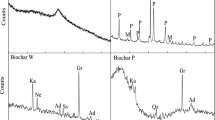
Abstract
Leonardite (LND), a low-grade coal, is an inexpensive material used to improve soil fertility in humic-deficient soils. Our objective was to quantify the ability of LND to immobilize atrazine (ATZ). To accomplish this, LND morphological properties, elemental composition, and spectroscopic properties were determined. We found that LND had a high organic matter content (40.53%, w/w) with a mesopore particle structure. Fourier-transform infrared spectroscopy spectral analysis confirmed the presence of aromatic molecules, all of which contributed to ATZ adsorption. The ATZ adsorption characteristics were quantified using batch adsorption kinetic and isotherm experiments. The ATZ adsorption kinetics were best described by a pseudo-second-order kinetic model while the concentrations of ATZ adsorbed (Cads) versus equilibrium concentrations (Ceq) had the best fit with the Freundlich equation compared with five other isotherms tested. The ability of LND to minimize ATZ leaching was tested using one-dimensional transport experiments with packing configurations and materials. The results revealed that up to 95% of ATZ was immobilized in the LND-amended soil. The extractable ATZ residues from both soil and LND corroborated the reduction in ATZ leaching. Overall, the adsorption results indicated that LND had a high affinity for ATZ adsorption and could reduce ATZ transport to the groundwater.

.





Similar content being viewed by others
References
Akinremi, O. O., Janzen, H. H., Lemke, R. L., & Larney, F. J. (2000). Response of canola, wheat and green beans to leonardite additions. Canadian Journal of Soil Science, 80(3), 437–443. https://doi.org/10.4141/S99-058.
Alekseeva, O. V., Rodionova, A. N., Bagrovskaya, N. A., z, A. V., & Noskov, A. V. (2019). Effect of the bentonite filler on structure and properties of composites based on hydroxyethyl cellulose. Arabian Journal of Chemistry, 12(3), 398–404. https://doi.org/10.1016/j.arabjc.2015.07.011.
ARO. (2018). Agricultural Regulatory Office, Report of imported agricultural hazardous material to Thailand in 2018. http://www.doa.go.th/ard/?page_id=386. Accessed 1 January 2020.
Ausavasukhi, A., Kampoosaen, C., & Kengnok, O. (2016). Adsorption characteristics of Congo red on carbonized leonardite. Journal of Cleaner Production, 134, 506–514. https://doi.org/10.1016/j.jclepro.2015.10.034.
Ayawei, N., Ebelegi, A. N., & Wankasi, D. (2017). Modelling and interpretation of adsorption isotherms. Journal of Chemistry, 2017, 11. https://doi.org/10.1155/2017/3039817.
Cavas, T. (2011). In vivo genotoxicity evaluation of atrazine and atrazine-based herbicide on fish Carassius auratus using the micronucleus test and the comet assay. Food Chemical Toxicology, 49(6), 1431–1435. https://doi.org/10.1016/j.fct.2011.03.038.
Cerejeira, M., Viana, P., Batista, S., Pereira, T., Silva, E., Valério, M., Silvaa, A., Ferreira, M., & Silva-Fernandesa, A. M. (2003). Pesticides in Portuguese surface and ground waters. Water Research, 37(5), 1055–1063. https://doi.org/10.1016/s0043-1354(01)00462-6.
Chammui, Y., Sooksamiti, P., Naksata, W., Thiansem, S., & Arqueropanyo, O.-A. (2014). Removal of arsenic from aqueous solution by adsorption on leonardite. Chemical Engineering Journal, 240, 202–210. https://doi.org/10.1016/j.cej.2013.11.083.
Demir, A., Ece, A., Filiz, D. B. U., & Y. (2019). A new screening index for pesticides leachability to groundwater. Journal of Environmental Management, 231, 1193–1202. https://doi.org/10.1016/j.jenvman.2018.11.007.
Ece, A., Saltali, K., Eryigit, N., & Uysal, F. (2007). The effects of leonardite applications on climbing bean (Phaseolus vulgaris L.) yield and the some soil properties. Journal of Agronomy, 6, 480–483.
Graymore, M., Stagnitti, F., & Allinson, G. (2001). Impacts of atrazine in aquatic ecosystems. Environment International, 26(7), 483–495. https://doi.org/10.1016/S0160-4120(01)00031-9.
