Abstract
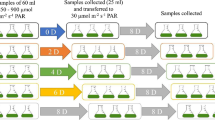
Egeria densa release allelochemicals harmful to other species, including cyanobacteria. Under natural conditions, E. densa and Microcystis aeruginosa can be found in various aquatic systems with different light levels. The effect that the coexistence of E. densa has on M. aeruginosa was tested under different photosynthetically active radiation (PAR) intensities. The growth, chlorophyll-a, oxidative stress, and antioxidant activities of M. aeruginosa were quantified after 7 days of coexistence with E. densa. A control experiment without E. densa was conducted for M. aeruginosa under the same conditions to distinguish the effect of the PAR intensity from the allelopathic effect. The allelopathy of E. densa on M. aeruginosa is significantly influenced by the PAR intensity. PAR intensities lower or higher than the favorable range further suppressed M. aeruginosa under coexistence. Research has revealed that the allelopathic effect of E. densa can be utilized to biologically suppress M. aeruginosa under various light conditions. Strong second-order polynomial distributions between the optical density (OD) and cellular H2O2 content of M. aeruginosa were found for both the control and coexistence conditions. In the practical application of E. densa allelopathy, OD can be adopted as a convenient method to obtain an approximate stress status on M. aeruginosa.









Similar content being viewed by others
References
Aebi, H. (1984). Catalase in vitro. Methods in Enzymology, 105(C), 121–126. https://doi.org/10.1016/S0076-6879(84)05016-3.
Amorim, C. A., Moura-Falcão, R. H., Valença, C. R., Souza, V. R., & Moura, A. N. (2019). Allelopathic effects of the aquatic macrophyte Ceratophyllum demersum L. on phytoplankton species: Contrasting effects between cyanobacteria and chlorophytes. Acta Limnologica Brasiliensia, 31. https://doi.org/10.1590/s2179-975x1419.
Anderson, D. M. (2009). Approaches to monitoring, control and management of harmful algal blooms (HABs). Ocean and Coastal Management, 52(7), 342. https://doi.org/10.1016/j.ocecoaman.2009.04.006.
Asaeda, T., Senavirathna, M. D. H. J., & Vamsi Krishna, L. (2020). Evaluation of habitat preferences of invasive macrophyte Egeria densa in different channel slopes using hydrogen peroxide as an indicator. Frontiers in Plant Science, 11, 422. https://doi.org/10.3389/fpls.2020.00422.
Atapaththu, K. S. S., Miyagi, A., Atsuzawa, K., Kaneko, Y., Kawai-Yamada, M., & Asaeda, T. (2015). Effects of water turbulence on variations in cell ultrastructure and metabolism of amino acids in the submersed macrophyte, Elodea nuttallii (Planch.) H. St. John. Plant Biology, 17(5), 997–1004. https://doi.org/10.1111/plb.12346.
Baird, R., & Bridgewater, L. (2017). Standard methods for the examination of water and wastewater (3rd edition)
Bortoli, S., Oliveira-Silva, D., Krüger, T., Dörr, F. A., Colepicolo, P., Volmer, D. A., & Pinto, E. (2014). Growth and microcystin production of a Brazilian Microcystis aeruginosa strain (LTPNA 02) under different nutrient conditions. Revista Brasileira de Farmacognosia, 24(4), 389–398. https://doi.org/10.1016/j.bjp.2014.07.019.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72(1–2), 248–254. https://doi.org/10.1016/0003-2697(76)90527-3.
Chen, J., Cao, T., Zhang, X., Xi, Y., Ni, L., & Jeppesen, E. (2016). Differential photosynthetic and morphological adaptations to low light affect depth distribution of two submersed macrophytes in lakes. Scientific Reports, 6, 34028. https://doi.org/10.1038/srep34028.
Clare, D. S., Spencer, M., Robinson, L. A., & Frid, C. L. J. (2016). Species-specific effects on ecosystem functioning can be altered by interspecific interactions. PLoS ONE, 11(11), e0165739. https://doi.org/10.1371/journal.pone.0165739.
Derks, A., Schaven, K., & Bruce, D. (2015). Diverse mechanisms for photoprotection in photosynthesis. Dynamic regulation of photosystem II excitation in response to rapid environmental change. Biochimica et Biophysica Acta, 1847(4–5), 468–485. https://doi.org/10.1016/j.bbabio.2015.02.008.
Espinosa-Rodríguez, C. A., Sarma, S. S. S., & Nandini, S. (2017). Effect of the allelochemicals from the macrophyte Egeria densa on the competitive interactions of pelagic and littoral cladocerans. Chemistry and Ecology, 33(3), 247–256. https://doi.org/10.1080/02757540.2017.1288226.
