Abstract
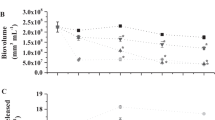
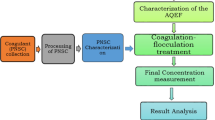
Cyanobacteria are microorganisms capable of releasing toxic metabolites in freshwater, deleterious to humans and other living organisms even in very low concentrations. Removing both cyanobacteria cells and their metabolites in conventional water treatment systems is still a challenge that needs to be addressed. This paper evaluated the use of Moringa oleifera saline extract and polyaluminum chloride (PACl) as coagulants in order to remove cells and metabolites of M. aeruginosa from water. Samples consisted of synthetic water spiked with humic acid and M. aeruginosa cells, with a final turbidity of 25 NTU. Coagulation/flocculation/dissolved air flotation (C/D/DAF) tests were performed with 50 mg/L of M. oleifera coagulant, combined with different proportions of PACl (10–50%). For removal of metabolites, 25 μg/L of microcystin and 50 ng/L of 2-MIB and geosmin were spiked in water samples. The best results were obtained with 70:30 M. oleifera:PACl, achieving removal efficiencies of 81.37%, 74.69%, and 71.06% for turbidity, color, and cell density, respectively. For microcystin, 2-MIB, and geosmin, after filtration (30 min), global removal efficiencies (clarification + filtration) of 99.45%, 62.37%, and 100%, respectively, were obtained, using Sand+GAC filter. The results suggest that the substitution of PACl by M. oleifera extract can be a good option to remove cyanobacteria cells from water, and high removal efficiencies of metabolites can be achieved after the complete proposed treatment.






Similar content being viewed by others
References
Abdulazeez, Q. M., Jami, M. S., & Alam, M. D. Z. (2016). Effective sludge dewatering using Moringa oleifera seed extract combined with aluminium sulfate. ARPN J. Eng. Appl. Sci., 11, 372–381.
Ali Ger, K., Faassen, E. J., Pennino, M. G., & Lurling, M. (2016). Effect of the toxin (microcystin) content of Microcystis on copepod grazing. Harmful Algae, 52, 34–45. https://doi.org/10.1016/j.hal.2015.12.008.
Almeida, C. R., Spiandorello, F. B., Giroldo, D., & Yunes, J. S. (2016). The effectiveness of conventional water treatment in removing Ceratium furcoides (Levander) Langhans. Microcystis sp. and microcystins. Water SA, 42, 606–611. https://doi.org/10.4314/wsa.v42i4.11.
APHA/WEF, Standard methods for the examination of water and wastewater, 22nd ed., American Public Health, Washington D.C., 2012
AWWA. (1999). American Water Works Association (ed.), Water quality and treatment: a handbook of community water supplies, 5 ed. USA: McGraw-Hill.
Basra, S. M. A., Iqbal, Z., Khalil-ur-Rehman, Hafeez-Ur-Rehman, & Ejaz, M. F. (2014). Time course changes in pH, electrical conductivity and heavy metals (Pb, Cr) of wastewater using Moringa oleifera Lam. seed and alum, a comparative evaluation. J. Appl. Res. Technol, 12, 560–567. https://doi.org/10.1016/S1665-6423(14)71635-9.
Beltrán-Heredia, J., & Sánchez-Martín, J. (2009). Removal of sodium lauryl sulphate by coagulation/flocculation with Moringa oleifera seed extract. J. Hazard. Mater., 164, 713–719. https://doi.org/10.1016/j.jhazmat.2008.08.053.
Beltrán-Heredia, J., Sánchez-Martín, J., & Barrado-Moreno, M. (2012). Long-chain anionic surfactants in aqueous solution. Removal by Moringa oleifera coagulant. Chem. Eng J., 180, 128–136. https://doi.org/10.1016/j.cej.2011.11.024.
Bhatti, Z. A., Mahmood, Q., & Raja, I. A. (2009). Sewage water pollutants removal efficiency correlates to the concentration gradient of amendments. J. Chem. Soc. Pak., 31, 665–673.
