Abstract
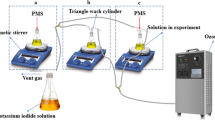
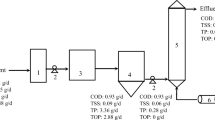
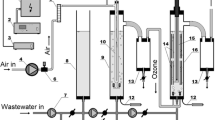
The large use of plasticizers in industry produced large amounts of wastewater. The treatment of industrial wastewater with advanced oxidation processes (AOPs) has attracted widespread interest from scientists in recent years. Comparing with several common AOPs such as activated persulfate, Fenton, UV, and H2O2, the ozone oxidation technology has the advantages of not introducing other chemical reagents, not bringing secondary pollution, lower energy consumption, safety, and non-flammability or explosion. Using the ozone microbubble process to treat high-concentration acidic plasticizer wastewater is in line with the concepts of green, energy-saving, and environment friendly. This work studied the changes of chemical oxygen demand (COD), pH, and dissolved oxygen (DO) in wastewater by adjusting the reaction time, system pressure, and reaction temperature, and revealed the best working conditions of ozone microbubble technology to treat plasticizer wastewater. The experiment shows that with the condition of the reaction time of 45 h, the pressure of 0.150 MPa, the ozone concentration of 100%, and the gas flow of 0.7 L/min, the dissolved oxygen (DO) of the wastewater increased from 3.8 to 4.5 mg/L, while the pH value increased from 3.23 to 7.54, and the COD removal rate reached up to 94.18%. This work discussed the mechanism of ozone microbubble technology to degrade plasticizer wastewater, and also confirmed that ozone microbubble technology can generate high hydroxyl radicals, even under acidic media. In addition, this technology does not require the addition of any additional chemical reagents and does not form a precipitate in the reaction to cause secondary pollution to the environment. Meanwhile, the water treatment costs of unit tons using this technology have also been analyzed. This technique has great practical application prospects in treating high concentration organic acid wastewater.







Similar content being viewed by others
References
Agarwal, A., Ng, W. J., & Liu, Y. (2011). Principle and applications of microbubble and nanobubble technology for water treatment. Chemosphere, 84, 1175–1180.
Beauchesne, I., Barnabe, S., Cooper, D. G., & Nicell, J. A. (2008). Plasticizers and related toxic degradation products in wastewater sludges. Water Science and Technology, 57, 367–374.
Chu, L. B., Xing, X. H., Yu, A. F., Sun, X. L., & Jurcik, B. (2008). Enhanced treatment of practical textile wastewater by microbubble ozonation. Process Safety and Environmental Protection, 86, 389–393.
Comninellis, C., Kapalka, A., Malato, S., Parsons, S. A., Poulios, L., & Mantzavinos, D. (2008). Advanced oxidation processes for water treatment: advances and trends for R&D. Journal of Chemical Technology and Biotechnology, 83, 769–776.
Fan, X., Rui, Y., Han, X., Yang, J., Wang, Y., & Zhang, Q. (2020). Spray-coated monodispersed SnO2 microsphere films as scaffold layers for efficient mesoscopic perovskite solar cells. Journal of Power Sources, 448, 227405.
Gasanov, A. A. (2018). Application of countercurrent extraction treatment of wastewater for its repeated use. Journal of Water Chemistry and Technology, 40, 151–156.
Kalabukhova, T. N. (1998). The van’t Hoff rule as applied to studying different phases of hibernation of ground squirrel Citellus undulatus Pallas: physicochemical way of cold adaptation. Biophysics, 43, 1027–1031.
Kasprzyk-Hordern, B., Ziolek, M., & Nawrocki, J. (2003). Catalytic ozonation and methods of enhancing molecular ozone reactions in water treatment. Applied Catalysis B-Environmental, 46, 639–669.
Khuntia, S., Majumder, S. K., & Ghosh, P. (2015a). A pilot plant study of the degradation of Brilliant Green dye using ozone microbubbles: mechanism and kinetics of reaction. Environmental Technology, 36, 336–347.
Khuntia, S., Majumder, S. K., & Ghosh, P. (2015b). Quantitative prediction of generation of hydroxyl radicals from ozone microbubbles. Chemical Engineering Research and Design, 98, 231–239.
Lee, Y.-G., Park, Y., Lee, G., Kim, Y., & Chon, K. (2019a). Enhanced degradation of pharmaceutical compounds by a microbubble ozonation process: effects of temperature, pH, and humic acids. Energies, 12, 4373.
Lee, Y. S., Lee, S., Lim, J. E., & Moon, H. B. (2019b). Occurrence and emission of phthalates and non-phthalate plasticizers in sludge from wastewater treatment plants in Korea. Science of the Total Environment, 692, 354–360.
Li, P., Takahashi, M., & Chiba, K. (2009). Degradation of phenol by the collapse of microbubbles. Chemosphere, 75, 1371–1375.
Li, Q., Wang, L., Fang, X., Zhang, L., Li, J., & Xie, H. (2019a). Synergistic effect of photocatalytic degradation of hexabromocyclododecane in water by UV/TiO2/persulfate. Catalysts, 9, 189.
Li, S., Chen, J., Jiang, W., Liu, Y., Ge, Y., & Liu, J. (2019b). Facile construction of flower-like bismuth oxybromide/bismuth oxide formate p-n heterojunctions with significantly enhanced photocatalytic performance under visible light. Journal of Colloid and Interface Science, 548, 12–19.
