Abstract
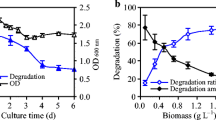
Decachlorobiphenyl (DCB) is one of the 209 polychlorinated biphenyls congeners characterized by its high toxicity and chemical stability. It is produced by industrial activities. A possible strategy to eliminate DCB is by bacterial degradation. The main objective of this study was to define the optimal conditions for biodegradation and bioaccumulation of DCB by Pseudomonas extremaustralis ADA-5 isolated from a worm intestine. Bacterial growth kinetics were determined in minimal medium with added biphenyl and DCB. By GC coupled to mass spectrometry, we found that the strain had the ability to degrade 9.75% of available DCB, using it as a carbon source and was able to accumulate 19.98% of this pollutant in biomass. Membrane lipids may be altered by DCB. Phosphatidylethanolamine (PE), phosphatidylglycerol (PG), and cardiolipin (CL) were identified by thin-layer chromatography as the membrane lipids of the cell. At 250 mg L−1 of DCB in the culture medium, membranes showed a 30% decrease in the PE concentration, an 18% increase in the PG, and a 12% increase in CL. ADA-5 was able to catabolize DCB and may be used for bioremediation of highly chlorinated toxic compounds in soil.




Similar content being viewed by others
References
Anyasi, R. O., & Atagana, H. I. (2011). Biological remediation of polychlorinated biphenyls (PCB) in the environment by microorganisms and plants. African Journal of Biotechnology, 10, 18916–18938. https://doi.org/10.5897/AJB10.557.
Bedard, D. L., Haberl, M. L., May, R. J., et al. (1987). Evidence for novel mechanisms of polychlorinated biphenyl metabolism in Alcaligenes eutrophus H850. Applied and Environmental Microbiology, 53, 1103–1112. https://doi.org/10.1128/AEM.53.5.1103-1112.1987.
Berg, J. M., Tymoczko, J. L., & Stryer, L. (2006). Biochemistry (5th ed.p. 1514). WH Freeman and Company.
Bernal, P., Muñoz-Rojas, J., & Hurtado, A. (2007). Pseudomonas putida cardiolipine synthesis mutant exhibits increased sensitivity to drugs related to transport functionality. Environmental Microbiology Reports, 9, 135–1145. https://doi.org/10.1111/j.1462-2920.2006.01236.x.
Bernat, P., Gajewska, E., Szewczyk, R., et al. (2013). Tributyltin (TBT) induces oxidative stress and modifies lipid profile in the filamentous fungus Cunninghamella elegans. Environmental Science and Pollution Research International, 21, 4228–4235. https://doi.org/10.1007/s11356-013-2375-5.
Bernat, P., Siewiera, P., Soboń, A., et al. (2014). Phospholipids and protein adaptation of Pseudomonas sp. to the xenoestrogen tributyltin chloride (TBT). World Journal of Microbiology and Biotechnology, 9, 2343–2350. https://doi.org/10.1007/s11274-014-1659-3.
Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37, 911–917. https://doi.org/10.1139/o59-099.
Chakraborty, J., & Das, S. (2016). Characterization of the metabolic pathway and catabolic gene expression in biphenyl degrading marine bacterium Pseudomonas aeruginosa JP-11. Chemosphere, 144, 1706–1714. https://doi.org/10.1016/j.chemosphere.2015.10.059.
Costabeber, I., Sifuentes, D. S. J., Odorissi, X. A., et al. (2006). Levels of polychlorinated biphenyls (PCBs) in meat and meat products from the state of Rio Grande do Sul, Brazil. Food and Chemical Toxicology, 44, 1–7. https://doi.org/10.1016/j.fct.2005.01.005.
De Limae Silva, M. R., Correa, R. C., Sakamoto, I. K., et al. (2018). Microbial characterization of methanogenic and iron-reducing consortium in reactors with polychlorinated biphenyls. Current Microbiology, 75, 666–676. https://doi.org/10.1007/s00284-018-1431-2.
