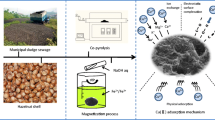
Abstract
In this study, an efficient nano-zero-valent iron supported by alkali-modified sludge biochar (nZVI@NaOH-SBC) composites were synthesized by a liquid-phase reduction method for Cu2+ removal. The preparation conditions (sodium hydroxide concentration, pyrolysis temperature, solid–liquid ratio, and modification time) of sludge biochar (SBC) were optimized by orthogonal experiments. The results showed that the best SBC was prepared under the conditions of sodium hydroxide concentration of 3 mol/L, pyrolysis temperature of 500 °C, a solid–liquid ratio of 1:20, and modification time of 24 h, and a maximum adsorption capacity was 183.22 mg/g, which is 2.43 times that of the pristine sludge biochar. The adsorption behavior was in accordance with a pseudo-second-order kinetic model, indicating that the removal of Cu2+ was mainly affected by chemisorption. The adsorption isotherm conformed to the Langmuir model, and the maximum adsorption capacity of Cu2+ was 358.42 mg/g at pH = 5, and 25 °C. Reusability tests confirmed the good reusability and stability of the material. Batch experiments and characterization reveal that the mechanism of Cu removal by nZVI@NaOH-SBC composites involves multiple processes: adsorption, precipitation, electrostatic attraction, and reduction. This study proposes a new biochemical adsorbent, which provides a good choice for the treatment of copper-containing wastewater and the resource utilization of sludge.









Similar content being viewed by others
Data Availability
All data generated and analyzed in this study are included in this paper and the Supplementary Materials.
References
Ahmed, M., Zhou, J., Ngo, H. H., Guo, W., & Chen, M. (2016). Progress in the preparation and application of modified biochar for improved contaminant removal from water and wastewater. Bioresource Technology, 214, 836–851. https://doi.org/10.1016/j.biortech.2016.05.057
Badruddoza, A. Z. M., Tay, A. S. H., Tan, P., Hidajat, K., & Uddin, M. S. (2011). Carboxymethyl-β-cyclodextrin conjugated magnetic nanoparticles as nano-adsorbents for removal of copper ions: Synthesis and adsorption studies. Journal of Hazardous Materials, 185(2–3), 1177–1186.
Bahar, M. M., Mahbub, K. R., Naidu, R., & Megharaj, M. (2018). As(V) removal from aqueous solution using a low-cost adsorbent coir pith ash: Equilibrium and kinetic study. Environmental Technology & Innovation, 9, 198–209. https://doi.org/10.1016/j.eti.2017.12.005
Chen, Q., Yao, Y., Li, X., Lu, J., Zhou, J., & Huang, Z. (2018). Comparison of heavy metal removals from aqueous solutions by chemical precipitation and characteristics of precipitates. Journal of Water Process Engineering, 26, 289–300. https://doi.org/10.1016/j.jwpe.2018.11.003
Chowdhury, Z. Z., Hasan, M. R., Hamid, S. B. A., Samsudin, E. M., Zain, S. M., & Khalid, K. (2015). Catalytic pretreatment of biochar residues derived from lignocellulosic feedstock for equilibrium studies of manganese, Mn(II) cations from aqueous solution. Rsc Advances, 5, 6345–6356. https://doi.org/10.1039/C4RA09709B
Chung, S. G., Ryu, J. C., Song, M. K., An, B., Kim, S. B., Lee, S. H., & Choi, J. W. (2014). Modified composites based on mesostructured iron oxyhydroxide and synthetic minerals: A potential material for the treatment of various toxic heavy metals and its toxicity. Journal of Hazardous Materials, 267, 161–168. https://doi.org/10.1016/j.jhazmat.2013.12.056
Ding, Z., Hu, X., Wan, Y., Wang, S., & Gao, B. (2016). Removal of lead, copper, cadmium, zinc, and nickel from aqueous solutions by alkali-modified biochar: Batch and column tests. Journal of Industrial and Engineering Chemistry, 33, 239–245. https://doi.org/10.1016/j.jiec.2015.10.007
Duan, P., Ma, T., Yue, Y., Li, Y., Zhang, X., Shang, Y., Gao, B., Zhang, Q., Yue, Q., & Xu, X. (2019). Fe/Mn nanoparticles encapsulated in nitrogen-doped carbon nanotubes as a peroxymonosulfate activator for acetamiprid degradation. Environmental Science:Nano, 6, 1799–1811. https://doi.org/10.1039/C9EN00220K
