis. Powder Technology, 383, 371–380. https://doi.org/10.1016/j.powtec.2021.01.065
Zhang, F., Zhang, Y., Wang, Y., Zhu, A., & Zhang, Y. (2022). Efficient photocatalytic reduction of aqueous Cr (VI) by Zr4+ doped and polyaniline coupled SnS2 nanoflakes. Separation and Purification Technology, 283, 120161. https://doi.org/10.1016/j.seppur.2021.120161
Zhang, H., Shi, Z., Xiao, Y., Zhang, X., Yin, X., & Tian, L. (2022). Preparation of 3D graphene by 3D printing and its application in water treatment. Chemical Industry and Engineering Progress, 41(5), 2231–2242. https://doi.org/10.16085/j.issn.1000-6613.2021-1277
Zu, Y., Zhang, Y., Zhao, X., Shan, C., Zu, S., Wang, K., Li, Y., & Ge, Y. (2012). Preparation and characterization of chitosan–polyvinyl alcohol blend hydrogels for the controlled release of nano-insulin. International Journal of Biological Macromolecules, 50(1), 82–87. https://doi.org/10.1016/j.ijbiomac.2011.10.006
Acknowledgements
We also thank the “Hongliu Excellent Young” talents support program of Lanzhou University of Technology.
Funding
This work was financially supported by the China Postdoctoral Science Foundation (2019M653796), Natural Science Foundation of Gansu Province (20JR10RA197; 22JR5RA254) and Science and Technology Innovation Fund of Gansu Academy of Sciences (2019QN-08).
Author information
Authors and Affiliations
Contributions
Huining Zhang proposed the general idea and methodology of the project.
Xingmao Liu wrote the original manuscript.
Lihong Tian, Yuling Tang, Zhongyu Shi, and Yankui Xiao performed the synthesis and adsorption experiments as well as analyzed data.
Zhiguo Wu, Ying Zhu, Qi Guo, and Zhangpu Peng supplemented the experiments, analyzed the data, reviewed, and wrote the relevant parts for the revised manuscript.
All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, H., Liu, X., Tian, L. et al. Preparation of Functionalized Graphene Oxide Composite Spheres and Removal of Cu2+ and Pb2+ from Wastewater. Water Air Soil Pollut 233, 512 (2022). https://doi.org/10.1007/s11270-022-05971-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05971-5