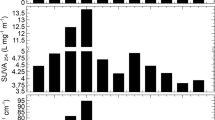
Abstract
Tropical peat water dissolved organic matter (DOM) fractions spectral parameters correlate with the formation of carcinogenic disinfection by-products (DBPs), haloacetic acids (HAAs). Peat water DOM has been fractionated using Superlite DAX-8, Amberlite XAD-4, and Amberlite IRA-958 resins to separate hydrophobic acid (HPOA), transphilic (TPH), hydrophilic-charged (HPIC), and hydrophilic-neutral (HPIN). The formation potential of HAA was determined using the total concentration of 5 HAAs (HAA5). The DOM characteristic was determined using dissolved organic carbon (DOC) and ultraviolet–visible (UV–Vis) absorbance was evaluated at 200–700 nm. The HPOA fraction dominates the DOM of tropical peat water with a 40% DOC concentration when the HPIC fraction was the slighter. The spectral absorbance ratios (E4/E6 and A253/A203) and spectral slopes (S206–213, S251–280, S281–295, and S>295) have a strong positive correlation with HAA5 formation potential (HAA5FP). Similarly, the absorbance ratio A210/A254 and spectral slopes S200–205 are strongly correlated to HAA5FP, but in a negative direction. In contrast, the E2/E3 and A280/A350 spectral ratio, and spectral slopes (S214-227, and S228–250), has a weaker correlation with HAA5FP. The E2/E3 and A280/A350 ratios and S251–280 S200–205 spectral slope show a strong relationship with the brominated HAA5FP (B-HAA5FP). The spectral parameters of DOM fractions can be used as surrogate parameters of HAA5FP of peat water.





Similar content being viewed by others
Data availability
Not applicable.
Code Availability
Not applicable.
References
APHA, A. P. H. A. (1998). Standard methods for the examination of water and wastewater. American Physical Education Review, 24(9), 481–486.
Asgari, G., Mohammadi, A. S., & Ebrahimi, A. (2012). Performance of the catalytic ozonation process with pumice in removal of humic acids from aqueous solutions. International Journal of Environmental Health Engineering, 1(3), 1–7. https://doi.org/10.4103/2277-9183.99327
Bing-zhi, D., Yan, C., Nai-yun, G., & Jin-chu, F. (2007). Effect of coagulation pretreatment on the fouling of ultrafiltration membrane. Journal of Environmental Sciences, 19(May 2006), 278–283. https://doi.org/10.1016/S1001-0742(07)60045-X
Bond, T., Goslan, E. H., Parsons, S. A., & Jefferson, B. (2012). A critical review of trihalomethane and haloacetic acid formation from natural organic matter surrogates. Environmental Technology Reviews, 1(1), 93–113. https://doi.org/10.1080/09593330.2012.705895
Chaukura, N., Ndlangamandla, N. G., Moyo, W., Msagati, T. A. M., Mamba, B. B., & Nkambule, T. T. I. (2018). Natural organic matter in aquatic systems – a South African perspective. Water S.A, 44(4), 624–635. https://doi.org/10.4314/wsa.v44i4.11
Chen, Y., Senesi, N., & Schnitzer, M. (1976). Information provided on humic substances by E4/E6 ratios. Soil Science Society of America Journal, 606, 352–358. https://doi.org/10.2136/sssaj1977.03615995004100020037x
Dickenson, E. R. V., Summers, R. S., Croué, J., & Gallard, H. (2008). Haloacetic acid and trihalomethane formation from the chlorination and bromination of aliphatic -dicarbonyl acid model compounds. Environmental Science and Technology, 42(9), 3226–3233. https://doi.org/10.1021/es0711866
Edzwald, J. K., & Tobiason, J. E. (1999). Enhanced coagulation: US requirements and a broader view. Water Science and Technology, 40(9), 63–70. https://doi.org/10.1016/S0273-1223(99)00641-1
Feng, H., Ruan, Y., Wu, R., Zhang, H., & Lam, P. K. S. (2019). Occurrence of disinfection by-products in sewage treatment plants and the marine environment in Hong Kong. Ecotoxicology and Environmental Safety, 181, 404–411. https://doi.org/10.1016/j.ecoenv.2019.06.034
Gough, R., Holliman, P. J., Cooke, G. M., & Freeman, C. (2015). Characterisation of algogenic organic matter during an algal bloom and its implications for trihalomethane formation. Sustainability of Water Quality and Ecology, 6, 11–19. https://doi.org/10.1016/j.swaqe.2014.12.008
He, K., Okuta, E., Cordero, J. A., Echigo, S., Asada, Y., & Itoh, S. (2018). Formation of chlorinated haloacetic acids by chlorination of low molecular weight compounds listed on pollutant release and transfer registers (PRTRs). Journal of Hazardous Materials, 351(2010), 98–107. https://doi.org/10.1016/j.jhazmat.2018.02.028
Helms, J. R., Stubbins, A., Ritchie, J. D., Minor, E. C., Kieber, D. J., & Mopper, K. (2008). Absorption spectral slopes and slope ratios as indicators of molecular weight, source, and photobleaching of chromophoric dissolved organic matter. Limnology and Oceanography, 53(3), 955–969.