Hamdaoui, O., & Naffrechoux, E. (2007). Modeling of adsorption isotherms of phenol and chlorophenols onto granular activated carbon: Part I. two-parameter models and equations allowing determination of thermodynamic parameters. Journal of Hazardous Materials, 147(1), 381–394. https://doi.org/10.1016/j.jhazmat.2007.01.021.
Helal, A. A., Murad, G. A., & Helal, A. A. (2011). Characterization of different humic materials by various analytical techniques. Arabian Journal of Chemistry, 4(1), 51–54. https://doi.org/10.1016/j.arabjc.2010.06.018.
Hildebrandt, A., Guillamón, M., Lacorte, S., Tauler, R., & Barceló, D. (2008). Impact of pesticides used in agriculture and vineyards to surface and groundwater quality (North Spain). Water Research, 42(13), 3315–3326. https://doi.org/10.1016/j.watres.2008.04.009.
Ho, Y.-S. (2006). Isotherms for the sorption of lead onto peat: Comparison of linear and non-linear methods. Polish Journal of Environmental Studies, 15(1), 81–86.
Huang, Y., Liu, Z., He, Y., & Li, Y. (2015). Impact of soil primary size fractions on sorption and desorption of atrazine on organo-mineral fractions. Environmental Science and Pollution Research, 22(6), 4396–4405. https://doi.org/10.1007/s11356-014-3684-z.
Jantawongsri, K., Thammachoti, P., Kitana, J., Khonsue, W., Varanusupakul, P., & Kitana, N. (2015). Altered immune response of the rice frog Fejervarya limnocharis living in agricultural area with intensive herbicide utilization at Nan Province, Thailand. Environment Asia, 8, 68–73. https://doi.org/10.14456/ea.2015.9.
Jovanovic, D. S. (1969). Physical adsorption of gases I: Isotherms for monolayer and multilayer adsorption. Colloid and Polymer Science, 235, 1203.
Kananizadeh, N., Chokejaroenrat, C., Li, Y., & Comfort, S. (2015). Modeling improved ISCO treatment of low permeable zones via viscosity modification: Assessment of system variables. Journal of Contaminant Hydrology, 173, 25–37. https://doi.org/10.1016/j.jconhyd.2014.11.009.
Kaplan, D. I. (2003). Influence of surface charge of an Fe-oxide and an organic matter dominated soil on iodide and pertechnetate sorption. Radiochimica Acta, 91, 173.
Khashirova, S. Y., Musaev, Y. I., Mikitaev, A. K., Malkanduev, Y. A., & Ligidov, M. K. (2009). Hybrid nanocomposites based on guanidine methacrylate monomer and polymer and layered aluminosilicates: Synthesis, structure, and properties. Polymer Science Series B, 51(9), 377. https://doi.org/10.1134/S1560090409090085.
Ladu, J., Ladu, C., & Zhang, D.-R. (2011). Modeling atrazine transport in soil columns with HYDRUS-1D. Water Science and Engineering, 4, 258–269. https://doi.org/10.3882/j.issn.1674-2370.2011.03.003.
Lei, Z., Ye, C., & Wang, X. (2001). Hydrolysis kinetics of atrazine and influence factors. Journal of Environmental Sciences (China), 13, 99–103.
Lian, F., Sun, B., Song, Z., Zhu, L., Qi, X., & Xing, B. (2014). Physicochemical properties of herb-residue biochar and its sorption to ionizable antibiotic sulfamethoxazole. Chemical Engineering Journal, 248, 128–134. https://doi.org/10.1016/j.cej.2014.03.021.
Limousin, G., Gaudet, J. P., Charlet, L., Szenknect, S., Barthès, V., & Krimissa, M. (2007). Sorption isotherms: A review on physical bases, modeling and measurement. Applied Geochemistry, 22(2), 249–275. https://doi.org/10.1016/j.apgeochem.2006.09.010.
Luo, Z., Gao, M., Ye, Y., & Yang, S. (2015). Modification of reduced-charge montmorillonites by a series of Gemini surfactants: Characterization and application in methyl orange removal. Applied Surface Science, 324, 807–816. https://doi.org/10.1016/j.apsusc.2014.11.043.
Lupul, I., Yperman, J., Carleer, R., & Gryglewicz, G. (2015). Adsorption of atrazine on hemp stem-based activated carbons with different surface chemistry. Adsorption, 21(6–7), 489–498. https://doi.org/10.1007/s10450-015-9689-1.
