Abstract
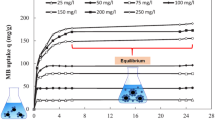
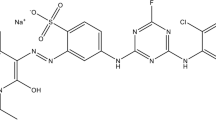
This study measures and evaluates the capabilities of desiccated coconut waste, a waste by-product produced from coconut milk processing, as an adsorbent precursor for the removal of azo dyes in wastewater. The results show that the desiccated coconut waste performed the highest adsorption capacity towards the Congo Red dye in pH 2 condition where the Qmax = 0.071 mmol/g. The adsorption capacity of the coconut waste-based adsorbent is relatively high in azo dye removal among the tested waste precursors such as coir pith, tamarind fruit shells, hazelnut shells, and banana peel. The analysis of azo dye adsorption equilibrium and kinetics using appropriate isotherm and kinetic model shows excellent fitting with Langmuir isotherm and pseudo-second-order kinetic model, where the determination coefficient, R2 ≈ 1, indicates a monolayer chemical adsorption process, with a maximum monolayer capacity of 0.004 mmol/g. These findings support that coconut waste-based adsorbent is highly potential for application of Congo Red removal in wastewater.







Similar content being viewed by others
Data availability (Data Transparency)
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability (Software Application or Custom Code)
Not applicable.
References
Abdul Rahim, A. R., Mohsin, H. M., Thanabalan, M., Rabat, N. E., Saman, N., Mat, H., & Johari, K. (2020). Effective carbonaceous desiccated coconut waste adsorbent for application of heavy metal uptakes by adsorption: Equilibrium, kinetic and thermodynamics analysis. Biomass and Bioenergy, 142(August), 105805. https://doi.org/10.1016/j.biombioe.2020.105805
Ahmad, N., Sereshti, H., Mousazadeh, M., Rashidi, H., Afzal, M., & Mohamad, S. (2019). New magnetic silica-based hybrid organic-inorganic nanocomposite for the removal of lead (II) and nickel (II) ions from aqueous solutions. Materials Chemistry and Physics, 226(July 2016), 73–81. https://doi.org/10.1016/j.matchemphys.2019.01.002
Al-Degs, Y. S., El-Barghouthi, M. I., El-Sheikh, A. H., & Walker, G. M. (2008). Effect of solution pH, ionic strength, and temperature on adsorption behavior of reactive dyes on activated carbon. Dyes and Pigments, 77(1), 16–23. https://doi.org/10.1016/j.dyepig.2007.03.001
Aljeboree, A. M., Alshirifi, A. N., & Alkaim, A. F. (2017). Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon. Arabian Journal of Chemistry, 10, S3381–S3393. https://doi.org/10.1016/j.arabjc.2014.01.020
Carletto, R. A., Chimirri, F., Bosco, F., & Ferrero, F. (2008). Adsorption of congo red dye on hazelnut shells and degradation with Phanerochaete chrysosporium. Adsorption Journal of the International Adsorption Society, 3, 1146–1155.
Chakraborty, J. N. (2011a). 14 - Sulphur dyes. In M. Clark (Ed.), Handbook of textile and industrial dyeing Vol. 1 (pp. 446–465). Woodhead Publishing.
Chakraborty, J. N. (2011b). Metal-complex dyes. In Handbook of textile and industrial dyeing: Principles, processes and types of dyes (Vol. 1). https://doi.org/10.1533/9780857093974.2.446
Chequer, F. M. D., Dorta, D. J., & de Oliveira, D. P. (2011). Azo dyes and their metabolites: does the discharge of the azo dye into water bodies represent human and ecological risks? Advances in Treating Textile Effluent:
de Farias, R. S., de Buarque, H. L., & B., da Cruz, M. R., Cardoso, L. M. F., Gondim, T. de A., & de Paulo, V. R. . (2018). Adsorption of congo red dye from aqueous solution onto amino-functionalized silica gel. Engenharia Sanitaria e Ambiental, 23(6), 1053–1060. https://doi.org/10.1590/s1413-41522018172982
El-Idreesy, T. T., Khoshala, O., Firouzi, A., & Elazab, H. A. (2021). Equilibrium and kinetic study on the biosorption of trypan blue from aqueous solutions using avocado seed powder. Biointerface Research in Applied Chemistry, 11(3), 11042–11053. https://doi.org/10.33263/BRIAC113.1104211053
Elkhider, K. H. A., Ihsanullah, I., Zubair, M., Manzar, M. S., & Mu’azu, N. D., & Al-Harthi, M. A. . (2020). Synthesis, characterization and dye adsorption performance of strontium ferrite decorated bentonite-CoNiAl magnetic composite. Arabian Journal for Science and Engineering, 45(9), 7397–7408. https://doi.org/10.1007/s13369-020-04544-0
Etim, U. J., Umoren, S. A., & Eduok, U. M. (2016). Coconut coir dust as a low cost adsorbent for the removal of cationic dye from aqueous solution. Journal of Saudi Chemical Society, 20, S67–S76. https://doi.org/10.1016/j.jscs.2012.09.014
Jabbar, Z., Angham, A., & Sami, G. H. F. (2014). Removal of azo dye from aqueous solutions using chitosan. Oriental Journal of Chemistry, 30(2).