Her, N., Amy, G., Sohn, J., & von Gunten, U. (2008). UV absorbance ratio index with size exclusion chromatography (URI-SEC) as an NOM property indicator. Journal of Water Supply: Research and Technology-Aqua, 57(1), 35–44. https://doi.org/10.2166/aqua.2008.029
Hu, Y., Qian, Y., Chen, Y., Guo, J., Song, J., & An, D. (2021). Characteristics of trihalomethane and haloacetic acid precursors in filter backwash and sedimentation sludge waters during drinking water treatment. Science of the Total Environment, 775, 145952. https://doi.org/10.1016/j.scitotenv.2021.145952
Hua, G., & Reckhow, D. A. (2007). Characterization of disinfection by-product precursors based on hydrophobicity and molecular size. Environmental Science and Technology, 41, 3309–3315.
Hua, G., & Reckhow, D. A. (2013). Effect of pre-ozonation on the formation and speciation of DBPs. Water Research, 47(13), 4322–4330. https://doi.org/10.1016/j.watres.2013.04.057
Kanokkantapong, V., Marhaba, T. F., Pavasant, P., & Panyapinyophol, B. (2006). Characterization of haloacetic acid precursors in source water. Journal of Environmental Management, 80(3), 214–221. https://doi.org/10.1016/j.jenvman.2005.09.006
Korshin, G., Chow, C. W. K., Fabris, R., & Drikas, M. (2009). Absorbance spectroscopy-based examination of effects of coagulation on the reactivity of fractions of natural organic matter with varying apparent molecular weights. Water Research, 43(6), 1541–1548. https://doi.org/10.1016/j.watres.2008.12.041
Korshin, G. V., Li, C., & Benjamin, M. M. (1997). Monitoring the properties of natural organic matter through UV spectroscopy: A consistent theory. Water Research, 31(7), 1787–1795. https://doi.org/10.1016/S0043-1354(97)00006-7
Kuokkanen, V., Kuokkanen, T., Rämö, J., & Lassi, U. (2015). Electrocoagulation treatment of peat bog drainage water containing humic substances. Water Research, 79, 79–87. https://doi.org/10.1016/j.watres.2015.04.029
Liu, S., Benedetti, M. F., Han, W., & Korshin, G. V. (2020). Comparison of the properties of standard soil and aquatic fulvic and humic acids based on the data of differential absorbance and fl uorescence spectroscopy. Chemosphere, 261, 128189. https://doi.org/10.1016/j.chemosphere.2020.128189
Lu, J., Zhang, T., Ma, J., & Chen, Z. (2009). Evaluation of disinfection by-products formation during chlorination and chloramination of dissolved natural organic matter fractions isolated from a filtered river water. Journal of Hazardous Materials, 162(1), 140–145. https://doi.org/10.1016/j.jhazmat.2008.05.058
Mahmud, Abdi, C., & Mu’min, B. (2013). Removal natural organic matter (NOM) in peat water from wetland area by coagulation-ultrafiltration hybrid process with pretreatment two-stage coagulation. Journal of Wetlands Environmental Management, 1(1), 42–49. https://doi.org/10.20527/jwem.v1i1.88
Marhaba, T. F., & Van, D. (2000). The variation of mass and disinfection by-product formation potential of dissolved organic matter fractions along a conventional surface water treatment plant. Journal of Hazardous Materials, 74(3), 133–147. https://doi.org/10.1016/S0304-3894(99)00190-9
Moyo, W., Chaukura, N., Msagati, T. A. M., & Mamba, B. B. (2019). The properties and removal efficacies of natural organic matter fractions by South African drinking water treatment plants. Journal of Environmental Chemical Engineering, 7(3), 103101. https://doi.org/10.1016/j.jece.2019.103101
