Abstract
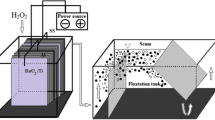
The electrooxidation process, one of the advanced oxidation processes, is one of the effective treatment processes used in treating various industrial wastewaters. This study investigated the treatment of olive mill wastewater using the electrooxidation process. This study includes the effects of different experimental parameters on chemical oxygen demand and total phenol removal efficiencies in olive mill wastewater. Ti/IrO2/RuO2 mesh plates as anode material and Ti mesh plates as cathode material were used in the study. The effects of stirring rate, dilution factor, pH, type of support electrolyte, the concentration of support electrolyte, and current density on chemical oxygen demand and total phenol removal efficiencies were examined in the experiments using a batch reactor. The study found that the chemical oxygen demand and total phenol removal rates were 96.93% and 100% under optimum conditions, respectively. According to the treatment data obtained, it can be said that olive mill wastewater can be treated by the electrooxidation method and can be proposed as a pretreatment system before entering biological treatment.











Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Adhoum, N., & Monser, L. (2004). Decolourization and removal of phenolic compounds from olive mill wastewater by electrocoagulation. Chemical Engineering and Processing: Process Intensification, 43(10), 1281–1287. https://doi.org/10.1016/j.cep.2003.12.001
Al-Bsoul, A., Al-Shannag, M., Tawalbeh, M., Al-Taani, A. A., Lafi, W. K., Al-Othman, A., & Alsheyab, M. (2020). Optimal conditions for olive mill wastewater treatment using ultrasound and advanced oxidation processes. Science of the Total Environment, 700, 134576. https://doi.org/10.1016/j.scitotenv.2019.134576
APHA. (2005). Standard methods for the examination of water and wastewater. American Public Health Association
Atanassova, D., Kefalas, P., Petrakis, C., Mantzavinos, D., Kalogerakis, N., & Psillakis, E. (2005). Sonochemical reduction of the antioxidant activity of olive mill wastewater. Environment International, 31(2), 281–287. https://doi.org/10.1016/j.envint.2004.10.004
Barbera, A., Maucieri, C., Cavallaro, V., Ioppolo, A., & Spagna, G. (2013). Effects of spreading olive mill wastewater on soil properties and crops, a review. Agricultural Water Management, 119, 43–53. https://doi.org/10.1016/j.agwat.2012.12.009
Bashir, M. J. K., Lim, J. W., Aziz, S. Q., & Amr, S. S. A. (2014). Electrochemical methods: Electrochemical oxidation process contribution in remediating complicated wastewaters. In H. A. Aziz & A. Mojiri (Eds.), Wastewater engineering: Advanced wastewater treatment systems (pp. 96–106). IJSR Publications.
Bayar, S., Yıldız, Y. Ş., Yılmaz, A. E., & İrdemez, Ş. (2011). The effect of stirring speed and current density on removal efficiency of poultry slaughterhouse wastewater by electrocoagulation method. Desalination, 280(1–3), 103–107. https://doi.org/10.1016/j.desal.2011.06.061
Bingül, Z., İrdemez, Ş, Kul, S., Ekmekyapar Torun, F., & Demircioğlu, N. (2021a). Investigation of organic and inorganic matters removal from tannery wastewater using iron plate electrode by electrocoagulation process. International Journal of Environmental Analytical Chemistry. https://doi.org/10.1080/03067319.2021.1953002
Bingül, Z., İrdemez, Ş, & Demircioğlu, N. (2021b). Effect of controlled and uncontrolled pH on tannery wastewater treatment by the electrocoagulation process. International Journal of Environmental Analytical Chemistry. https://doi.org/10.1080/03067319.2021.1925261
Bottino, A., Capannelli, G., Comite, A., Costa, C., Firpo, R., Jezowska, A., & Pagliero, M. (2020). Treatment of olive mill wastewater through integrated pressure-driven membrane processes. Membranes, 10(11), 334. https://doi.org/10.3390/membranes10110334
Boubaker, F., & Ridha, B. C. (2007). Anaerobic co-digestion of olive mill wastewater with olive mill solid waste in a tubular digester at mesophilic temperature. Bioresource Technology, 98(4), 769–774. https://doi.org/10.1016/j.biortech.2006.04.020
Chatzisymeon, E., Dimou, A., Mantzavinos, D., & Katsaounis, A. (2009). Electrochemical oxidation of model compounds and olive mill wastewater over DSA electrodes: 1. The case of Ti/IrO2 anode. Journal of Hazardous Materials, 167(1–3), 268–274. https://doi.org/10.1016/j.jhazmat.2008.12.117
Chatzisymeon, E., Xekoukoulotakis, N. P., Diamadopoulos, E., Katsaounis, A., & Mantzavinos, D. (2009). Boron-doped diamond anodic treatment of olive mill wastewaters: Statistical analysis, kinetic modeling and biodegradability. Water Research, 43(16), 3999–4009. https://doi.org/10.1016/j.watres.2009.04.007
Chinarro, E., Pérez Orosa, L., García-Alegre, M., & Guinea, D. (2020). Graphite electrodes for hydrogen generation in alkali electrolysis assisted by an organic waste water compound: Olive mill wastewater. Grupo Español Del Carbón, 55, 8–12.