Johari, K., Saman, N., Song, S. T., Mat, H., & Stuckey, D. C. (2013). Utilization of coconut milk processing waste as a low-cost mercury sorbent. Industrial and Engineering Chemistry Research, 52(44), 15648–15657. https://doi.org/10.1021/ie401470w
Johari, K., Saman, N., Tien, S. S., Chin, C. S., Kong, H., & Mat, H. (2016). Removal of elemental mercury by coconut pith char adsorbents. Procedia Engineering, 148, 1357–1362. https://doi.org/10.1016/j.proeng.2016.06.588
Khaniabadi, Y. O., Basiri, H., Nourmoradi, H., Mohammadi, M. J., Yari, A. R., Sadeghi, S., & Amrane, A. (2018). Adsorption of congo red dye from aqueous solutions by montmorillonite as a low-cost adsorbent. International Journal of Chemical Reactor Engineering, 16(1). https://doi.org/10.1515/ijcre-2016-0203
Langmuir, I. (1917). The constitution and fundamental properties of solids and liquids. Part II.-Liquids. Journal of the Franklin Institute, 184(5), 721. https://doi.org/10.1016/s0016-0032(17)90088-2
Lellis, B., Fávaro-Polonio, C. Z., Pamphile, J. A., & Polonio, J. C. (2019). Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnology Research and Innovation, 3(2), 275–290. https://doi.org/10.1016/j.biori.2019.09.001
M. Shah. (2018). Azo dye removal technologies. Austin Journal of Biotechnology & Bioengineering, 5(1).
Man, H. C., Akinbile, C. O., & Jun, C. X. (2015). Coconut husk adsorbent for the removal of methylene blue dye from wastewater. BioResources, 10(2), 2859–2872.
Manzar, M. S., Zubair, M., Khan, N. A., Husain Khan, A., Baig, U., Aziz, M. A., … Abdel-Magid, H. I. M. (2020). Adsorption behaviour of green coffee residues for decolourization of hazardous congo red and eriochrome black T dyes from aqueous solutions. International Journal of Environmental Analytical Chemistry, 00(00), 1–17https://doi.org/10.1080/03067319.2020.1811260
Mondal, N. K., & Kar, S. (2018). Potentiality of banana peel for removal of Congo red dye from aqueous solution: Isotherm, kinetics and thermodynamics studies. Applied Water Science, 8(6), 1–12. https://doi.org/10.1007/s13201-018-0811-x
Namasivayam, C., & Kavitha, D. (2002). Removal of Congo Red from water by adsorption onto activated carbon prepared from coir pith, an agricultural solid waste. 54, 47–58.
Okafor, P. C., Okon, P. U., Daniel, E. F., & Ebenso, E. E. (2012). Adsorption capacity of coconut (Cocos nucifera L.) shell for lead, copper, cadmium and arsenic from aqueous solutions. International Journal of Electrochemical Science, 7(12), 12354–12369.
Patel, B. H. (2011). 11 - Natural dyes. In M. Clark (Ed.), Handbook of textile and industrial dyeing (Vol. 1, pp. 395–424). Publishing.
Rahim, A. R. A., Ekmi, N., Johari, K., Saman, N., & Mat, H. (2019). Removal of lead (II) ions from aqueous solution using desiccated coconut waste as low-cost adsorbent. 7(December 2018), 169–174. https://doi.org/10.3303/CET1972029
Rahim, A. R. A., Johari, K., Shehzad, N., & Saman, N. (2021). Conversion of coconut waste into cost effective adsorbent for Cu ( II ) and Ni ( II ) removal from aqueous solutions. 0–1.