Ferrão-Filho, A. S., & Kozlowsky-Suzuki, B. (2011). Cyanotoxins: Bioaccumulation and effects on aquatic animals. Marine Drugs, 9(12), 2729–2772. https://doi.org/10.3390/md9122729.
Guidi, L., Lo Piccolo, E., & Landi, M. (2019). Chlorophyll fluorescence, photoinhibition and abiotic stress: Does it make any difference the fact to be a C3 or C4 species? Frontiers in Plant Science, 10, 174. https://doi.org/10.3389/fpls.2019.00174.
Gulati, R. D., Dionisio Pires, L. M. D., & Van Donk, E. (2008). Lake restoration studies: Failures, bottlenecks and prospects of new ecotechnological measures. Limnologica, 38(3–4), 233–247. https://doi.org/10.1016/j.limno.2008.05.008.
Hobbie, S. E., Finlay, J. C., Janke, B. D., Nidzgorski, D. A., Millet, D. B., & Baker, L. A. (2017). Contrasting nitrogen and phosphorus budgets in urban watersheds and implications for managing urban water pollution. Proceedings of the National Academy of Sciences of the United States of America, 114(16), 4177–4182. https://doi.org/10.1073/pnas.1618536114.
Howard, A. (1994). Problem cyanobacterial blooms: explanation and simulation modelling. Transactions of the Institute of British Geographers, 19(2), 213–224. https://doi.org/10.2307/622755.
Hu, H., & Hong, Y. (2008). Algal-bloom control by allelopathy of aquatic macrophytes – A review. Frontiers of Environmental Science & Engineering in China, 2(4), 421–438. https://doi.org/10.1007/s11783-008-0070-4.
Ilavarasi, A., Mubarakali, D., Praveenkum, R., Baldev, E., & Thajuddin, N. (2011). Optimization of various growth media to freshwater microalgae for biomass production. Biotechnology (Faisalabad), 10(6), 540–545. https://doi.org/10.3923/biotech.2011.540.545.
Jana, S., & Choudhuri, M. A. (1984). Effects of plant growth regulators on Hill activity of submerged aquatic plants during induced senescence. Aquatic Botany, 18(4), 371–376.
Kortmann, R. W., Knoecklein, G. W., & Bonnell, C. H. (1994). Aeration of stratified lakes: Theory and practice. Lake and Reservoir Management, 8(2), 99–120. https://doi.org/10.1080/07438149409354463.
Leblanc Renaud, S. L. B., Pick, F. R., & Fortin, N. (2011). Effect of light intensity on the relative dominance of toxigenic and nontoxigenic strains of Microcystis aeruginosa. Applied and Environmental Microbiology, 77(19), 7016–7022. https://doi.org/10.1128/AEM.05246-11.
Lürling, M., Waajen, G., & de Senerpont Domis, L. N. (2016). Evaluation of several end-of-pipe measures proposed to control cyanobacteria. Aquatic Ecology, 50(3), 499–519. https://doi.org/10.1007/s10452-015-9563-y.
Lürling, M., Mello, M. M. E., Van Oosterhout, F., de Senerpont Domis, L., & Marinho, M. M. (2018). Response of natural cyanobacteria and algae assemblages to a nutrient pulse and elevated temperature. Frontiers in Microbiology, 9(AUG), 1851. https://doi.org/10.3389/fmicb.2018.01851.
MacAdam, J. W., Nelson, C. J., & Sharp, R. E. (1992). Peroxidase activity in the leaf elongation zone of tall fescue: I. Spatial distribution of ionically bound peroxidase activity in genotypes differing in length of the elongation zone. Plant Physiology, 99(3), 872–878 http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1080558/.
Manganelli, M., Scardala, S., Stefanelli, M., Palazzo, F., Funari, E., Vichi, S., Buratti, M. F, & Testai, E. (2012). Emerging health issues of cyanobacterial blooms. Annali Dell’Istituto Superiore di Sanita, 48(4), 415–428. https://doi.org/10.4415/ANN_12_04_09
Mohamed, Z. A. (2017). Macrophytes-cyanobacteria allelopathic interactions and their implications for water resources management – A review. Limnologica, 63, 122–132. https://doi.org/10.1016/j.limno.2017.02.006.
Moosavi, A., Afshari, R. T., Asadi, A., & Gharineh, M. H. (2011). Allelopathic effects of aqueous extract of leaf stem and root of sorghum bicolor on seed germination and seedling growth of Vigna radiata L. Notulae Scientia Biologicae, 3, 114–118. https://doi.org/10.15835/nsb.3.2.5862.