Bondelind, M., Sasic, S., & Bergdahl, L. (2013). A model to estimate the size of aggregates formed in a dissolved air flotation unit. Appl. Math. Model., 37, 3036–3047. https://doi.org/10.1016/j.apm.2012.07.004.
Bradford, M. M. A. (1976). Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72, 248–254. https://doi.org/10.1016/0003-2697(76)90527-3.
Brasil. Ministério da Saúde. Portaria de Consolidação N° 5, de 28 de setembro de 2017. Dispõe sobre a Consolidação das normas sobre as ações e os serviços de saúde do Sistema Único de Saúde. Diário Oficial da União N° 190, República Federativa do Brasil, Brasília, DF, 03 out. 2017. Seção 1 – Suplemento – p. 360.
Camacho, F. P., Bongiovani, M. C., Arakawa, F. S., Shimabuku, Q. L., Vieira, A. M. S., & Bergamasco, R. (2013). Advanced processes of cyanobacteria and cyanotoxins removal in supply water treatment. Chem. Engineer. Trans., 32, 421–426. https://doi.org/10.3303/CET1332071.
Carvalho, M. S., Alves, B. R. R., Silva, M. F., Bergamasco, R., Coral, L. A., & Bassetti, F. (2016). CaCl2 applied to the extraction of Moringa oleifera seeds and the use for Microcystis aeruginosa removal. Chem. Eng. J., 304, 469–475. https://doi.org/10.1016/j.cej.2016.06.101.
Centurione Filho, P. L., & Di Bernardo, L. (2003). Procedures for benchscale flotation/filtration assays (in Portuguese: Procedimento para execução de ensaios de flotação/filtração em equipamento de bancada). Eng. Sanit. Ambient., 8, 39–44.
Chorus, I.; Bartram, J. (Ed.). Toxic cyanobacteria in water: a guide to their public health consequences, monitoring and management. E & FN SPON: London and New York, 1999.
Choy, S. Y., Prasad, K. M. N., Wu, T. Y., Raghunandan, M. E., & Ramanan, R. N. (2014). Utilization of plant-based natural coagulants as future alternatives towards sustainable water clarification. J. Environ. Sci., 26, 2178–2189. https://doi.org/10.1016/j.jes.2014.09.024.
Coelho, E. R. C., & Di Bernardo, L. (2012). Removal of atrazine and metabolites through slow filtration by sand and granular activated carbon in Portuguese: Remoção de atrazine e metabólitos pela filtração lenta com leito de areia e carvão ativado granular. Eng. Sanit. Ambient., 17, 269–276. https://doi.org/10.1590/S1413-41522012000300003.
Cook, D., & Newcombe, G. (2004). Can we predict the removal of MIB and geosmin with PAC by using water quality parameters? Water Sci. Technol.: Water Supply, 4, 221–226. https://doi.org/10.2166/ws.2004.0081.
Cook, D., Newcombe, G., & Sztajnbok, P. (2001). The application of powdered activated carbon for 2-MIB and geosmin removal: predicting PAC doses in four raw waters. Water Res., 35, 1325–1333. https://doi.org/10.1016/S0043-1354(00)00363-8.
Drikas, M., Chow, C. W. K., House, J., & Burch, M. D. (2001). Using coagulation, flocculation and settling to remove toxic cyanobacteria. J. Am. Water Works Ass., 93, 100–111. https://doi.org/10.1002/j.1551-8833.2001.tb09130.x.
Dugan, N. R., & Williams, D. J. (2006). Cyanobacteria passage through drinking water filters during perturbation episodes as a function of cell morphology, coagulant and initial filter loading rate. Harmful Algae, 5, 26–35. https://doi.org/10.1016/j.hal.2005.04.001.
EPA – United States Environmental Protection Agency. National Primary Drinking Water Regulations, EPA 816-F-09-004, Office of Ground Water and Drinking Water, Washington D.C. 2009.
