Abstract
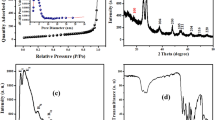
In this study, two methods were used to synthesize the NiO/C composite from agricultural waste. The mesoporous composite was successfully synthesized via a novel precipitation method in the presence of dissolved gases. The morphology of the composites was differentiated by using characterization techniques such as X-ray diffraction, the point of zero charge (pHpzc), field-emission scanning electron microscopy (FESEM), Fourier transform infrared spectroscopy, energy-dispersive X-ray analysis (EDAX), and vibrating sample magnetometry (VSM). Then, the mechanism of synthesis was elucidated using the above experimental characterization data. Results of FESEM and EDAX analyses of Ni(OH)2–carbon composite clearly showed the role of dissolved gases in the synthesis. Both the composites were subjected as the adsorbent to remove the toxic Pb(II) ions from the wastewater. Batch adsorption experiments were carried out to compare the Pb(II) ion removal capability of both the composite materials. The parameters such as the effect of pH, the dosage of the adsorbents, and initial concentration were studied. At the optimized conditions, isotherm studies for each of the adsorbent were also carried out. The isotherm results revealed that the maximum removal capacity qe (mg/g) was 30.78 for PJNC and 43.48 for PJGNC. The VSM analysis confirmed that both the adsorbents were soft magnetic materials. Hence, they could be competently separated from salted/treated water using a magnetic field.













Similar content being viewed by others
References
Acharya, J., Sahu, J. N., Mohanty, C. R., & Meikap, B. C. (2009). Removal of lead(II) from wastewater by activated carbon developed from Tamarind wood by zinc chloride activation. Chemical Engineering Journal, 149(1–3), 249–262. https://doi.org/10.1016/j.cej.2008.10.029
Ahilandeswari, E., Kanna, R. R., & Sakthipandi, K. (2020). Synthesis of neodymium-doped barium nanoferrite: Analysis of structural, optical, morphological, and magnetic properties. Physica B: Condensed Matter, 599, 412425. https://doi.org/10.1016/j.physb.2020.412425
Ahrouch, M., Gatica, J. M., Draoui, K., Bellido, D., & Vidal, H. (2019). Lead removal from aqueous solution by means of integral natural clays honeycomb monoliths. Journal of Hazardous Materials, 365, 519–530. https://doi.org/10.1016/j.jhazmat.2018.11.037
Bharath, G., Abdul Hai, K., Rambabu, T. P., Hasan, S. W., & Banat, F. (2021). Designed assembly of Ni/MAX (Ti3AlC2) and porous graphene-based asymmetric electrodes for capacitive deionization of multivalent ions. Chemosphere, 266, 129048. https://doi.org/10.1016/j.chemosphere.2020.129048
Bouabidi, Z. B., El-Naas, M. H., Cortes, D., & McKay, G. (2018). Steel-Making dust as a potential adsorbent for the removal of lead (II) from an aqueous solution. Chemical Engineering Journal, 334(Ii), 837–844. https://doi.org/10.1016/j.cej.2017.10.073
Cai, Y., Zheng, L., & Fang, Z. (2015). Selective adsorption of Cu(II) from an aqueous solution by ion imprinted magnetic chitosan microspheres prepared from steel pickling waste liquor. RSC Advances, 5(118), 97435–97445. https://doi.org/10.1039/c5ra16547d
Chandrasekaran, A., Patra, C., Narayanasamy, S., & Subbiah, S. (2020). Adsorptive removal of Ciprofloxacin and Amoxicillin from single and binary aqueous systems using acid-activated carbon from Prosopis juliflora. Environmental Research, 188, 109825. https://doi.org/10.1016/j.envres.2020.109825
Dada AO, Olalekan AP, Olatunya AM, Dada O (2012) Langmuir, Freundlich, Temkin and Dubinin – Radushkevich isotherms studies of equilibrium sorption of Zn 2 + unto phosphoric acid modified rice husk. 3(1), 38–45.