Navalon, S., Alvaro, M., Alcaina, I., & Garcia, H. (2010). Multi-method characterization of DOM from the Turia river (Spain). Applied Geochemistry, 25(11), 1632–1643. https://doi.org/10.1016/j.apgeochem.2010.08.011
Notodarmojo, S., Mahmud, & Larasati, A. (2017). Adsorption of natural organic matter (NOM) in peat water by local Indonesia tropical clay soils. International Journal of GEOMATE, 13(38), 111–119. https://doi.org/10.21660/2017.38.30379
Peuravuori, J., & Pihlaja, K. (1997). Molecular size distribution and spectroscopic properties of aquatic humic substances. Analytica Chimica Acta, 337, 133–149. https://doi.org/10.1016/S0003-2670(96)00412-6
Postigo, C., Emiliano, P., Barceló, D., & Valero, F. (2018). Chemical characterization and relative toxicity assessment of disinfection byproduct mixtures in a large drinking water supply network. Journal of Hazardous Materials, 359, 166–173. https://doi.org/10.1016/j.jhazmat.2018.07.022
Qadafi, M., Notodarmojo, S., & Zevi, Y. (2021). Performance of microbubble ozonation on treated tropical peat water: Effects on THM4 and HAA5 precursor formation based on DOM hydrophobicity fractions. Chemosphere, 279, 130642. https://doi.org/10.1016/j.chemosphere.2021.130642
Qadafi, M., Notodarmojo, S., Zevi, Y., & Maulana, Y. E. (2020). Trihalomethane and haloacetic acid formation potential of tropical peat water: Effect of tidal and seasonal variations. International Journal of GEOMATE, 18(66), 111–117. https://doi.org/10.21660/2020.66.9487
Raghunandan, M. E., & Sriraam, A. S. (2017). An overview of the basic engineering properties of Malaysian peats. Geoderma Regional, 11(February), 1–7. https://doi.org/10.1016/j.geodrs.2017.08.003
Ritson, J. P., Bell, M., Graham, N. J. D., Templeton, M. R., Brazier, R. E., Verhoef, A., et al. (2014). Simulated climate change impact on summer dissolved organic carbon release from peat and surface vegetation: Implications for drinking water treatment. Water Research, 67, 66–76. https://doi.org/10.1016/j.watres.2014.09.015
Siddique, A., Saied, S., Mumtaz, M., Hussain, M. M., & Khwaja, H. A. (2015). Multipathways human health risk assessment of trihalomethane ex- posure through drinking water. Ecotoxicology and Environmental Safety, 116, 129–136. https://doi.org/10.1016/j.ecoenv.2015.03.011
Sillanpää, M., Ncibi, M. C., & Matilainen, A. (2018). Advanced oxidation processes for the removal of natural organic matter from drinking water sources: A comprehensive review. Journal of Environmental Management, 208, 56–76. https://doi.org/10.1016/j.jenvman.2017.12.009
Sururi, M. R., Notodarmojo, S., & Roosmini, D. (2019). Aquatic organic matter characteristics and THMFP occurrence in a tropical river. International Journal of GEOMATE, 17(62), 203–211. https://doi.org/10.21660/2019.62.85393
Tada, Y., Cordero, J. A., Echigo, S., & Itoh, S. (2021). Effect of coexisting manganese ion on the formation of haloacetic acids during chlorination. Chemosphere, 263, 127862. https://doi.org/10.1016/j.chemosphere.2020.127862
Twardowski, M. S., Boss, E., Sullivan, J. M., & Donaghay, P. L. (2004). Modeling the spectral shape of absorption by chromophoric dissolved organic matter. Marine Chemistry, 89, 69–88. https://doi.org/10.1016/j.marchem.2004.02.008
USEPA. (1995). Method 552.2: Determination of haloacetic acids and dalapon in drinking water by liquid-liquid extraction, derivatization and gas chromatography with electron capture detection. National exposure research laboratory office of research and development U.S. Environmental protection agency, 1–32.