Feria-Díaz, J. J., Polo-Corrales, L., & Hernandez-Ramos, E. J. (2016). Evaluation of coagulation sludge from raw water treated with Moringa oleifera for agricultural use. Ing. Invest, 36, 14–20. https://doi.org/10.15446/ing.investig.v36n2.56986.
Ferreira Filho, S. S., & Waelkens, B. E. (2009). Minimization of water treatment plant sludge production with polyaluminium chloride application and its disposal in wastewater treatment plants. (in Portuguese: Minimização da produção de lodo no tratamento de águas de abastecimento mediante uso do cloreto de polialumínio e sua disposição em estações de tratamento de esgotos). Eng. Sanit. Ambient., 14, 317–326. https://doi.org/10.1590/S1413-41522009000300005.
García-Fayos, B., Arnal, J. M., Monforte-Monleon, L., & Sancho, M. (2015). Alternatives to the use of synthetic organic coagulant aids in drinking water treatment: improvements in the application of the crude extract of Moringa oleifera seed. Desalin. Water Treat., 55, 3635–3645. https://doi.org/10.1080/19443994.2014.939487.
García-Villada, L., Rico, M., Altamirano, M., Sánchez-Martín, L., López-Rodas, V., & Costas, E. (2004). Occurrence of copper resistant mutants in the toxic cyanobacteria Microcystis aeruginosa: characterisation and future implications in the use of copper sulphate as algaecide. Water Res., 38, 2207–2213. https://doi.org/10.1016/j.watres.2004.01.036.
Ghebremichael, K. A., & Hultman, B. (2004). Alum sludge dewatering using Moringa oleifera as a conditioner. Water Air Soil Poll., 158, 153–167. https://doi.org/10.1023/B:WATE.0000044852.23764.9c.
Hafuka, A., & Nagasato, H. (2019). Yamamura, Application of graphene oxide for adsorption removal of geosmin and 2-Methylisoborneol in the presence of natural organic matter. Int. J. Envir. Res. Public Health, 16, 1–8. https://doi.org/10.3390/ijerph16111907.
Hamid, S. H. A., Lananan, F., Din, W. N. S., Lam, S. S., Khatoon, H., Endut, A., & Jusoh, A. (2014). Harvesting microalgae, Chlorella sp. by bio-flocculation of Moringa oleifera seed derivatives from aquaculture wastewater phytoremediation. Int. Biodeter. Biodegr, 95, 270–275. https://doi.org/10.1016/j.ibiod.2014.06.021.
He, X., Liu, Y., Conklin, A., Westrick, J., Weavers, L. K., Dionysiou, D. D., & Walker, H. W. (2016). Toxic cyanobacteria and drinking water: impacts, detection, and treatment. Harmful Algae, 54, 174–193. https://doi.org/10.1016/j.hal.2016.01.001.
He, L., Huang, Y., Guo, Q., Zeng, H., Zheng, C., Wang, J., Chen, J.-A., Wang, L., & Shu, W. (2018). Chronic microcystin-LR exposure induces hepatocarcinogenesis via increased Gankyrin in vitro and in vivo. Cell. Physiol. Biochem., 49, 1420–1430. https://doi.org/10.1159/000493446.
Health Canada. (2019). Guidelines for Canadian Drinking Water Quality — Summary Table. Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario: Water and Air Quality Bureau.
Henderson, R. K., Parsons, S. A., & Jefferson, B. (2008a). Successful removal of algae through the control of zeta potential. Sep. Sci. Technol., 43, 1653–1666. https://doi.org/10.1080/01496390801973771.
Henderson, R., Chips, M., Cornwell, N., Hitchins, P., Holden, B., Hurley, S., Parsons, S. A., Wetherill, A., & Jefferson, B. (2008b). Experiences of algae in UK waters: a treatment perspective. Water Environ. J., 22, 184–192. https://doi.org/10.1111/j.1747-6593.2007.00100.x.