El-Keblawy, A., & Abdelfatah, M. A. (2014). Impacts of native and invasive exotic prosopis congeners on soil properties and associated flora in the arid united arab emirates. Journal of Arid Environments, 100–101, 1–8. https://doi.org/10.1016/j.jaridenv.2013.10.001
Estela MCCB, Juliana S, Matos TTS, Mayara RF (2018) Effect of surface and porosity of biochar on water holding capacity aiming indirectly at preservation of the Amazon biome, (November 2017), 1–9. https://doi.org/10.1038/s41598-018-28794-z
Feygenson, M., Kou, A., Kreno, L. E., Tiano, A. L., Patete, J. M., Zhang, F., & Aronson, M. C. (2010). Properties of highly crystalline NiO and Ni nanoparticles prepared by high-temperature oxidation and reduction. PHYSICAL REVIEW, B, 81(014420), 1–9. https://doi.org/10.1103/PhysRevB.81.014420
Fu, W., Wang, X., & Huang, Z. (2019). Science of the Total Environment Remarkable reusability of magnetic Fe 3 O 4 -encapsulated C 3 N 3 S 3 polymer / reduced graphene oxide composite : A highly effective adsorbent for Pb and Hg ions. Science of the Total Environment, 659, 895–904. https://doi.org/10.1016/j.scitotenv.2018.12.303
Gerçel, Ö., & Gerçel, H. F. (2007). Adsorption of lead(II) ions from aqueous solutions by activated carbon prepared from biomass plant material of Euphorbia rigida. Chemical Engineering Journal, 132(1–3), 289–297. https://doi.org/10.1016/j.cej.2007.01.010
Ghaemi, N., Zereshki, S., & Heidari, S. (2017). Removal of lead ions from water using PES-based nanocomposite membrane incorporated with polyaniline modified GO nanoparticles: Performance optimization by central composite design. Process Safety and Environmental Protection, 111, 475–490. https://doi.org/10.1016/j.psep.2017.08.011
Goel, J., Kadirvelu, K., Rajagopal, C., & Garg, V. K. (2005). Removal of lead(II) by adsorption using treated granular activated carbon: Batch and column studies. Journal of Hazardous Materials, 125(1–3), 211–220. https://doi.org/10.1016/j.jhazmat.2005.05.032
Gupta, V. K., Agarwal, S., & Saleh, T. A. (2011). Synthesis and characterization of alumina-coated carbon nanotubes and their application for lead removal. Journal of Hazardous Materials, 185(1), 17–23. https://doi.org/10.1016/j.jhazmat.2010.08.053
Habtegebrel, M. M., & Khan, M. A. (2018). Removal of Zn ( II ) and Cu ( II ) ions from aqueous solution by dried Prosopis juliflora. 6(1), 6–14. https://doi.org/10.11648/j.mc.20180601.12
Hale, D. K. (1976). Review The physical properties of composite materials. Journal of Materials Science, 11, 2105–2141.
Hameed, B. H., Tan, I. A. W., & Ahmad, A. L. (2008). Adsorption isotherm, kinetic modeling and mechanism of 2,4,6-trichlorophenol on coconut husk-based activated carbon. Chemical Engineering Journal, 144(2), 235–244. https://doi.org/10.1016/j.cej.2008.01.028
Hua, M., Zhang, S., Pan, B., Zhang, W., Lv, L., & Zhang, Q. (2012). Heavy metal removal from water/wastewater by nanosized metal oxides: A review. Journal of Hazardous Materials, 211–212, 317–331. https://doi.org/10.1016/j.jhazmat.2011.10.016
Huang, X. H., Tu, J. P., Zhang, C. Q., Chen, X. T., Yuan, Y. F., & Wu, H. M. (2007). Spherical NiO-C Composite for Anode Material of Lithium Ion Batteries, 52, 4177–4181. https://doi.org/10.1016/j.electacta.2006.11.034
Indhu, S., & Muthukumaran, K. (2018). Removal and recovery of reactive yellow 84 dye from wastewater and regeneration of functionalised Borassus flabellifer activated carbon. Journal of Environmental Chemical Engineering, 6(2), 3111–3121. https://doi.org/10.1016/j.jece.2018.04.027
Inoue, M., & Hirasawa, I. (2013). The relationship between crystal morphology and XRD peak intensity. Journal of Crystal Growth, 380, 169–175. https://doi.org/10.1016/j.jcrysgro.2013.06.017
