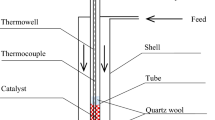
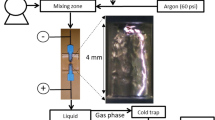
Abstract
Effects of discharge power, O2 content, reaction temperature, catalyst introduction, and presence of NO and dichloromethane (DCM) on the formation of nitrogen oxides (N2O, NO, and NO2) by discharge in N2-O2 mixture have been systematically investigated using a dielectric barrier discharge (DBD) reactor. Results show that discharge in N2-O2 mixture always produces several to hundreds ppm of nitrogen oxides as byproducts. The production of nitrogen oxides increases with the increase of O2 content and the introduction of Al2O3 or RuO2/Al2O3 catalyst. N2O production first increases and then decreases/levels off with increasing discharge power at room temperature, but increases monotonously at 300 °C. NO and NO2 are produced only at relatively high discharge power at room temperature but are produced at all discharge power tested at 300 °C. Increasing the reaction temperature from room temperature to 300 °C significantly reduces the production of N2O but increases that of NO and NO2. The presence of hundreds ppm NO in N2-O2 mixture significantly reduces the production of N2O due to the effective quenching of the vital species for N2O formation (N2(A3Σu+)) by NO. The presence of hundreds ppm DCM, however, hardly affects the production of nitrogen oxides, demonstrating the precedence of nitrogen oxide production over DCM decomposition in N2-O2 plasma.






Similar content being viewed by others
References
Allah, Z. A., Whitehead, J. C., & Martin, P. (2014). Remediation of dichloromethane (CH2Cl2) using non-thermal, atmospheric pressure plasma generated in a packed-bed reactor. Environmental Science & Technology, 48(1), 558–565.
Bai, Y. H., Chen, J. R., Li, X. Y., & Zhang, C. H. (2009). Non-thermal plasmas chemistry as a tool for environmental pollutants abatement. Reviews of Environmental Contamination & Toxicology, 201, 117–136.
Chang, J. S. (2008). Physics and chemistry of plasma pollution control technology. Plasma Sources Science & Technology, 17(4), 045004.
Chen, H. L., Lee, H. M., Chen, S. H., Chang, M. B., Yu, S. J., & Li, S. N. (2009). Removal of volatile organic compounds by single-stage and two-stage plasma catalysis systems: a review of the performance enhancement mechanisms, current status, and suitable applications. Environmental Science & Technology, 43(7), 2216–2227.
Durme, J. V., Dewulf, J., Leys, C., & Langenhove, H. V. (2008). Combining non-thermal plasma with heterogeneous catalysis in waste gas treatment: a review. Applied Catalysis B: Environmental, 78(3–4), 324–333.
Fan, X., Kang, S. J., Li, J., & Zhu, T. L. (2018). Conversion of dilute nitrous oxide (N2O) in N2 and N2-O2 mixtures by plasma and plasma-catalytic processes. RSC Advances, 8, 26998–27007.
Fan, X., Zhu, T. L., Wang, M. Y., & Li, X. M. (2009). Removal of low-concentration BTX in air using a combined plasma catalysis system. Chemosphere, 75(10), 1301–1306.
Fan, X., Zhu, T. L., Sun, Y. F., & Yan, X. (2011). The roles of various plasma species in the plasma and plasma-catalytic removal of low-concentration formaldehyde in air. Journal of Hazardous Materials, 196, 380–385.
Fan, X., Zhu, T. L., Wan, Y. J., & Yan, X. (2010). Effects of humidity on the plasma-catalytic removal of low-concentration BTX in air. Journal of Hazardous Materials, 180(1), 616–621.
Fitzsimmons, C., Ismail, F., Whitehead, J. C., & Wilman, J. J. (2000). The chemistry of dichloromethane destruction in atmospheric-pressure gas streams by a dielectric packed-bed plasma reactor. The Journal of Physical Chemistry A, 104(25), 6032–6038.
Futamura, S., Zhang, A., Einaga, H., & Kabashima, H. (2002). Involvement of catalyst materials in nonthermal plasma chemical processing of hazardous air pollutants. Catalysis Today, 72(3–4), 259–265.
Guan, B., Lin, H., Cheng, Q., & Huang, Z. (2011). Removal of NOx with selective catalytic reduction based on nonthermal plasma preoxidation. Industrial & Engineering Chemistry Research, 50(9), 5401–5413.
Herron, J. T., & Green, D. S. (2001). Chemical kinetics database and predictive schemes for nonthermal humid air plasma chemistry. Part II. Neutral species reactions. Plasma Chemistry and Plasma Processing, 21(3), 459–481.
Jiang, N., Guo, L., Qiu, C., Zhang, Y., Shang, K., Lu, N., Li, J., & Wu, Y. (2018). Reactive species distribution characteristics and toluene destruction in the three-electrode DBD reactor energized by different pulsed modes. Chemical Engineering Journal, 350, 12–19.
Jiang, N., Qiu, C., Guo, L., Shang, K., Lu, N., Li, J., & Wu, Y. (2017). Post plasma-catalysis of low concentration VOC over alumina-supported silver catalysts in a surface/packed-bed hybrid discharge reactor. Water, Air, & Soil Pollution, 228(3), 113.
Jo, J., Trinh, Q. H., Kim, S. H., & Mok, Y. S. (2018). Plasma-catalytic decomposition of nitrous oxide over γ-alumina-supported metal oxides. Catalysis Today, 310, 42–48.