Mowe, M. A. D., Mitrovic, S. M., Lim, R. P., Furey, A., & Yeo, D. C. J. (2015). Tropical cyanobacterial blooms: A review of prevalence, problem taxa, toxins and influencing environmental factors. Journal of Limnology, 73(AoP). https://doi.org/10.4081/jlimnol.2014.1005.
Muhetaer, G., Asaeda, T., Senavirathna, M. D. H. J., Baniya, M. B., Abeynayaka, H. D. L., Rashid, M. H., & Yan, H. (2020). Effects of light intensity and exposure period on the growth and stress responses of two cyanobacteria species: Pseudanabaena galeata and Microcystis aeruginosa. Water, 12, 407. https://doi.org/10.3390/w12020407.
Mulderij, G., Mau, B., van Donk, E., & Gross, E. M. (2007). Allelopathic activity of Stratiotes aloides on phytoplankton—towards identification of allelopathic substances. Hydrobiologia, 584, 89–100. https://doi.org/10.1007/s10750-007-0602-0.
Nakai, S., Zou, G., Okuda, T., Nishijima, W., & Okada, M. (2012). Polyphenols and fatty acids responsible for anti-cyanobacterial allelopathic effects of submerged macrophyte Myriophyllum spicatum. Water Science and Technology, 66, 993–999. https://doi.org/10.2166/wst.2012.272.
Nakano, Y., & Asada, K. (1981). Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant and Cell Physiology, 22(5), 867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232.
Paerl, H. W., & Huisman, J. (2009). Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environmental Microbiology Reports, 1(1), 27–37. https://doi.org/10.1111/j.1758-2229.2008.00004.x.
Princiotta, S. D., Hendricks, S. P., & White, D. S. (2019). Production of cyanotoxins by Microcystis aeruginosa mediates interactions with the mixotrophic flagellate cryptomonas. Toxins, 11(4). https://doi.org/10.3390/toxins11040223.
Rastogi, R. P., Madamwar, D., & Incharoensakdi, A. (2015). Bloom dynamics of cyanobacteria and their toxins: Environmental health impacts and mitigation strategies. Frontiers in Microbiology, 6, 1254. https://doi.org/10.3389/fmicb.2015.01254.
Riis, T., Olesen, B., Clayton, J. S., Lambertini, C., Brix, H., & Sorrell, B. K. (2012). Growth and morphology in relation to temperature and light availability during the establishment of three invasive aquatic plant species. Aquatic Botany, 102, 56–64. https://doi.org/10.1016/j.aquabot.2012.05.002.
Rodrigues, R. B., & Thomaz, S. M. (2010). Photosynthetic and growth responses of Egeria densa to photosynthetic active radiation. Aquatic Botany, 92(4), 281–284. https://doi.org/10.1016/j.aquabot.2010.01.009.
Salvador, D., Churro, C., & Valério, E. (2016). Evaluating the influence of light intensity in mcyA gene expression and microcystin production in toxic strains of Planktothrix agardhii and Microcystis aeruginosa. Journal of Microbiological Methods, 123, 4–12. https://doi.org/10.1016/j.mimet.2016.02.002.
Senavirathna, M. D. H. J., Muhetaer, G., Zhaozhi, L., & Fujino, T. (2020). Allelopathic influence of low concentration Microcystis aeruginosa on Egeria densa under different light intensities. Chemistry and Ecology, 36(10), 903–921. https://doi.org/10.1080/02757540.2020.1798939.
Senousy, H. H., Abd Ellatif, S., & Ali, S. (2020). Assessment of the antioxidant and anticancer potential of different isolated strains of cyanobacteria and microalgae from soil and agriculture drain water. Environmental Science and Pollution Research International, 27(15), 18463–18474. https://doi.org/10.1007/s11356-020-08332-z.
Shao, J., Li, R., Lepo, J. E., & Gu, J. D. (2013). Potential for control of harmful cyanobacterial blooms using biologically derived substances: Problems and prospects. Journal of Environmental Management, 125, 149–155. https://doi.org/10.1016/j.jenvman.2013.04.001.
Shi, K., Zhang, Y., Zhou, Y., Liu, X., Zhu, G., Qin, B., & Gao, G. (2017). Long-term MODIS observations of cyanobacterial dynamics in Lake Taihu: Responses to nutrient enrichment and meteorological factors. Scientific Reports, 7, 40326. https://doi.org/10.1038/srep40326.
Sigee, D. C., Glenn, R., Andrews, M. J., Bellinger, E. G., Butler, R. D., Epton, H. A. S., & Hendry, R. D. (1999). Biological control of cyanobacteria: Principles and possibilities. Hydrobiologia, 395, 161–172. https://doi.org/10.1023/A:1017097502124.