Jaiswal, A., Mani, R., Banerjee, S., Gautam, R. K., & Chattopadhyaya, M. C. (2015). Synthesis of novel nano-layered double hydroxide by urea hydrolysis method and their application in removal of chromium(VI) from aqueous solution: Kinetic, thermodynamic and equilibrium studies. Journal of Molecular Liquids, 202, 52–61. https://doi.org/10.1016/j.molliq.2014.12.004
Jayaram, K., & Prasad, M. N. V. (2009). Removal of Pb(II) from aqueous solution by seed powder of Prosopis juliflora DC. Journal of Hazardous Materials, 169(1–3), 991–997. https://doi.org/10.1016/j.jhazmat.2009.04.048
Khalil HPSA, Yusra AFI, Bhat AH, Jawaid M (2010) Cell wall ultrastructure , anatomy , lignin distribution , and chemical composition of Malaysian cultivated kenaf fiber. 31, 113–121. https://doi.org/10.1016/j.indcrop.2009.09.008
Khandanlou, R., Ahmad, M. B., Masoumi, H. R. F., Shameli, K., Basri, M., & Kalantari, K. (2015). Rapid adsorption of copper(II) and lead(II) by rice straw/Fe3O4nanocomposite: Optimization, equilibrium isotherms, and adsorption kinetics study. PLoS ONE, 10(3), 1–19. https://doi.org/10.1371/journal.pone.0120264
Khozhaenko, E., Kovalev, V., Podkorytova, E., & Khotimchenko, M. (2016). Science of the total environment removal of the metal ions from aqueous solutions by nanoscaled low molecular pectin isolated from seagrass Phyllospadix iwatensis. Science of the Total Environment. https://doi.org/10.1016/j.scitotenv.2016.01.108
Kloss, S., Zehetner, F., Dellantonio, A., Hamid, R., Ottner, F., Liedtke, V., … Soja, G. (2012). Characterization of slow pyrolysis biochars: Effects of Feedstocks and Pyrolysis Temperature on Biochar Properties. https://doi.org/10.2134/jeq2011.0070
Kosmulski, M. (2009). pH-dependent surface charging and points of zero charge. IV. Update and new approach. Journal of Colloid and Interface Science, 337, 439–448. https://doi.org/10.1016/j.jcis.2009.04.072
Li, Y., Li, W., Liu, Q., Meng, H., Lu, Y., & Li, C. (2018). Alkynyl carbon materials as novel and efficient sorbents for the adsorption of mercury(II) from wastewater. Journal of Environmental Sciences (china), 68(Chunxi Li), 169–176. https://doi.org/10.1016/j.jes.2016.12.016
Luzardo, F. H. M., Velasco, F. G., Correia, I. K. S., Silva, P. M. S., & Salay, L. C. (2017). Removal of lead ions from water using a resin of mimosa tannin and carbon nanotubes. Environmental Technology and Innovation, 7, 219–228. https://doi.org/10.1016/j.eti.2017.03.002
Mahmoud, A. M., Ibrahim, F. A., Shaban, S. A., & Youssef, N. A. (2015). Adsorption of heavy metal ion from aqueous solution by nickel oxide nano catalyst prepared by different methods. Egyptian Journal of Petroleum, 24(1), 27–35. https://doi.org/10.1016/j.ejpe.2015.02.003
Modwi, A., Khezami, L., Taha, K., Al-Duaij, O. K., & Houas, A. (2017). Fast and high efficiency adsorption of Pb(II) ions by Cu/ZnO composite. Materials Letters, 195(Ii), 41–44. https://doi.org/10.1016/j.matlet.2017.02.089
Mousa, S. M., Ammar, N. S., & Ibrahim, H. A. (2016). Removal of lead ions using hydroxyapatite nano-material prepared from phosphogypsum waste. Journal of Saudi Chemical Society, 20(3), 357–365. https://doi.org/10.1016/j.jscs.2014.12.006
Pallarés, J., González-cencerrado, A., & Arauzo, I. (2018). Biomass and bioenergy production and characterization of activated carbon from barley straw by physical activation with carbon dioxide and steam. Biomass and Bioenergy, 115(January), 64–73. https://doi.org/10.1016/j.biombioe.2018.04.015
Patra, C., Shahnaz, T., Subbiah, S., & Narayanasamy, S. (2020). Comparative assessment of raw and acid-activated preparations of novel Pongamia pinnata shells for adsorption of hexavalent chromium from simulated wastewater. Environmental Science and Pollution Research, 27(13), 14836–14851. https://doi.org/10.1007/s11356-020-07979-y