Deligiorgis, A., Xekoukoulotakis, N. P., Diamadopoulos, E., & Mantzavinos, D. (2008). Electrochemical oxidation of table olive processing wastewater over boron-doped diamond electrodes: Treatment optimization by factorial design. Water Research, 42(4–5), 1229–1237. https://doi.org/10.1016/j.watres.2007.09.014
Deng, Y., & Englehardt, J. D. (2007). Electrochemical oxidation for landfill leachate treatment. Waste Management, 27(3), 380–388. https://doi.org/10.1016/j.wasman.2006.02.004
Ekmekyapar Torun, F., Cengiz, İ, & Kul, S. (2020). Zeytin Karasuyunun İleri Oksidasyon Prosesleri İle Arıtımının İncelenmesi. Journal of the Institute of Science and Technology, 10(3), 1597–1606. https://doi.org/10.21597/jist.687345
Elkacmi, R., Boudouch, O., Hasib, A., Bouzaid, M., & Bennajah, M. (2020). Photovoltaic electrocoagulation treatment of olive mill wastewater using an external-loop airlift reactor. Sustainable Chemistry and Pharmacy, 17, 100274. https://doi.org/10.1016/j.scp.2020.100274
Fıl, B. A., Boncukcuoğlu, R., Yilmaz, A. E., & Bayar, S. (2014). Electro-oxidation of pistachio processing industry wastewater using graphite anode. Clean-Soil Air Water, 42(9), 1232–1238. https://doi.org/10.1002/clen.201300560
Folin, O., & Ciocalteu, V. (1927). On tyrosine and tryptophane determinations in proteins. Journal of Biological Chemistry, 73(2), 627–650. https://doi.org/10.1016/S0021-9258(18)84277-6
Ghahrchi, M., Rezaee, A., & Adibzadeh, A. (2021). Study of kinetic models of olive oil mill wastewater treatment using electrocoagulation process. Desalination and Water Treatment, 211, 123–130. https://doi.org/10.5004/dwt.2021.26516
Giannis, A., Kalaitzakis, M., & Diamadopoulos, E. (2007). Electrochemical treatment of olive mill wastewater. Journal of Chemical Technology & Biotechnology: International Research in Process, Environmental & Clean Technology, 82(7), 663–671. https://doi.org/10.1002/jctb.1725
Görmez, F., Görmez, Ö., Yabalak, E., & Gözmen, B. (2020). Application of the central composite design to mineralization of olive mill wastewater by the electro/FeII/persulfate oxidation method. SN Applied Sciences, 2(178), 1–11. https://doi.org/10.1007/s42452-020-1986-y
Gotsi, M., Kalogerakis, N., Psillakis, E., Samaras, P., & Mantzavinos, D. (2005). Electrochemical oxidation of olive oil mill wastewaters. Water Research, 39(17), 4177–4187. https://doi.org/10.1016/j.watres.2005.07.037
Khani, M. R., Mahdizadeh, H., Kannan, K., Kalankesh, L. R., Kamarehei, B., Baneshi, M. M., & Shahamat, Y. D. (2020). Olive mill wastewater (OMW) treatment by hybrid processes of electrocoagulation/catalytic ozonation and biodegradation. Environmental Engineering & Management Journal (EEMJ), 19(8), 1401–1410.