Hoeger, S., Hitzfeld, B. C., & Dietrich, D. R. (2005). Occurrence and elimination of cyanobacterial toxins in drinking water treatment plants. Toxicol. Appl. Pharm., 203, 231–242. https://doi.org/10.1016/j.taap.2004.04.015.
Huang, W., Cheng, B., & Cheng, Y. (2007). Adsorption of microcystin-LR by three types of activated carbon. J. Hazard. Mater., 141, 115–122. https://doi.org/10.1016/j.jhazmat.2006.06.122.
Jahn, S. A. A. (1988). Using Moringa oleifera seeds as coagulant in developing countries. J. Am. Water Works Ass., 6, 43–50. https://doi.org/10.1002/j.1551-8833.1988.tb03052.x.
Jerri, H. A., Adolfsen, K. J., McCullough, L. R., Velegol, D., & Velegol, S. B. (2012). Antimicrobial sand via adsorption of cationic Moringa oleifera protein. Langmuir, 28, 2262–2268. https://doi.org/10.1021/la2038262.
Konradt-Moraes, L. C., Bergamasco, R., Tavares, C. R. G., Hennig, D., & Bongiovani, M. C. (2008). Utilization of the coagulation diagram in the evaluation of the natural organic matter (NOM) removal for obtaining potable water. Int. J. Chem. React. Eng., 6, A87. https://doi.org/10.2202/1542-6580.1441.
Kumar, K., & Balasundaram, N. (2017). Efficiency of PAC in water treatment plant & disposal of its sludge. Int. J. Appl. Eng. Res., 12, 3253–3262.
Kumari, P., Srivastava, S., & Srivastava, M. (2006). Biosorption studies on shelled Moringa oleifera Lamarck powder: Removal and recovery of arsenic from aqueous system. Int. J. Miner. Process., 78, 131–139. https://doi.org/10.1016/j.minpro.2005.10.001.
Li, X., Zhao, Q., Zhou, W., Xu, L., & Wang, Y. (2015). Effects of chronic exposure to microcystin-LR on hepatocyte mitochondrial DNA replication in mice. Environ. Sci. Technol., 49, 4665–4672. https://doi.org/10.1021/es5059132.
Luz, L. A., Santos, A. F. S., Pontual, E. V., Napoleão, T. H., Paiva, P. M. G., & Coelho, L. C. B. B. (2015). Moringa oleifera: resource management and multiuse life tree. Adv. Res., 4, 388–402. https://doi.org/10.9734/AIR/2015/18177.
Ma, X., Gao, N., Chen, B., Li, Q., Zhang, Q., & Gu, G. (2007). Detection of geosmin and 2-methylisoborneol by liquid-liquid extraction-gas chromatograph mass spectrum (LLEGCMS) and solid phase extraction-gas chromatograph mass spectrum (SPEGCMS). Front. Environ. Sci. Engin. China, 1, 286–291. https://doi.org/10.1007/s11783-007-0048-7.
Ma, C., Xu, H., Zhang, L., Pei, H., & Jin, Y. (2018). Use of fluorescence excitation–emission matrices coupled with parallel factor analysis to monitor C- and N-DBPs formation in drinking water recovered from cyanobacteria-laden sludge dewatering. Sci. Total Environ., 640-641, 609–618. https://doi.org/10.1016/j.scitotenv.2018.05.369.
Madrona, G. S., Serpelloni, G. B., Vieira, A. M. S., Nishi, L., Cardoso, K. C., & Bergamasco, R. (2010). Study of the effect of saline solution on the extraction of the Moringa oleifera seed´s active component for water treatment. Water Air Soil Poll., 211, 409–415. https://doi.org/10.1007/s11270-009-0309-0.
Matsui, Y., Yoshida, T., Nakao, S., Knappe, D. R. U., & Matsushita, T. (2012). Characteristics of competitive adsorption between 2-methylisoborneol and natural organic matter on superfine and conventionally sized powdered activated carbons. Water Res., 46, 4741–4789. https://doi.org/10.1016/j.watres.2012.06.002.