Poinern, G. E., Brundavanam, R. K., Mondinos, N., & Jiang, Z. T. (2009). Synthesis and characterisation of nanohydroxyapatite using an ultrasound assisted method. Ultrasonics Sonochemistry, 16(4), 469–474. https://doi.org/10.1016/j.ultsonch.2009.01.007
Priya, N. S., Kamala, S. S. P., Anbarasu, V., Azhagan, S. A., & Saravanakumar, R. (2018). Characterization of CdS thin films and nanoparticles by a simple chemical bath technique. Materials Letters, 220, 161–164. https://doi.org/10.1016/j.matlet.2018.03.009
Saranya, N., Suganya, E., Narayanasamy, S., Sivaprakasam, S., Sivasubramanian, V., Pandian, S., & Selvaraj, R. (2020). 3-level Box-Behnkenoptimization of hexavalent chromium reduction by chromate resistant Trichoderma asperellum cells from simulated and industrial effluent. Environmental Technology and Innovation, 19, 101024. https://doi.org/10.1016/j.eti.2020.101024
Saravanakumar, R., Muthukumaran, K., & Selvaraju, N. (2019). Enhanced Pb ( II ) ions removal by using magnetic NiO / biochar composite enhanced Pb ( II ) ions removal by using magnetic NiO / biochar composite. Mater Res Express, 6, 105504.
Saravanakumar, S. S., Kumaravel, A., Nagarajan, T., Sudhakar, P., & Baskaran, R. (2013). Characterization of a novel natural cellulosic fiber from Prosopis juliflora bark. Carbohydrate Polymers, 92(2), 1928–1933. https://doi.org/10.1016/j.carbpol.2012.11.064
Sdiri, A., & Higashi, T. (2013). Simultaneous removal of heavy metals from aqueous solution by natural limestones. Applied Water Science, 3(1), 29–39. https://doi.org/10.1007/s13201-012-0054-1
Selvaraju, G., Kartini, N., & Bakar, A. (2018). Eco-friendly activated carbon from artocarpus integer bio-waste by. Journal of the Taiwan Institute of Chemical Engineers, 1–13. https://doi.org/10.1016/j.jtice.2018.08.011
Shackleton, R. T., Le Maitre, D. C., Van Wilgen, B. W., & Richardson, D. M. (2015). The impact of invasive alien Prosopis species (mesquite) on native plants in different environments in South Africa. South African Journal of Botany, 97, 25–31. https://doi.org/10.1016/j.sajb.2014.12.008
Shahnaz, P., Subbiah, S., & Narayanasamy, S. (2020). Comparative assessment of raw and acid-activated preparations of novel Pongamia pinnata shells for adsorption of hexavalent chromium from simulated wastewater. Environmental Science and Pollution Research, 27(13), 14836–14851. https://doi.org/10.1007/s11356-020-07979-y
Shahnaz, T., Vishnu Priyan, V., Pandian, S., & Narayanasamy, S. (2021). Use of nanocellulose extracted from grass for adsorption abatement of Ciprofloxacin and Diclofenac removal with phyto, and fish toxicity studies. Environmental Pollution, 268, 115494. https://doi.org/10.1016/j.envpol.2020.115494
Shaheen, S. M., Niazi, N. K., Hassan, N. E. E., Bibi, I., Wang, H., Tsang, D. C. W., & Rinklebe, J. (2018). Wood-based biochar for the removal of potentially toxic elements in water and wastewater: A critical review. International Materials Reviews, 1–32. https://doi.org/10.1080/09506608.2018.1473096
Shen, D. K., & Gu, S. (2009). Bioresource Technology The mechanism for thermal decomposition of cellulose and its main products. Bioresource Technology, 100(24), 6496–6504. https://doi.org/10.1016/j.biortech.2009.06.095
Shi, Q., Terracciano, A., Zhao, Y., Wei, C., Christodoulatos, C., & Meng, X. (2019). Evaluation ofmetal oxides and activated carbon for lead removal: Kinetics, isotherms, column tests, and the role of co-existing ions. Science of the Total Environment, 648, 176–183. https://doi.org/10.1016/j.scitotenv.2018.08.013