Manzotti, F., & dos Santos, O. A. A. (2019). Evaluation of removal and adsorption of different herbicides on commercial organophilic clay. Chemical Engineering Communications, 206(11), 1526–1543. https://doi.org/10.1080/00986445.2019.1601626.
Meng, F., Yuan, G., Larson, S., Ballard, J., Waggoner, C., Arslan, Z., & Han, F. X. (2017). Removing uranium (VI) from aqueous solution with insoluble humic acid derived from leonardite. Journal of Environmental Radioactivity, 180, 1–8. https://doi.org/10.1016/j.jenvrad.2017.09.019.
Mohamed, I., Zhang, G.-S., Li, Z.-G., Liu, Y., Chen, F., & Dai, K. (2015). Ecological restoration of an acidic cd contaminated soil using bamboo biochar application. Ecological Engineering, 84, 67–76. https://doi.org/10.1016/j.ecoleng.2015.07.009.
Mudhoo, A., & Garg, V. K. (2011). Sorption, transport and transformation of atrazine in soils, minerals and composts: a review. Pedosphere, 21(1), 11–25. https://doi.org/10.1016/S1002-0160(10)60074-4.
Murphy, I. J., & Coats, J. R. (2011). The capacity of switchgrass (Panicum virgatum) to degrade atrazine in a phytoremediation setting. Environmental Toxicology and Chemistry, 30(3), 715–722. https://doi.org/10.1002/etc.437.
Nasseri, S., Dehghani, M., Amin, S., Naddafi, K., & Zamanian, Z. (2009). Fate of atrazine in the agricultural soil of corn fields in Fars province of Iran. Iranian Journal of Environmental Health Science & Engineering, 6, 223–232.
Ou, L., Gannon, T. W., Arellano, C., & Polizzotto, M. L. (2018). A global meta-analysis to predict atrazine sorption from soil properties. Journal of Environmental Quality, 47(6), 1389–1399. https://doi.org/10.2134/jeq2017.11.0429.
Ouypornkonchakorn, S., & Klamtet, J. (2019). Adsorption of cadmium from aqueous solution using leonardite-bentonite ceramic as adsorbent. NU International Journal of Science, 16(1), 12–23.
Panuwet, P., Siriwong, W., Prapamontol, T., Ryan, P., Fiedler, N., Robson, M., & Barr, D. B. (2012). Agricultural pesticide Management in Thailand: Situation and population health risk. Environmental Science & Policy, 17, 72–81. https://doi.org/10.1016/j.envsci.2011.12.005.
Park, Y., Sun, Z., Ayoko, G., & Frost, R. (2014). Bisphenol a sorption by organo-montmorillonite: Implications for the removal of organic contaminants from water. Chemosphere, 107, 249–256. https://doi.org/10.1016/j.chemosphere.2013.12.050.
Pérez-de-Mora, A., Madejón, P., Burgos, P., Cabrera, F., Lepp, N. W., & Madejón, E. (2011). Phytostabilization of semiarid soils residually contaminated with trace elements using by-products: Sustainability and risks. Environmental Pollution, 159(10), 3018–3027. https://doi.org/10.1016/j.envpol.2011.04.015.
Petrov, D., Tunega, D., Gerzabek, M. H., & Oostenbrink, C. (2017). Molecular dynamics simulations of the standard leonardite humic acid: Microscopic analysis of the structure and dynamics. Environmental Science & Technology, 51(10), 5414–5424. https://doi.org/10.1021/acs.est.7b00266.
Prata, F. B., Lavorenti, A., Vanderborght, J., Burauel, P., & Vereecken, H. (2003). Miscible displacement, sorption and desorption of atrazine in a Brazilian Oxisol. Vadose Zone Journal, 2(4), 728–738. https://doi.org/10.2113/2.4.728.
Pusino, A., Fiori, M. G., Braschi, I., & Gessa, C. (2003). Adsorption and desorption of triasulfuron by soil. Journal of Agricultural and Food Chemistry, 51(18), 5350–5354. https://doi.org/10.1021/jf030105w.
Rambabu, N., Guzman, C., Soltan, J., & Vurimindi, H. (2012). Adsorption characteristics of atrazine on granulated activated carbon and carbon nanotubes. Chemical Engineering & Technology, 35, 272–280. https://doi.org/10.1002/ceat.201100376.
Saikia, B. J., Parthasarathy, G., & Sarmah, N. C. (2008). Fourier transform infrared spectroscopic estimation of crystallinity in SiO2 based rocks. Bulletin of Materials Science, 31(5), 775–779. https://doi.org/10.1007/s12034-008-0123-0.