Fonseca, M. G., Oliveira, M. M., Arakaki, L. N., Espinola, J. G. P., & Airoldi, C. (2005). Natural vermiculite as an exchanger support for heavy cations in aqueous solution. Journal of Colloid and Interface Science, 285(1), 50–55. https://doi.org/10.1016/j.jcis.2004.11.031
Gan, C., Liu, Y., Tan, X., Wang, S., Zeng, G., Zheng, B., Li, T., Jiang, Z., & Liu, W. (2015). Effect of porous zinc–biochar nanocomposites on Cr(VI) adsorption from aqueous solution. Rsc Advances, 5, 35107–35115. https://doi.org/10.1039/C5RA04416B
Gao, B., Chang, Q., Cai, J., Xi, Z., Li, A., & Yang, H. (2021). Removal of fluoroquinolone antibiotics using actinia-shaped lignin-based adsorbents: Role of the length and distribution of branched-chains. Journal of Hazardous Materials, 403, 123603. https://doi.org/10.1016/j.jhazmat.2020.123603
Hasan, S. H., & Srivastava, P. (2009). Batch and continuous biosorption of Cu2+ by immobilized biomass of Arthrobacter sp. Journal of Environmental Management, 90(11), 3313–3321. https://doi.org/10.1016/j.jenvman.2009.05.005
Hori, M., Shozugawa, K., & Matsuo, M. (2015). Reduction process of Cr(VI) by Fe(II) and humic acid analyzed using high time resolution XAFS analysis. Journal of Hazardous Materials, 285, 140–147. https://doi.org/10.1016/j.jhazmat.2014.11.047
Hua, Y., Wang, W., Huang, X., Gu, T., Ding, D., Ling, L., & Zhang, W. (2018). Effect of bicarbonate on aging and reactivity of nanoscale zerovalent iron (nZVI) toward uranium removal. Chemosphere, 201, 603–611. https://doi.org/10.1016/j.chemosphere.2018.03.041
Huang, H., Huang, H., & Wei, Y. (2017). Reduction of toxic Cr(VI)-humic acid in an ionic liquid. Spectrochimica Acta Part b: Atomic Spectroscopy, 133, 9–13. https://doi.org/10.1016/j.sab.2017.04.007
Huang, X., Wei, D., Zhang, X., Fan, D., Sun, X., Du, B., & Wei, Q. (2019). Synthesis of amino-functionalized magnetic aerobic granular sludge-biochar for Pb(II) removal: Adsorption performance and mechanism studies. Science of the Total Environment, 685, 681–689. https://doi.org/10.1016/j.scitotenv.2019.05.429
Kolodynska, D., Krukowska, J., & Thomas, P. (2017). Comparison of sorption and desorption studies of heavy metal ions from biochar and commercial active carbon. Chemical Engineering Journal, 307, 353–363. https://doi.org/10.1016/j.cej.2016.08.088
Larous, S., Meniai, A. H., & Lehocine, M. B. (2005). Experimental study of the removal of copper from aqueous solutions by adsorption using sawdust. Desalination, 185(1–3), 483–490. https://doi.org/10.1016/j.desal.2005.03.090
Li, S., Wang, W., Yan, W., & Zhang, W. X. (2014). Nanoscale zero-valent iron (nZVI) for the treatment of concentrated Cu(II) wastewater: A field demonstration. Environmental Science: Processes & Impacts, 16, 524–533. https://doi.org/10.1039/C3EM00578J
Li, J., Li, M., Wang, S., Yang, X., Liu, F., & Liu, X. (2020). Key role of pore size in Cr(VI) removal by the composites of 3-dimentional mesoporous silica nanospheres wrapped with polyaniline. Science of the Total Environment, 729, 139009. https://doi.org/10.1016/j.scitotenv.2020.139009
Ling, L., Huang, X., & Zhang, W. (2018). Enrichment of precious metals from wastewater with core–shell nanoparticles of iron. Advanced Materials, 30(17), 1705703. https://doi.org/10.1002/adma.201705703
Liu, Z., Zhang, F., & Wu, J. (2010). Characterization and application of chars produced from pinewood pyrolysis and hydrothermal treatment. Fuel, 89, 510–514. https://doi.org/10.1016/j.fuel.2009.08.042
Liu, F., Shan, C., Zhang, X., Zhang, Y., Zhang, W., & Pan, W. (2017). Enhanced removal of EDTA-chelated Cu(II) by polymeric anion-exchanger supported nanoscale zero-valent iron. Journal of Hazardous Materials, 321, 290–298. https://doi.org/10.1016/j.jhazmat.2016.09.022