USEPA. (1998). Final rule. Federal Register, 63(241), 69390–69476. https://www.federalregister.gov/documents/1998/12/16/98-32887/national-primary-drinking-water-regulations-disinfectants-and-disinfection-byproducts. Accessed 11/06/2020
USEPA. (2003). Method 415.3 determination of total organic carbon and specific UV absorbance at 254 nm in source water and drinking water. National exposure research laboratory office of research and development U.S. Environmental protection agency, 28(1), 17–59. https://doi.org/10.35362/rie280958
Vergnoux, A., Rocco, R. D., Domeizel, M., Guiliano, M., Doumenq, P., & Théraulaz, F. (2011). Effects of forest fires on water extractable organic matter and humic substances from Mediterranean soils: UV–vis and fl uorescence spectroscopy approaches. Geoderma, 160(3–4), 434–443. https://doi.org/10.1016/j.geoderma.2010.10.014
Vieira, R. F., Berenguel, A. T., Silva, M. A., Vilaça, J. S., Domingues, V. F., & Figueiredo, S. A. (2012). Natural organic matter fractionation along the treatment of water for human consumption. Global Nest Journal, 14(4), 399–406. https://doi.org/10.30955/gnj.000828
Wang, Y., Zhu, G., & Engel, B. (2019). Health risk assessment of trihalomethanes in water treatment plants in Jiangsu Province , China. Ecotoxicology and Environmental Safety, 170(December 2018), 346–354. https://doi.org/10.1016/j.ecoenv.2018.12.004
Xie, Y. (2001). Analyzing haloacetic acids using gas chromatography/mass spectrometry. Water Research, 35(6), 1599–1602. https://doi.org/10.1016/S0043-1354(00)00397-3
Yan, M., Korshin, G., Wang, D., & Cai, Z. (2012). Characterization of dissolved organic matter using high-performance liquid chromatography ( HPLC )– size exclusion chromatography ( SEC ) with a multiple wavelength absorbance detector. Chemosphere, 87(8), 879–885. https://doi.org/10.1016/j.chemosphere.2012.01.029
Yu, H. W., Oh, S. G., Kim, I. S., Pepper, I., Snyder, S., & Jang, A. (2015). Formation and speciation of haloacetic acids in seawater desalination using chlorine dioxide as disinfectant. Journal of Industrial and Engineering Chemistry, 26, 193–201. https://doi.org/10.1016/j.jiec.2014.10.046
Zhang, C., Han, X., Korshin, G. V., Kuznetsov, A. M., & Yan, M. (2021a). Interpretation of the differential UV-visible absorbance spectra of metal-NOM complexes based on the quantum chemical simulations for the model compound esculetin. Chemosphere, 276, 130043. https://doi.org/10.1016/j.chemosphere.2021.130043
Zhang, C., Roccaro, P., Yan, M., & Korshin, G. V. (2021b). Interpretation of the formation of unstable halogen-containing disinfection by-products based on the differential absorbance spectroscopy approach. Chemosphere, 268, 129241. https://doi.org/10.1016/j.chemosphere.2020.129241
Zhang, H., Qu, J., Liu, H., & Zhao, X. (2009). Characterization of isolated fractions of dissolved organic matter from sewage treatment plant and the related disinfection by-products formation potential. Journal of Hazardous Materials, 164(2–3), 1433–1438. https://doi.org/10.1016/j.jhazmat.2008.09.057
Zhang, M., & He, Z. (2015). Characteristics of dissolved organic carbon revealed by ultraviolet – visible absorbance and fluorescence spectroscopy : The current status and future exploration. In SSSA Special Publications (Vol. 62, pp. 1–22). https://doi.org/10.2136/sssaspecpub62.2014.0032
Zhong, X., Cui, C., & Yu, S. (2017a). Formation of aldehydes and carboxylic acids in humic acid ozonation. Water, Air, and Soil Pollution, 228(6), 1–11. https://doi.org/10.1007/s11270-017-3418-1
Zhong, X., Cui, C., & Yu, S. (2017b). The determination and fate of disinfection by-products from ozonation-chlorination of fulvic acid. Environmental Science and Pollution Research, 24(7), 6472–6480. https://doi.org/10.1007/s11356-016-8350-1
Acknowledgements
The authors express gratitude to the Integrated Laboratory, Poltekkes Kemenkes Bandung, for providing a laboratory analysis facility.
Funding
This research was funded by The Indonesia Endowment Fund for Education (Lembaga Pengelola Dana Pendidikan/LPDP), Ministry of Finance Indonesia, Grant No. 201705210110920.
Author information
Authors and Affiliations
Contributions
Muammar Qadafi: conceptualization, methodology, investigation, data analysis, writing—original draft, project administration.
Suprihanto Notodarmojo: Supervision, conceptualization.
Yuniati Zevi: Supervision, validation, investigation.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Qadafi, M., Notodarmojo, S. & Zevi, Y. Haloacetic Acids Formation Potential of Tropical Peat Water DOM Fractions and Its Correlation with Spectral Parameters. Water Air Soil Pollut 232, 319 (2021). https://doi.org/10.1007/s11270-021-05271-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05271-4