Sakulthaew, C., Chokejaroenrat, C., Poapolathep, A., Satapanajaru, T., & Poapolathep, S. (2017). Hexavalent chromium adsorption from aqueous solution using carbon nano-onions (CNOs). Chemosphere, 184, 1168–1174. https://doi.org/10.1016/j.chemosphere.2017.06.094.
Sass, J. B., & Colangelo, A. (2006). European Union bans atrazine, while the United States negotiates continued use. International Journal of Occupational and Environmental Health, 12(3), 260–267. https://doi.org/10.1179/oeh.2006.12.3.260.
Sayjumpa, J., Jomhataikool, B., Faungnawakij, K., Kuboon, S., Kraithong, W., Fuji, M., & Eiad-ua, A. (2019). Porous carbon adsorbent from humin derived from thai leonardite for methylene blue dye adsorption. Current Applied Science and Technology, 19, 1–8. https://doi.org/10.14456/cast.2019.1.
Stratton, G. (1984). Effects of the herbicide atrazine and its degradation products, alone and in combination, on phototrophic microorganisms. Archives of Environmental Contamination and Toxicology, 13, 35–42. https://doi.org/10.1007/BF01055644.
Tan, I. A. W., Ahmad, A. L., & Hameed, B. H. (2009). Adsorption isotherms, kinetics, thermodynamics and desorption studies of 2,4,6-trichlorophenol on oil palm empty fruit bunch-based activated carbon. Journal of Hazardous Materials, 164(2), 473–482. https://doi.org/10.1016/j.jhazmat.2008.08.025.
Tang, Z., Zhang, W., & Chen, Y. (2009). Adsorption and desorption characteristics of monosulfuron in Chinese soils. Journal of Hazardous Materials, 166(2), 1351–1356. https://doi.org/10.1016/j.jhazmat.2008.12.052.
Wang, B., Zeng, D., Chen, Y., Belzile, N., Bai, Y., Zhu, J., et al. (2019). Adsorption behaviors of phenanthrene and bisphenol A in purple paddy soils amended with straw-derived DOM in the West Sichuan Plain of China. Ecotoxicology and Environmental Safety, 169, 737–746. https://doi.org/10.1016/j.ecoenv.2018.11.096.
Yuan, B., Liang, S., Jin, Y.-X., Zhang, M.-J., Zhang, J.-B., & Kim, N.-H. (2017). Toxic effects ofatrazine on porcine oocytes and possible mechanisms of action. PLoS One, 12(6), e0179861. https://doi.org/10.1371/journal.pone.0179861.
Zeledón-Toruño, Z. C., Lao-Luque, C., de las Heras, F. X. C., & Sole-Sardans, M. (2007). Removal of PAHs from water using an immature coal (leonardite). Chemosphere, 67(3), 505–512. https://doi.org/10.1016/j.chemosphere.2006.09.047.
Zhang, W., Zheng, J., Zheng, P., & Qiu, R. (2015). Atrazine immobilization on sludge derived biochar and the interactive influence of coexisting Pb(II) or Cr(VI) ions. Chemosphere, 134, 438–445. https://doi.org/10.1016/j.chemosphere.2015.05.011.
Acknowledgments
Our appreciation is expressed to Associate Professor Dr. Artit Ausavasukhi from the Applied Chemistry Department, Rajamangala University of Technology Isan, for valuable guidance regarding the leonardite material and Dr. Kanitta Wongyai, Level 10th Scientist from the Electricity Generating Authority of Thailand (EGAT) at the Mae Moh lignite mine, for providing the leonardite for our research. The authors also thank Mrs. Sunee Rittirat, the owner of the agricultural farm, for granting access to collect soil samples and Ms. Kornkanok Jutarvutikul for creating our graphical abstract.
Funding
This research was funded by the Suranaree University Technology Research Fund fiscal year 2016 (SUT7-715-59-12-39) and was partially funded by the Environmental Technology for Emerging Issue Management Research Unit under supporting funds from the Faculty of Environment, Kasetsart University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 1097 kb)
Rights and permissions
About this article
Cite this article
Chokejaroenrat, C., Watcharenwong, A., Sakulthaew, C. et al. Immobilization of Atrazine Using Oxidized Lignite Amendments in Agricultural Soils. Water Air Soil Pollut 231, 249 (2020). https://doi.org/10.1007/s11270-020-04608-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04608-9