De Rudder, K. E., Thomas-Oates, J. E., & Geiger, O. (1997). Rhizobium meliloti mutants deficient in phospholipid N-methyltransferase still contain phosphatidylcholine. Journal of Bacteriology, 179, 6921–6928. https://doi.org/10.1128/jb.179.22.6921-6928.1997.
Del Puerto, C., Iglesias, E., Morales, T., et al. (2009). Organización y manejo de la colección de cepas de referencia del Instituto Finlay. VacciMonitor, 18, 20–24 Available on-line at: https://www.redalyc.org/articulo.oa?id=203414612004.
Denich, T. J., Beaudette, L. A., Lee, H., et al. (2003). Effect of selected environmental and physico-chemical factors on bacterial cytoplasmic membranes. Journal of Microbiological Methods, 52, 149–182. https://doi.org/10.1016/S0167-7012(02)00155-0.
Dercová, K., Murínová, S., Dudášová, H., et al. (2018). The adaptation mechanisms of bacteria applied in bioremediation of hydrophobic toxic environmental pollutants: How indigenous and introduced bacteria can respond to persistent organic pollutants-induced stress? Persistent organic pollutants, Stephen Kudom Donyinah. IntechOpen. https://doi.org/10.5772/intechopen.79646.
Donato, M. M., Jurado, A. S., & Antunes-Madeira, M. C. (1997). Effects of a lipophilic environmental pollutant (DDT) on the phospholipid and fatty acid contents of Bacillus stearothermophilus. Archives of Environmental Contamination and Toxicology, 33, 341–349. https://doi.org/10.1007/s002449900263.
Escárcega-González, C. E., Garza-Cervantes, J. A., Vázquez-Rodríguez, A., et al. (2018). Bacterial exopolysaccharides as reducing and/or stabilizing agents during synthesis of metal nanoparticles with biomedical applications. International Journal of Polymer Science, 7045852, 15. https://doi.org/10.1155/2018/7045852.
Geel-Schutten, G., Flesch, F., Ten, B. B., et al. (1998). Screening and characterization of Lactobacillus strains producing large amounts of exopolysaccharides. Applied Microbiology and Biotechnology, 50, 697–703. https://doi.org/10.1007/s002530051353.
Green, M. R., & Joseph, S. (2012). Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press.
Han, X., O’Connor, J. C., Donner, E. M., et al. (2009). Noncoplanar 2,2′, 3,3′, 4,4′, 5,5′, 6,6′-decachlorobiphenyl (PCB 209) did not induce cytochrome P450 enzyme activities in primary cultured rat hepatocytes, was not genotoxic, and did not exhibit endocrine-modulating activities. Toxicology, 255, 177–186. https://doi.org/10.1016/j.tox.2008.10.013.
Hatamian-Zarmi, A., Shojaosadati, S. A., Vasheghani-Farahani, E., et al. (2009). Extensive biodegradation of highly chlorinated biphenyl and Aroclor 1242 by Pseudomonas aeruginosa TMU56 isolated from contaminated soils. International Biodeterioration and Biodegradation, 63, 788–794. https://doi.org/10.1016/j.ibiod.2009.06.009.
Huang, L., Su, G., & Liu, Y. (2014). Effect of NiFe2O4 on PCDF by products formation during termal degradation of decachlorobiphenyl. RSC Advances, 4, 25453–25460. https://doi.org/10.1039/c4ra02580f.
Hughes, D., & Clark, B. R. (2011). Biodegradation of polyfluorinated biphenyl in bacteria. Biodegradation, 22, 741–749. https://doi.org/10.1007/s10532-010-9411-7.
Martínez-Toledo, A., & Rodríguez-Vázquez, R. (2011). Response surface methodology (Box-Behnken) to improve a liquid media formulation to produce biosurfactant and phenanthrene removal by Pseudomonas putida. Annals of Microbiology, 61, 605–613. https://doi.org/10.1007/s13213-010-0179-0.