Liu, X. H. Y., Zhao, Z., & Chen, X. (2018). Study on removal of copper from nickel-copper mixed solution by membrane electrolysis. Hydrometallurgy, 180, 153–157. https://doi.org/10.1016/j.hydromet.2018.07.019
Liu, L., Zhao, J., Bai, S., Lin, H., & Wang, D. (2021). Reduction and removal of As(V) in aqueous solution by biochar derived from nano zero-valent-iron (nZVI) and sewage sludge. Chemosphere, 277, 130273. https://doi.org/10.1016/j.chemosphere.2021.130273
Lyu, C., Yang, X., Zhang, S., Zhang, Q., & Su, X. (2019). Preparation and performance of manganese-oxide-coated zeolite for the removal of manganese-contamination in groundwater. Environmental Technology, 40(7), 878–887. https://doi.org/10.1080/09593330.2017.1410579
Meng, J., Wang, L., Liu, X., Wu, J., Brookes, P. C., & Xu, J. (2013). Physicochemical properties of biochar produced from aerobically composted swine manure and its potential use as an environmental amendment. Bioresource Technology, 142, 641–646. https://doi.org/10.1016/j.biortech.2013.05.086
Oliveira, F. R., Patel, A. K., Jaisi, D. P., Adhikari, S., Lu, H., & Khanal, S. K. (2017). Environmental application of biochar: Current status and perspectives. Bioresource Technology, 246, 110–122. https://doi.org/10.1016/j.biortech.2017.08.122
Peiris, C., Wathudura, P. D., Gunatilake, S. R., Gajanayake, B., Wewalwela, J. J., Abeysundara, S., & Vithanage, M. (2022). Effect of acid modified tea-waste biochar on crop productivity of red onion (Allium cepa L.). Chemosphere, 288, 132551. https://doi.org/10.1016/j.chemosphere.2021.132551
Peng, Y., Yu, W., & Zheng, Y. (2018). Recovery of magnetite from waste ferrous sulfate using polyethylene glycol (PEG) as a dispersant. Transactions of Nonferrous Metals Society of China, 28(7), 1465–1474. https://doi.org/10.1016/S1003-6326(18)64785-5
Qambrani, N. A., Rahman, M. M., Won, S., Shim, S., & Ra, C. (2017). Biochar properties and eco-friendly applications for climate change mitigation, waste management, and wastewater treatment: A review. Renewable and Sustainable Energy Reviews, 79, 255–273. https://doi.org/10.1016/j.rser.2017.05.057
Ren, N., Tang, Y., & Li, M. (2018). Mineral additive enhanced carbon retention and stabilization in sewage sludge-derived biochar. Process Safety and Environmental Protection, 115, 70–78. https://doi.org/10.1016/j.psep.2017.11.006
Shang, X., Yang, L., Ouyang, D., Zhang, B., Zhang, W., Gu, M., Li, J., Chen, M., Huang, L., & Qian, L. (2020). Enhanced removal of 1,2,4-trichlorobenzene by modified biochar supported nanoscale zero-valent iron and palladium. Chemosphere, 249, 126518. https://doi.org/10.1016/j.chemosphere.2020.126518
Shao, Y., Gao, Y., Yue, Q., Kong, W., Gao, B., Wang, W., & Jiang, W. (2020). Degradation of chlortetracycline with simultaneous removal of copper (II) from aqueous solution using wheat straw-supported nanoscale zero-valent iron. Chemical Engineering Journal, 379, 122384. https://doi.org/10.1016/j.cej.2019.122384
Shen, B., Li, G., Wang, F., Wang, Y., He, C., Zhang, M., & Singh, S. (2015). Elemental mercury removal by the modified bio-char from medicinal residues. Chemical Engineering Journal, 272, 28–37. https://doi.org/10.1016/j.cej.2015.03.006
Tan, P., Sun, J., Hu, Y., Fang, Z., Bi, Q., Chen, Y., & Cheng, J. (2015). Adsorption of Cu2+, Cd2+ and Ni2+ from aqueous single metal solutions on graphene oxide membranes. Journal of Hazardous Materials, 297, 251–260. https://doi.org/10.1016/j.jhazmat.2015.04.068
Tang, Y., Li, Y., Zhan, L., Wu, D., Zhang, S., Pang, R., & Xie, B. (2022). Removal of emerging contaminants (bisphenol A and antibiotics) from kitchen wastewater by alkali-modified biochar. Science of the Total Environment, 805, 150158. https://doi.org/10.1016/j.scitotenv.2021.150158
Trakal, L., Sigut, R., Sillerova, H., Faturikova, D., & Komarek, M. (2014). Copper removal from aqueous solution using biochar: Effect of chemical activation. Arabian Journal of Chemistry, 7(1), 43–52. https://doi.org/10.1016/j.arabjc.2013.08.001