Li, S., Chen, J., Liu, Y., Xu, K., & Liu, J. (2019c). In situ anion exchange strategy to construct flower-like BiOCl/BiOCOOH p-n heterojunctions for efficiently photocatalytic removal of aqueous toxic pollutants under solar irradiation. Journal of Alloys and Compounds, 781, 582–588.
Li, S., Chen, J., Hu, S., Jiang, W., Liu, Y., & Liu, J. (2020). A novel 3D Z-scheme heterojunction photocatalyst: Ag6Si2O7 anchored on flower-like Bi2WO6 and its excellent photocatalytic performance for the degradation of toxic pharmaceutical antibiotics. Inorganic Chemistry Frontiers, 7, 529–541.
Lin, H., & Zhang, H. (2015). Treatment of organic pollutants using electro-Fenton and electro-Fenton-like process in aqueous solution. Progress in Chemistry, 27, 1123–1132.
Meyer, J., & Bester, K. (2004). Organophosphate flame retardants and plasticisers in wastewater treatment plants. Journal of Environmental Monitoring, 6, 599–605.
Petrie, B., Lopardo, L., Proctor, K., Youdan, J., Barden, R., & Kasprzyk-Hordern, B. (2019). Assessment of bisphenol-A in the urban water cycle. Science of the Total Environment, 650, 900–907.
Quan, X., Luo, D., Wu, J., Li, R., Cheng, W., & Ge, S. (2017). Ozonation of acid red 18 wastewater using O3/Ca(OH)2 system in a micro bubble gas-liquid reactor. Journal of Environmental Chemical Engineering, 5, 283–291.
Sehested, K., Holcman, J., Bjergbakke, E., & Hart, E. J. (1984). A pulse radiolytic study of the reaction hydroxyl + ozone in aqueous medium. The Journal of Physical Chemistry, 88, 4144–4147.
Sehested, K., Holcman, J., Bjergbakke, E., & Hart, E. J. (1987). Ozone decomposition in aqueous acetate solutions. Journal of Physical Chemistry, 91, 2359–2361.
Sehested, K., Corfitzen, H., Holcman, J., Fischer, C. H., & Hart, E. J. (1991). The primary reaction in the decomposition of ozone in acidic aqueous-solutions. Environmental Science & Technology, 25, 1589–1595.
Soliman, M. A., Pedersen, J. A., Park, H., Castaneda-Jimenez, A., Stenstrom, M. K., & Suffet, I. H. (2007). Human pharmaceuticals, antioxidants, and plasticizers in wastewater treatment plant and water reclamation plant effluents. Water Environment Research, 79, 156–167.
Takahashi, M., Chiba, K., & Li, P. (2007). Formation of hydroxyl radicals by collapsing ozone microbubbles under strongly acidic conditions. Journal of Physical Chemistry B, 111, 11443–11446.
Xia, Z., & Hu, L. (2019). Theoretical model for micro-nano-bubbles mass transfer during contaminant treatment. Journal of Environmental Engineering and Science, 14, 157–167.
Xiao, S., Cheng, M., Zhong, H., Liu, Z., Liu, Y., Yang, X., & Liang, Q. (2020). Iron-mediated activation of persulfate and peroxymonosulfate in both homogeneous and heterogeneous ways: a review. Chemical Engineering Journal, 384, 123265.
Xu, G., Zhang, L., Yu, W., Sun, Z., Guan, J., Zhang, J., Lin, J., Zhou, J., Fan, J., Murugadoss, V., & Guo, Z. (2020). Low optical dosage heating-reduced viscosity for fast and large-scale cleanup of spilled crude oil by reduced graphene oxide melamine nanocomposite adsorbents. Nanotechnology, 31, 225402.
Yang, C. F., Wang, C. C., & Chen, C. H. (2014). Di-n-butyl phthalate removal by strain Deinococcus sp R5 in batch reactors. International Biodeterioration & Biodegradation, 95, 55–60.
Yasuda, K., Matsushima, H., & Asakura, Y. (2019). Generation and reduction of bulk nanobubbles by ultrasonic irradiation. Chemical Engineering Science, 195, 455–461.
Yasui, K., Tuziuti, T., & Kanematsu, W. (2019). Mechanism of OH radical production from ozone bubbles in water after stopping cavitation. Ultrasonics Sonochemistry, 58, 104707.
Zhang, M., Feng, Y., Zhang, D., Dong, L., & Pan, X. (2019a). Ozone-encapsulated colloidal gas aphrons for in situ and targeting remediation of phenanthrene-contaminated sediment-aquifer. Water Research, 160, 29–38.
Zhang, M., Wang, Y., Wang, Y., Li, M., Zhang, D., Qiang, Z., & Pan, X. (2019b). Efficient elimination and regrowth inhibition of harmful bloom-forming cyanobacteria using surface-functionalized microbubbles. Water Research, 161, 473–485.
Funding
The project was funded by the Natural Science Foundation of China (nos. 21806101, 51476094, 51590901), the Natural Science Foundation of Shanghai (nos. 16ZR1412600, 15ZR1416900), the Gaoyuan Discipline of Shanghai-Environmental Science and Engineering (Resource Recycling Science and Engineering), the Shanghai Eastern Professorship grant, ShuGuang project supported by the Shanghai Municipal Education Commission and Shanghai Education Development Foundation (no. 15SG52).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wan, X., Zhang, L., Sun, Z. et al. Treatment of High Concentration Acid Plasticizer Wastewater by Ozone Microbubble Oxidation. Water Air Soil Pollut 231, 367 (2020). https://doi.org/10.1007/s11270-020-04735-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04735-3