Mathews, S., & Sithebe, P. (2018). The role of bacteria on the breakdown of recalcitrant polychlorinated biphenyls (PCBs) compounds in wastewater. Waste Water and Water Quality. https://doi.org/10.5772/intechopen.75400.
Mishra, A., & Jha, B. (2013). Microbial Exopolysaccharides. In E. Rosenberg, E. F. De Long, S. Lory, E. Stackebrandt, & F. Thompson (Eds.), The Prokaryotes. Springer. https://doi.org/10.1007/978-3-642-31331-8-25.
Murínová, S., & Dercová, K. (2014). Response mechanisms of bacterial degraders to environmental contaminants on the level of cell walls and cytoplasmic membrane. International Journal of Microbiology, 873081, 16. https://doi.org/10.1155/2014/873081.
Polak, M. L., Zlatic, E., Demšar, L., et al. (2016). Degradation of PCBs in dry fermented sausages during drying/ripening. Food Chemistry, 213, 246–250. https://doi.org/10.1016/j.foodchem.2016.06.051.
Prossnigg, F., Hickel, A., Pabst, G., et al. (2010). Packing behaviour of two predominant anionic phospholipids of bacterial cytoplasmic membranes. Biophysical Chemistry, 50, 129–135. https://doi.org/10.1016/j.bpc.2010.04.004.
Qiu, L., Wang, H., & Wang, X. (2016). Isolation and characterization of a cold-resistant PCB209-degrading bacterial strain from river sediment and its application in bioremediation of contaminated soil. Journal of Environmental Science and Health Part C, 51, 204–212. https://doi.org/10.1080/10934529.2015.1094324.
Rühl, J., Hein, E. M., & Hayen, H. (2012). The glycerophospholipid inventory of Pseudomonas putida is conserved between strains and enables growth condition-related alterations. Microbial Biotechnology, 5, 45–58. https://doi.org/10.1111/j.1751-7915.2011.00286.x.
Sánchez-Pérez, B. N., Zenteno-Rojas, A., Rincón-Molina, C. I., et al. (2019). Rhizosphere and endophytic bacteria associated to Ocimum basilicum L. with decaclorobiphenyl removal potential. Water, Air, & Soil Pollution, 231, 134. https://doi.org/10.1007/s11270-020-04481-6.
Sandri, F., Fedi, S., & Zannoni, D. (2017). Biphenyl modulates the expression and function of respiratory oxidases in the polychlorinated-biphenyls degrader Pseudomonas pseudoalcaligenes KF707. Frontiers in Microbiology, 8, 1223. https://doi.org/10.3389/fmicb.2017.01223.
She, J., Holden, A., Sharp, M., et al. (2007). Polybrominated diphenyl ethers (PBDEs) and polychlorinated biphenyls (PCBs) in breast milk from the Pacific Northwest. Chemosphere, 67, S307–S317. https://doi.org/10.1016/j.chemosphere.2006.05.154.
Tandlich, R., Vrana, B., & Balaz, S. (2011). Biodegradation mechanism of biphenyl by a strain of Pseudomonas stutzeri. Journal of Environmental Science and Health, Part C, 46, 337–344. https://doi.org/10.1080/10934529.2011.542383.
Tharakan, J., Tomlinson, D., Addagada, A., et al. (2006). Biotransformation of PCBs in contaminated sludge: potential for novel biological technologies. Engineering in Life Sciences, 6, 43–50. https://doi.org/10.1002/elsc.200620117.
Valenzuela-Encinas, C., Neria-Gonzalez, I., Alcantara-Hernández, R. J., et al. (2008). Phylogenetic analysis of the archaeal community in an alkaline saline soil of the former lake Texcoco (México). Extremophiles, 12, 247–254. https://doi.org/10.1007/s00792-007-0121-y.
Vidali, M. (2001). Bioremediation an overview. Pure and Applied Chemistry, 73, 1163–1172. https://doi.org/10.1351/pac200173071163.