Wang, H., Gao, B., Wang, S., Fang, J., Xue, Y., & Yang, K. (2015). Removal of Pb(II), Cu(II), and Cd(II) from aqueous solutions by biochar derived from KMnO4 treated hickory wood. Bioresource Technology, 197, 356–362. https://doi.org/10.1016/j.biortech.2015.08.132
Wu, H., Feng, Q., Yang, H., Lu, P., Gao, B., & Alansari, A. (2019). Enhanced phenanthrene removal in aqueous solution using modified biochar supported nano zero-valent iron. Environmental Technology, 40(23), 3114–3123. https://doi.org/10.1080/09593330.2018.1549104
Xu, Z., Lin, Y., Lin, Y., Yang, D., & Zheng, H. (2021). Adsorption behaviors of paper mill sludge biochar to remove Cu, Zn and As in wastewater. Environmental Technology & Innovation, 23, 101616. https://doi.org/10.1016/j.eti.2021.101616
Yang, Y., Lin, X., Wei, B., Zhao, Y., & Wang, J. (2014). Evaluation of adsorption potential of bamboo biochar for metal-complex dye: Equilibrium, kinetics and artificial neural network modeling. International Journal of Environmental Science and Technology, 11, 1093–1100. https://doi.org/10.1007/s13762-013-0306-0
Yang, D., Wang, L., Li, Z., Tang, X., He, M., Yang, S., Liu, X., & Xu, J. (2020). Simultaneous adsorption of Cd(II) and As(III) by a novel biochar-supported nanoscale zero-valent iron in aqueous systems. Science of the Total Environment, 708, 134823. https://doi.org/10.1016/j.scitotenv.2019.134823
Yu, Y., Qiao, N., Wang, D., Zhu, Q., Fu, F., Cao, R., Wang, R., Liu, W., & Xu, B. (2019). Fluffy honeycomb-like activated carbon from popcorn with high surface area and well-developed porosity for ultra-high efficiency adsorption of organic dyes. Bioresource Technology, 285, 121340. https://doi.org/10.1016/j.biortech.2019.121340
Zhang, J., Chen, S., Zhang, H., & Wang, X. (2017). Removal behaviors and mechanisms of hexavalent chromium from aqueous solution by cephalosporin residue and derived chars. Bioresource Technology, 238, 484–491. https://doi.org/10.1016/j.biortech.2017.04.081
Zhang, Y., Zheng, Y., Yang, Y., Huang, J., Zimmerman, A. R., Chen, H., Hu, X., & Gao, B. (2021). Mechanisms and adsorption capacities of hydrogen peroxide modified ball milled biochar for the removal of methylene blue from aqueous solutions. Bioresource Technology, 337, 125432. https://doi.org/10.1016/j.biortech.2021.125432
Acknowledgements
This work was supported by the Natural Science Foundation of Shandong Province, China [grant no. ZR2021ME214] and the Jinan City Talent Development Project in 2021 (2021GXRC067 and, 2021GXRC124).
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study’s conception and design. Hao Xue: Conceptualization, methodology, investigation, writing—original draft, and writing—review and editing; Yingying Shao: methodology and, investigation; Xinhua Shi: methodology and, investigation; Yanqiu Shao: writing—review and editing; Weiyi Zhang: writing—review and editing; Ying Zhu: funding acquisition, supervision, and writing—review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. Sodium hydroxide-modified sludge biochar (NaOH-SBC) was used to stabilize zero-valent iron nanoparticles.
2. Cu(II) removal by three types of biochar materials was compared.
3. Effects of the initial pH of the solution, initial concentration of Cu(II), adsorbent dose, reaction time, coexisting ions, and humic acid (HA) concentration on the removal of Cu(II) were investigated.
4. The mechanism of Cu(II) removal by nZVI supported by alkali-modified sludge biochar was probed.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xue, H., Shao, Y., Shi, X. et al. Removal of Cu2+ in Aqueous Solution with Nano-zero-Valent Iron (nZVI) Supported by Alkali-Modified Sludge Biochar. Water Air Soil Pollut 233, 515 (2022). https://doi.org/10.1007/s11270-022-05909-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05909-x