Jõgi, I., Erme, K., Levoll, E., Raud, J., & Stamate, E. (2018). Plasma and catalyst for the oxidation of NOx. Plasma Sources Science & Technology, 27(3), 035001.
Kim, H. H. (2004). Nonthermal plasma processing for air-pollution control: a historical review, current issues, and future prospects. Plasma Processes & Polymers, 1(2), 91–110.
Kim, H. H., Oh, S. M., Ogata, A., & Futamura, S. (2005). Decomposition of gas-phase benzene using plasma-driven catalyst (PDC) reactor packed with Ag/TiO2 catalyst. Applied Catalysis B: Environmental, 56(3), 213–220.
Kossyi, I. A., Kostinsky, A. Y., Matveyev, A. A., & Silakov, V. P. (1992). Kinetic scheme of the non-equilibrium discharge in nitrogen-oxygen mixtures. Plasma Sources Science & Technology, 1(3), 207–220.
Krawczyk, K. (2009). Conversion of nitrous oxide by positive pulsed corona discharge. IEEE Transactions on Plasma Science, 37(6), 884–889.
Lee, H. M., & Chang, M. B. (1998). Destruction of VOCs via silent discharge plasmas. Chemical Engineering & Technology, 21(12), 987–989.
Mok, Y. S., & Huh, Y. J. (2005). Simultaneous removal of nitrogen oxides and particulate matters from diesel engine exhaust using dielectric barrier discharge and catalysis hybrid system. Plasma Chemistry and Plasma Processing, 25(6), 625–639.
Oda, T. (2003). Non-thermal plasma processing for environmental protection: decomposition of dilute VOCs in air. Journal of Electrostatics, 57(3–4), 293–311.
Pekridis, G., Athanasiou, C., Konsolakis, M., Yentekakis, I. V., & Marnellos, G. E. (2009). N2O abatement over γ-Al2O3 supported catalysts: effect of reducing agent and active phase nature. Topics in Catalysis, 52(13), 1880–1887.
Schiavon, M., Torretta, V., Casazza, A., & Ragazzi, M. (2017). Non-thermal plasma as an innovative option for the abatement of volatile organic compounds: a review. Water, Air, & Soil Pollution, 228(10), 388.
Song, Y. H., Kim, S. J., Choi, K. I., & Yamamoto, T. (2002). Effects of adsorption and temperature on a nonthermal plasma process for removing VOCs. Journal of Electrostatics, 55, 189–201.
Tang, X., Wang, J., Yi, H., Zhao, S., Gao, F., Huang, Y., Zhang, R., & Yang, Z. (2017). N2O formation characteristics in dielectric barrier discharge reactor for environmental application: effect of operating parameters. Energy & Fuels, 31(12), 13901–13908.
Trinh, H. Q., & Mok, Y. S. (2014). Plasma-catalytic oxidation of acetone in annular porous monolithic ceramic-supported catalysts. Chemical Engineering Journal, 251, 199–206.
Trinh, Q., Kim, S. H., & Mok, Y. S. (2016). Removal of dilute nitrous oxide from gas streams using a cyclic zeolite adsorption-plasma decomposition process. Chemical Engineering Journal, 302, 12–22.
Urashima, K., & Chang, J. S. (2000). Removal of volatile organic compounds from air streams and industrial flue gases by non-thermal plasma technology. IEEE Transactions on Dielectrics & Electrical Insulation, 7(5), 602–614.
Wan, Y. J., Fan, X., & Zhu, T. L. (2011). Removal of low-concentration formaldehyde in air by DC corona discharge plasma. Chemical Engineering Journal, 171(1), 314–319.
Wang, P., & Chen, J. (2009). Numerical modelling of ozone production in a wire-cylinder corona discharge and comparison with a wire-plate corona discharge. Journal of Physics D: Applied Physics, 42(3), 035202.
Yamamoto, T. (1997). VOC decomposition by nonthermal plasma processing—a new approach. Journal of Electrostatics, 42(1–2), 227–238.
Zhang, Y., Tang, X., Yi, H., Yu, Q., Wang, J., Gao, F., Gao, Y., Li, D., & Cao, Y. (2016). The byproduct generation analysis of the NOx conversion process in dielectric barrier discharge plasma. RSC Advances, 6(68), 63946–63953.
Zhao, G. B., Garikipati, S., Hu, X. D., Argyle, M. D., & Radosz, M. (2005). Effect of oxygen on nonthermal plasma reactions of nitrogen oxides in nitrogen. AICHE Journal, 51(6), 1800–1812.
Zhao, G. B., Hu, X. D., Argyle, M. D., & Radosz, M. (2004). N atom radicals and N2(A3Σu +) found to be responsible for nitrogen oxides conversion in nonthermal nitrogen plasma. Industrial & Engineering Chemistry Research, 43(17), 5077–5088.
Zhu, X., Gao, X., Zheng, C., Wang, Z., Ni, M., & Tu, X. (2014). Plasma-catalytic removal of a low concentration of acetone in humid conditions. RSC Advances, 4(71), 37796–37805.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 21707004, 51638001) and the Natural Science Foundation of Beijing Municipality (grant number 8152011).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic Supplementary Material
ESM 1
(DOCX 757 kb)
Rights and permissions
About this article
Cite this article
Fan, X., Kang, S., Li, J. et al. Formation of Nitrogen Oxides (N2O, NO, and NO2) in Typical Plasma and Plasma-Catalytic Processes for Air Pollution Control. Water Air Soil Pollut 229, 351 (2018). https://doi.org/10.1007/s11270-018-4011-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-018-4011-y