Su, Q., Pan, B., Pan, B., Zhang, Q., Zhang, W., Lv, L., & Wang, X. (2009). Fabrication of polymer-supported nanosized hydrous manganese dioxide (HMO) for enhanced lead removal from waters. Science of the Total Environment, 407, 5471–5477. https://doi.org/10.1016/j.scitotenv.2009.06.045
Sud, D., Mahajan, G., & Kaur, M. P. (2008). Agricultural waste material as potential adsorbent for sequestering heavy metal ions from aqueous solutions - A review. Bioresource Technology, 99(14), 6017–6027. https://doi.org/10.1016/j.biortech.2007.11.064
Suresh, S., Jayamoorthy, K., & Karthikeyan, S. (2016). Fluorescence sensing of potential NLO material by bunsenite NiO nanoflakes : Room temperature magnetic studies. Sensors & Actuators: B. Chemical. https://doi.org/10.1016/j.snb.2016.03.149
Tang, N., Niu, C., Li, X., Liang, C., Guo, H., Lin, L., & Zeng, G. (2018). Efficient removal of Cd2+ and Pb2+ from aqueous solution with amino- and thiol-functionalized activated carbon: Isotherm and kinetics modeling. Science of the Total Environment, 635, 1331–1344. https://doi.org/10.1016/j.scitotenv.2018.04.236
Tang, Q., Shi, C., Shi, W., Huang, X., Ye, Y., Jiang, W., & Li, D. (2019). Preferable phosphate removal by nano-La(III) hydroxides modified mesoporous rice husk biochars: Role of the host pore structure and point of zero charge. Science of the Total Environment, 662, 511–520. https://doi.org/10.1016/j.scitotenv.2019.01.159
Wan Ngah, W. S., & Hanafiah, M. A. K. M. (2008). Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: A review. Bioresource Technology, 99(10), 3935–3948. https://doi.org/10.1016/j.biortech.2007.06.011
Wu, M. S., & Hsieh, H. H. (2008). Nickel oxide/hydroxide nanoplatelets synthesized by chemical precipitation for electrochemical capacitors. Electrochimica Acta, 53(8), 3427–3435. https://doi.org/10.1016/j.electacta.2007.12.005
Xiang, L., Deng, X. Y., & Jin, Y. (2002). Experimental study on synthesis of NiO nano-particles. Scripta Materialia, 47(4), 219–224. https://doi.org/10.1016/S1359-6462(02)00108-2
Xing, W., Li, F., Yan, Z. F., & Lu, G. Q. (2004). Synthesis and electrochemical properties of mesoporous nickel oxide. Journal of Power Sources, 134(2), 324–330. https://doi.org/10.1016/j.jpowsour.2004.03.038
Yang, X., Yi, H., Tang, X., Zhao, S., Yang, Z., Ma, Y., & Cui, X. (2018). Behaviors and kinetics of toluene adsorption-desorption on activated carbons with varying pore structure. Journal of Environmental Sciences (china), 67, 104–114. https://doi.org/10.1016/j.jes.2017.06.032
Yao, S., Sun, S., Wang, S., & Shi, Z. (2016). Adsorptive removal of lead ion from aqueous solution by activated carbon/iron oxide magnetic composite. Indian Journal of Chemical Technology, 23(2), 146–152.
Yuan, G. H., Jiang, Z. H., Aramata, A., & Gao, Y. Z. (2005). Electrochemical behavior of activated-carbon capacitor material loaded with nickel oxide. Carbon, 43(14), 2913–2917. https://doi.org/10.1016/j.carbon.2005.06.027
Zachariades, C., Hoffmann, J. H., & Roberts, A. P. (2011). Biological control of mesquite ( Prosopis Species) (Fabaceae) in South Africa. African Entomology, 19(2), 402–415. https://doi.org/10.4001/003.019.0230
Zhang, X., Lin, Q., Luo, S., Ruan, K., & Peng, K. (2018). Preparation of novel oxidized mesoporous carbon with excellent adsorption performance for removal of malachite green and lead ion. Applied Surface Science, 442, 322–331. https://doi.org/10.1016/j.apsusc.2018.02.148
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saravanakumar, R., Muthukumaran, K., . Sivasankari, C. et al. Role of Purged Air in the Synthesis of the Mesoporous NiO/C Composite and Its Application in Wastewater Treatment. Water Air Soil Pollut 233, 53 (2022). https://doi.org/10.1007/s11270-022-05527-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05527-7