Slim, K., Fadel, A., Atoui, A., Lemaire, B. J., Vinçon-Leite, B., & Tassin, B. (2014). Global warming as a driving factor for cyanobacterial blooms in Lake Karaoun, Lebanon. Desalination and Water Treatment, 52(10–12), 2094–2101. https://doi.org/10.1080/19443994.2013.822328.
Sodaeizadeh, H., Rafieiolhossaini, M., Havlík, J., & Van Damme, P. (2009). Allelopathic activity of different plant parts of Peganum harmala L. and identification of their growth inhibitors substances. Plant Growth Regulation, 59, 227–236. https://doi.org/10.1007/s10725-009-9408-6.
Stroom, J. M., & Kardinaal, W. E. A. (2016). How to combat cyanobacterial blooms: Strategy toward preventive lake restoration and reactive control measures. Aquatic Ecology, 50(3), 541–576. https://doi.org/10.1007/s10452-016-9593-0.
Tan, K., Huang, Z., Ji, R., Qiu, Y., Wang, Z., & Liu, J. (2019). A review of allelopathy on microalgae. Microbiology, 165(6), 587–592. https://doi.org/10.1099/mic.0.000776.
Tanabe, Y., Hodoki, Y., Sano, T., Tada, K., & Watanabe, M. M. (2018). Adaptation of the freshwater bloom-forming cyanobacterium Microcystis aeruginosa to brackish water is driven by recent horizontal transfer of sucrose genes. Frontiers in Microbiology, 9(JUN), 1150. https://doi.org/10.3389/fmicb.2018.01150.
Van Hullebusch, E., Deluchat, V., Chazal, P. M., & Baudu, M. (2002). Environmental impact of two successive chemical treatments in a small shallow eutrophied lake: Part II. Case of copper sulfate. Environmental Pollution, 120(3), 627–634. https://doi.org/10.1016/S0269-7491(02)00191-4.
Visser, P. M., Ibelings, B. W., Bormans, M., & Huisman, J. (2016). Artificial mixing to control cyanobacterial blooms: A review. Aquatic Ecology, 50(3), 423–441. https://doi.org/10.1007/s10452-015-9537-0.
Wang, H., Zhong, G., Yan, H., Liu, H., Wang, Y., & Zhang, C. (2012). Growth control of cyanobacteria by three submerged macrophytes. Environmental Engineering Science, 29(6), 420–425. https://doi.org/10.1089/ees.2010.0286.
Wang, Y., Chen, X., Liu, J., Hong, Y., He, Q., Yu, D., Liu, C., & Dingshanbayi, H. (2019). Greater performance of exotic Elodea nuttallii in response to water level may make it a better invader than exotic Egeria densa during winter and spring. Frontiers in Plant Science, 10, 144. https://doi.org/10.3389/fpls.2019.00144.
Wells, M. L., Trainer, V. L., Smayda, T. J., Karlson, B. S. O., Trick, C. G., Kudela, R. M., Ishikawa, A., Bernard, S., Wulff, A., Anderson, D. M., & Cochlan, W. P. (2015). Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae, 49, 68–93. https://doi.org/10.1016/j.hal.2015.07.009.
Wiedner, C., Visser, P. M., Fastner, J., Metcalf, J. S., Codd, G. A., Mur, L. R. (2003) Effects of Light on the Microcystin Content of Microcystis Strain PCC 7806. Applied and Environmental Microbiology 69(3), 1475-1481. https://doi.org/10.1128/AEM.69.3.1475-1481.2003.
Yu, H., Shen, N., Yu, S., Yu, D., & Liu, C. (2018). Responses of the native species Sparganium angustifolium and the invasive species Egeria densa to warming and interspecific competition. PLoS ONE, 13(6), e0199478. https://doi.org/10.1371/journal.pone.0199478.
Zhao, C. S., Shao, N. F., Yang, S. T., Ren, H., Ge, Y. R., Feng, P., Dong, B. E., & Zhao, Y. (2019). Predicting cyanobacteria bloom occurrence in lakes and reservoirs before blooms occur. Science of the Total Environment, 670, 837–848. https://doi.org/10.1016/j.scitotenv.2019.03.161.
Acknowledgements
We would like to acknowledge Mr. Zhaozhi Liu for assisting the research.
Funding
This research was funded by the Japanese Society for the Promotion of Science (JSPS), JSPS KAKENHI grant number 18K13833 and 2020 Young Researcher Support funding of Saitama University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Senavirathna, M.D.H.J., Muhetaer, G., Atapaththu, K.S.S. et al. Egeria densa Allelopathy on Microcystis aeruginosa Under Different Light Intensities and Preliminary Insight into Inter-Parameter Relationships. Water Air Soil Pollut 232, 135 (2021). https://doi.org/10.1007/s11270-021-05088-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05088-1