Matsui, Y., Nakao, S., Sakamoto, A., Taniguchi, T., Pan, L., Matsushita, T., & Shirasaki, N. (2015). Adsorption capacities of activated carbons for geosmin and 2-methylisoborneol vary with activated carbon particle size: effects of adsorbent and adsorbate characteristics. Water Res., 85, 95–102. https://doi.org/10.1016/j.watres.2015.08.017.
Ndabigengesere, A., & Narasiah, K. S. (1998). Quality of water treated by coagulation using Moringa oleifera seeds. Water Res., 32, 781–791. https://doi.org/10.1016/S0043-1354(97)00295-9.
Ndabigengesere, A., Narasiah, K. S., & Talbot, B. G. (1995). Active agents and mechanism of coagulation of turbid water using Moringa oleifera. Water Res., 29, 703–710. https://doi.org/10.1016/0043-1354(94)00161-Y.
Nishi, L., Madrona, G. S., Falavigna-Guilherme, A. L., Vieira, A. M. S., Araujo, A. A., Ambrosio-Ugri, M., & Bergamasco, R. (2011). Cyanobacteria removal by coagulation/flocculation with seeds of the natural coagulant Moringa oleifera Lam. Chem. Engineer. Trans., 24, 1129–1134. https://doi.org/10.3303/CET1124189.
Nkurunziza, T., Nduwayezu, J. B., Banadda, E. N., & Nhapi, I. (2009). The effect of turbidity levels and Moringa oleifera concentration on the effectiveness of coagulation in water treatment. Water Sci. Technol., 59, 1551–1558. https://doi.org/10.2166/wst.2009.155.
Obuseng, V., Nareetsile, F., & Kwaambwa, H. M. (2012). A study of the removal of heavy metals from aqueous solutions by Moringa oleifera seeds and amine-based ligand 1,4-bis[N,N-bis(2-picoyl)amino]butane. Anal. Chim. Acta, 730, 87–92. https://doi.org/10.1016/j.aca.2012.01.054.
Okuda, T., Baes, A. U., Nishijima, W., & Okada, M. (2001). Coagulation mechanism of salt solution extracted active component in Moringa oleifera seeds. Water Res., 35, 830–834. https://doi.org/10.1016/S0043-1354(00)00296-7.
Ometto, F., Pozza, C., Whitton, R., Smyth, B., Torres, A. G., Henderson, R. K., Jarvis, P., Jefferson, B., & Villa, R. (2014). The impacts of replacing air bubbles with microspheres for the clarification of algae from low cell-density culture. Water Res., 53, 168–179. https://doi.org/10.1016/j.watres.2014.01.012.
Pantelic, D., Svircev, Z., Simeunovic, J., Vidovic, M., & Trajkovic, I. (2013). Cyanotoxins: characteristics, production and degradation routes in drinking water treatment with reference to the situation in Serbia. Chemosphere, 91, 421–441. https://doi.org/10.1016/j.chemosphere.2013.01.003.
Paula, H. M., Ilha, M. S. O., & Andrade, L. S. (2014). Concrete plant wastewater treatment process by coagulation combining aluminum sulfate and Moringa oleifera powder. J. Clean. Prod., 76, 125–130. https://doi.org/10.1016/j.jclepro.2014.04.031.
Petersen, H. H., Petersen, T. B., Enemark, H. L., Olsen, A., & Dalsgaard, A. (2016). Removal of Cryptosporidium parvum oocysts in low quality water using Moringa oleifera seed extract as coagulant. Food and Waterborne Parasitol., 3, 1–8. https://doi.org/10.1016/j.fawpar.2016.03.002.
Pivokonsky, M., Kloucek, O., & Pivokonska, L. (2006). Evaluation of the production, composition and aluminum and iron complexation of algogenic organic matter. Water Res., 40, 3045–3052. https://doi.org/10.1016/j.watres.2006.06.028.