Rahim, A. R. A., Mohsin, H. M., Thanabalan, M., Rabat, N. E., Saman, N., Mat, H., & Johari, K. (2020). Effective carbonaceous desiccated coconut waste adsorbent for application of heavy metal uptakes by adsorption: Equilibrium, kinetic and thermodynamics analysis. Biomass & Bioenergy.
Sejie, P., & Nadiye-Tabbiruka, S. (2016). Removal of methyl orange (MO) from water by adsorption onto modified local clay (Kaolinite). Physical Chemistry, 6(2), 39–48. https://doi.org/10.5923/j.pc.20160602.02
Sekar, N. (2011a). 12 - Direct dyes. In M. Clark (Ed.), Handbook of textile and industrial dyeing Vol. 1. Woodhead Publishing.
Sekar, N. (2011b). 15 - Acid dyes. In M. Clark (Ed.), Handbook of textile and industrial dyeing Vol. 1 (pp. 486–514). Woodhead Publishing.
Singh, P. K., & Singh, R. L. (2017). Bio-removal of Azo dyes: A review. International Journal of Applied Sciences and Biotechnology, 5(2), 108–126.
Somasekhara Reddy, M. C. (2006). Removal of direct dye from aqueous solutions with an adsorbent made from tamarind fruit shell, an agricultural solid waste. Journal of Scientific and Industrial Research, 65(5), 443–446.
Song, C., Wu, S., Cheng, M., Tao, P., Shao, M., & Gao, G. (2014). Adsorption studies of coconut shell carbons prepared by KOH activation for removal of lead(ii) from aqueous solutions. Sustainability (switzerland), 6(1), 86–98. https://doi.org/10.3390/su6010086
Srebrenkoska, V., Zhezhova, Risteski, S., & Golomeova, S. (2014). Methods for waste waters treatment in textile industry.
Srivatsav, P., Bhargav, B. S., Shanmugasundaram, V., Arun, J., Gopinath, K. P., & Bhatnagar, A. (2020). Biochar as an eco-friendly and economical adsorbent for the removal of colorants (Dyes) from aqueous environment: A review. Water (switzerland), 12(12), 1–27. https://doi.org/10.3390/w12123561
Tejada-Tovar, C., Villabona-Ortiz, A., & Gonzalez-Delgado, A. D. (2021). Adsorption of azo-anionic dyes in a solution using modified coconut (Cocos nucifera) mesocarp: Kinetic and equilibrium study. Water, 13.
Wanyonyi, W. C., Onyari, J. M., & Shiundu, P. M. (2014). Adsorption of congo red dye from aqueous solutions using roots of eichhornia crassipes: Kinetic and equilibrium studies. Energy Procedia, 50, 862–869. https://doi.org/10.1016/j.egypro.2014.06.105
Zubair, M., Mu’azu, N. D., Jarrah, N., Blaisi, N. I., Aziz, H. A., & A. Al-Harthi, M. (2020). Adsorption behavior and mechanism of methylene blue, crystal violet, Eriochrome black T, and methyl orange dyes onto biochar-derived date palm fronds waste produced at different pyrolysis conditions. Water, Air, and Soil Pollution, 231(5). https://doi.org/10.1007/s11270-020-04595-x
Acknowledgements
The authors would like to thank the Department of Chemical Engineering, Universiti Teknologi PETRONAS, for providing the lab facilities for this project.
Author information
Authors and Affiliations
Contributions
Conceptualization: Abdul Rahman, Hanan, Khairiaihanna, Kausalya, investigation: Kausalya, methodology: Abdul Rahman, Khairiraihanna, supervision: Khairiraihanna, Norasikin, writing original draft: Abdul Rahman, Hanan, Kausalya, writing review and editing: Khairiraihanna, Norasikin.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdul Rahim, A.R., Mohsin, H.M., Chin, K.B.L. et al. Promising Low-cost Adsorbent from Desiccated Coconut Waste for Removal of Congo Red Dye from Aqueous Solution. Water Air Soil Pollut 232, 357 (2021). https://doi.org/10.1007/s11270-021-05308-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05308-8