Kul, S., Boncukcuoğlu, R., Yilmaz, A. E., & Fil, B. A. (2015). Treatment of olive mill wastewater with electro-oxidation method. Journal of the Electrochemical Society, 162(8), G41.
Kul, S., & Nuhoğlu, A. (2020). Removal kinetics of olive-mill wastewater in a batch-operated aerobic bioreactor. Journal of Environmental Engineering, 146(3), 04019122.
Kul, S., Nuhoğlu, A., & Değermenci, N. (2014). Zeytin Karasuyunun Respirometrik Analizi. Journal of the Institute of Science and Technology, 4(3), 35–40.
McNamara, C. J., Anastasiou, C. C., O’Flaherty, V., & Mitchell, R. (2008). Bioremediation of olive mill wastewater. International Biodeterioration & Biodegradation, 61(2), 127–134. https://doi.org/10.1016/j.ibiod.2007.11.003
Nogueira, V., Lopes, I., Rocha-Santos, T., Gonçalves, F., Duarte, A., & Pereira, R. (2016). Photocatalytic treatment of olive oil mill wastewater using TiO2 and Fe2O3 nanomaterials. Water, Air, & Soil Pollution, 227, 88. https://doi.org/10.1007/s11270-016-2787-1
Ozturk, D., & Yilmaz, A. E. (2019). Treatment of slaughterhouse wastewater with the electrochemical oxidation process: Role of operating parameters on treatment efficiency and energy consumption. Journal of Water Process Engineering, 31, 100834.
Panizza, M., & Cerisola, G. (2006). Olive mill wastewater treatment by anodic oxidation with parallel plate electrodes. Water Research, 40(6), 1179–1184. https://doi.org/10.1016/j.watres.2006.01.020
Papastefanakis, N., Mantzavinos, D., & Katsaounis, A. (2010). DSA electrochemical treatment of olive mill wastewater on Ti/RuO2 anode. Journal of Applied Electrochemistry, 40(4), 729–737. https://doi.org/10.1007/s10800-009-0050-9
Piya-Areetham, P., Shenchunthichai, K., & Hunsom, M. (2006). Application of electrooxidation process for treating concentrated wastewater from distillery industry with a voluminous electrode. Water Research, 40(15), 2857–2864. https://doi.org/10.1016/j.watres.2006.05.025
Scialdone, O., Randazzo, S., Galia, A., & Filardo, G. (2009). Electrochemical oxidation of organics at metal oxide electrodes: The incineration of oxalic acid at IrO2–Ta2O5 (DSA-O2) anode. Electrochimica Acta, 54(4), 1210–1217. https://doi.org/10.1016/j.electacta.2008.08.064
Tufaner, F. (2020). Zeytin Karasuyunun Anaerobik Arıtılabilirliği ve Biyogaz Üretim Potansiyelinin Araştırılması. Bitlis Eren Üniversitesi Fen Bilimleri Dergisi, 9(4), 1766–1778. https://doi.org/10.17798/bitlisfen.676940
Un, U. T., Altay, U., Koparal, A. S., & Ogutveren, U. B. (2008). Complete treatment of olive mill wastewaters by electrooxidation. Chemical Engineering Journal, 139(3), 445–452. https://doi.org/10.1016/j.cej.2007.08.009
Ün, Ü. T., Uğur, S., Koparal, A., & Öğütveren, Ü. B. (2006). Electrocoagulation of olive mill wastewaters. Separation and Purification Technology, 52(1), 136–141. https://doi.org/10.1016/j.seppur.2006.03.029
Acknowledgements
The authors are grateful for Atatürk University Department of Environmental Engineering’s laboratory support.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by Sinan Kul, Recep Boncukcuoğlu, Fatma Ekmekyapar Torun, Züleyha Reçber, Onur Sözüdoğru, and Erdinç Aladağ. Sinan Kul wrote the first draft of the manuscript and all authors commented on previous versions. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kul, S., Boncukcuoğlu, R., Ekmekyapar Torun, F. et al. Investigation of the Treatment of Olive Mill Wastewater by Electrooxidation. Water Air Soil Pollut 233, 421 (2022). https://doi.org/10.1007/s11270-022-05894-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05894-1