Pritchard, M., Craven, T., Mkandawire, T., Edmondson, A. S., & O’neill, J. G. (2010). A comparison between Moringa oleifera and chemical coagulants in the purification of drinking water – an alternative sustainable solution for developing countries. Phys. Chem. Earth Parts A/B/C, 35, 798–805. https://doi.org/10.1016/j.pce.2010.07.014.
Rico, T. E. F., Santos, L. M., Reis, E. M., Silva, F. F., & Zonetti, P. C. (2010). Treatment of tannery wastewater with use of moringa seeds Moringa oleifera L in Portuguese: Tratamento de água residuária de curtume com utilização de sementes de moringa Moringa oleifera L. AgroAmbiente, 4, 96–101. https://doi.org/10.18227/1982-8470ragro.v4i2.292.
Rozainy, M. R., Hasif, M., Syafalny, P., & Puganeshwary, A. A. (2014). Combination of chitosan and bentonite as coagulant agents in dissolved air flotation. APCBEE Proc., 10, 229–234. https://doi.org/10.1016/j.apcbee.2014.10.044.
Sánchez-Martín, J., Ghebremichael, K. A., & Beltrán-Heredia, J. (2010). Comparison of single step and two step purified coagulants from Moringa oleifera seed for turbidity and DOC removal. Bioresource Technol., 101, 6259–6261. https://doi.org/10.1016/j.biortech.2010.02.072.
Santos, A. F. S., Carneiro-Da-Cunha, M. G., Teixeira, J. A., Paiva, P. M. G., Coelho, L. C. B. B., & Nogueira, R. (2011). Interaction of Moringa oleifera seed lectin with humic acid. Chem. Papers, 65, 406–411. https://doi.org/10.2478/s11696-011-0025-2.
Saqrane, S., El Ghazali, I., Oudra, B., Bouarab, L., Dekayir, S., Mandi, L., Ouazzani, N., & Vasconcelos, V. M. (2009). Detection of microcystin contamination by the measurement of the variability of the in vivo chlorophyll fluorescence in aquatic plant Lemna gibba. Toxicon, 53, 9–14. https://doi.org/10.1016/j.toxicon.2008.10.004.
Sathishkumar, M., Pavagadhi, S., Vijayaraghavan, K., Balasubramanian, R., & Ong, S. L. (2011). Concomitant uptake of microcystin-LR and -RR by peat under various environmental conditions. Chem. Eng. J., 172, 754–762. https://doi.org/10.1016/j.cej.2011.06.058.
Shebek, K., Schantz, A. B., Sines, I., Lauser, K., Velegol, S., & Kumar, M. (2015). The flocculating cationic polypetide from Moringa oleifera seeds damages bacterial cell membranes by causing membrane fusion. Langmuir, 31, 4496–4502. https://doi.org/10.1021/acs.langmuir.5b00015.
Shen, Q., Zhu, J., Cheng, L., Zhang, J., Zhang, Z., & Xu, X. (2011). Enhanced algae removal by drinking water treatment of chlorination coupled with coagulation. Desalination, 271, 236–240. https://doi.org/10.1016/j.desal.2010.12.039.
Sing, K. S. W., Everett, D. H., Haul, R. A. W., Moscou, L., Pierotti, R. A., Rouquérol, J., & Siemieniewska, T. (1985). Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. International Union of Pure and Applied Chemistry – IUPAC. Pure Appl. Chemis., 57, 603–619.
Swati, M., & Govindan, V. S. (2005). Coagulation studies on natural seed extracts. J. Indian Water Works Ass., 37, 145–149.
Teixeira, M. R., & Rosa, M. J. (2006a). Integration of dissolved gas flotation and nanofiltration for M. aeruginosa and associated microcystins removal. Water Res., 40, 3612–3620. https://doi.org/10.1016/j.watres.2006.06.006.
Teixeira, M. R., & Rosa, M. J. (2006b). Comparing dissolved air flotation and conventional sedimentation to remove cyanobacterial cells of Microcystis aeruginosa Part I: The key operating conditions. Sep. Purif. Technol., 52, 84–94. https://doi.org/10.1016/j.seppur.2006.03.017.