Villacieros, M., Whelan, C., Mackova, M., et al. (2005). Polychlorinated biphenyl rhizoremediation by Pseudomonas fluorescens F113 derivatives, using a Sinorhizobium meliloti nod system to drive bph gene expression. Applied and Environmental Microbiology, 71, 2687–2694. https://doi.org/10.1128/AEM.71.5.2687-2694.2005.
Villalobos-Maldonado, J. J., Meza-Gordillo, R., Mancilla-Margalli, N. A., et al. (2015). Removal of decachlorobiphenyl in vermicomposting process amended with rabbit manure and peat moss. Water, Air, & Soil Pollution, 226, 159. https://doi.org/10.1007/s11270-015-2400-z.
Weber, F. J., & de Bont, J. A. M. (1996). Adaptation mechanisms of microorganisms to the toxic effects of organic solvents on membranes. Biochimica et Biophysica Acta, 1286, 225–245. https://doi.org/10.1016/S0304-4157(96)00010-X.
Weiland, B. N., Fischer, M. A., Schramm, K. W., et al. (2017). Polychlorinated biphenyl (PCB)-degrading potential of microbes present in a cryoconite of jamtalferner glacier. Frontier of Microbiology, 8, 110.5. https://doi.org/10.3389/fmicb.2017.01105.
Yu, Y., Yin, H., Peng, H., et al. (2020). Proteomic mechanism of decabromodiphenyl ether (BDE-209) biodegradation by Microbacterium Y2 and its potential in remediation of BDE-209 contaminated water-sediment system. Journal of Hazardous Materials, 387, 0304–3894. https://doi.org/10.1016/j.jhazmat.2019.121708.
Zenteno-Rojas, A., Martínez-Romero, E., Rincón-Molina, C. I., et al. (2019). Removal of high concentrations decachlorobiphenyl of earthworm Eisenia fetida and its symbiotic bacteria in a vermicomposting system. Water, Air, & Soil Pollution, 230, 116. https://doi.org/10.1007/s11270-019-4170-5.
Zhao, L., Hou, H., & Shimoda, K. (2012). Formation pathways of polychlorinated dibenzofurans (PCDFs) in sediments contaminated with PCBs during the thermal desorption process. Chemosphere, 88, 1368–1374. https://doi.org/10.1016/j.chemosphere.2012.05.042.
Zorádová, S., Dudášová, H., Lukáčová, L., et al. (2011). The effect of polychlorinated biphenyls (PCBs) on the membrane lipids of Pseudomonas stutzeri. International Biodeterioration and Biodegradation, 65, 1019–1023. https://doi.org/10.1016/j.ibiod.2011.03.012.
Acknowledgements
We thank the Biochemical Engineering Postgraduate Department-ITTG and CONACYT for granting a scholarship to Miguel Ángel Gómez López. We thank MA. Celina Lujan Hidalgo from the ITTG laboratory for technical assistance. We thank Michael Dunn for reading the manuscript.
Funding
Miguel Ángel Gómez López had a Conacyt scholarship during his Master of Science in Biochemical Engineering. Financial support was from Tecnologico Nacional de México 7676.20-P.
Author information
Authors and Affiliations
Contributions
All authors contributed to this study and have approved the final manuscript. RRR designed the study. MAGL and AZR performed laboratory experiments and data analysis. CIRM, LAMG, and MAVG contributed new reagents and analytical tools. VMRV and FARM data analysis. EMR, RRR, and AZR wrote the manuscript.
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 12 kb)
Rights and permissions
About this article
Cite this article
López, M.A.G., Zenteno-Rojas, A., Martinez-Romero, E. et al. Biodegradation and Bioaccumulation of Decachlorobiphenyl (DCB) by Native Strain Pseudomonas extremaustralis ADA-5. Water Air Soil Pollut 232, 192 (2021). https://doi.org/10.1007/s11270-021-05122-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05122-2