Teixeira, M. R., & Rosa, M. J. (2007). Comparing dissolved air flotation and conventional sedimentation to remove cyanobacterial cells of Microcystis aeruginosa Part II. The effect of water background organics. Sep. Purif. Technol., 53, 126–134. https://doi.org/10.1016/j.seppur.2006.03.017.
Teixeira, M. R., Sousa, V., & Rosa, M. J. (2010). Investigating dissolved air flotation performance with cyanobacterial cells and filaments. Water Res., 44, 3337–3344. https://doi.org/10.1016/j.watres.2010.03.012.
Valverde, K. C., Paccola, E. A. S., Pomini, A. M., Yamaguchi, N. U., & Bergamasco, R. (2018). Combined water treatment with extract of natural Moringa oleifera Lam and synthetic coagulant. Ambiente & Água - An Interd. J. Appl. Sci., 13, e2135. https://doi.org/10.4136/ambi-agua.2135.
Van Wichelen, J., Vanormelingen, P., Codd, G. A., & Vyverman, W. (2016). The common bloom-forming cyanobacterium Microcystis is prone to a wide array of microbial antagonists. Harmful Algae, 55, 97–111. https://doi.org/10.1016/j.hal.2016.02.009.
WHO - World Health Organization, Guidelines for drinking-water quality, 4th edition, incorporating the 1st addendum, Geneva, 631 p., Licence: CC BY-NC-SA 3.0 IGO, 2017.
Yang, Z., & Kong, F. (2012). Formation of large colonies: a defense mechanism of Microcystis aeruginosa under continuous grazing pressure by flagellate Ochromonas sp. J. Limnol., 71, 61–66. https://doi.org/10.4081/jlimnol.2012.e5.
Yuheng, W., Shengguang, Z., Na, L., & Yixin, W. (2011). Influences of various aluminum coagulants on algae floc structure, strength and flotation effect. Procedia Environ. Sci., 8, 75–80. https://doi.org/10.1016/j.proenv.2011.10.014.
Žegura, B., Zajc, I., Lah, T. T., & Filipič, M. (2008). Patterns of microcystin-LR induced alteration of the expression of genes involved in response to DNA damage and apoptosis. Toxicon, 51, 615–623. https://doi.org/10.1016/j.toxicon.2007.11.009.
Zonoozi, M. H., Alavi Moghaddam, M. R., & Arami, M. (2011). Study on the removal of acid dyes using chitosan as a natural coagulant/coagulant aid. Water Sci. Technol., 63, 403–409. https://doi.org/10.2166/wst.2011.234.
Zyska, A., & Jasik-Ślęzak, J. (2014). Mechanism and effects of cyanobacterial hepatotoxin action on human organism. Pol. J. Public Health, 124, 156–159. https://doi.org/10.2478/pjph-2014-0035.
Acknowledgments
This work was supported by the National Council of Scientific and Technology Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq) and the Post-Graduate Program in Environmental Science and Technology, from the Federal University of Technology, Paraná, Brazil. The Moringa oleifera seeds were donated by the Federal University, Paraíba, Brazil.
Funding
This research was funded by the National Council of Scientific and Technology Development (CNPq), grant number 487115/2013-9.
Author information
Authors and Affiliations
Contributions
Conceptualization, L.A.A.C. and F.J.B.; methodology, M.S.C.; formal analysis, M.S.C.; investigation, M.S.C.; resources, F.J.B.; writing (original draft preparation), M.S.C.; writing (review and editing), R.A.K., L.A.A.C., and F.J.B.; supervision, L.A.A.C. and F.J.B.; project administration, F.J.B.; and funding acquisition, F.J.B. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Carvalho, M.S., de Almeida Konzen, R., de Almeida Coral, L.A. et al. Evaluation of Moringa oleifera Extract as a Biocoagulant to Remove Microcystis aeruginosa Cells and Dissolved Metabolites. Water Air Soil Pollut 232, 163 (2021). https://doi.org/10.1007/s11270-021-05136-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05136-w