Abstract
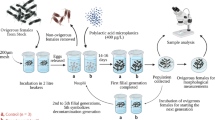
The impact of nanoparticles (NPs) in zooplankton is poorly studied, particularly when organisms are exposed through diet. Food, constituted mainly by unicellular algae, can act as an important route of contamination for zooplankton. Since unicellular algae have a high surface area in relation to their volume, NPs can interact with their cell membranes and walls, as well as with exopolysaccharides secreted by them. In the present research, we investigated both the acute effects of waterborne titanium dioxide nanoparticles (TiO2 NPs), and its chronic effects via dietary exposure on the Neotropical freshwater zooplankton Ceriodaphnia silvestrii Daday, 1902 (Crustacea: Cladocera). The observed acute effects served as support for chronic tests, in which we investigated the effects of TiO2 NPs on survival and life history parameters (body length, numbers of eggs, and neonates produced) of cladoceran adult females, using the freshwater cosmopolitan chlorophycean Raphidocelis subcapitata as source of contamination of TiO2 NPs for zooplankton. R. subcapitata cells were exposed to concentrations of 0, 0.01, 1, and 10 mg L−1 of TiO2 NPs for 96 h, and then provided as food for females of C. silvestrii until the third brood was released. Significant toxic effects were observed in body length and total number of neonates and eggs produced by females of C. silvestrii at concentrations of 1 and 10 mg L−1 of TiO2 NPs. Survival was the most sensitive parameter when exposure was given via food. From the concentration of 0.01 mg L−1 of TiO2 NPs, there was a decrease in the survival of C. silvestrii females. The quantification of TiO2 NPs in algae evidenced that they have retained NPs in their cells, being, therefore, an important route of exposure and toxicity of TiO2 NPs to the studied microcrustacean.








Similar content being viewed by others
References
ABNT, Associação Brasileira de Normas Técnicas (2009) NBR 12713. Ecotoxicologia aquática – toxicologia aguda – método de ensaio com Daphnia spp (Crustacea, Cladocera). Rio de Janeiro, 23 p.
ABNT, Associação Brasileira de Normas Técnicas. (2010). NBR 13373. Ecotoxicologia aquática – toxicidade crônica – método de ensaio com Ceriodaphnia spp (Crustacea, Cladocera). Rio de Janeiro, 18 p.
ABNT, Associação Brasileira de Normas Técnicas. (2011). NBR 12648. Ecotoxicologia aquática – toxicidade crônica – método de ensaio com algas (Chlorophyceae). Rio de Janeiro, 24 p.
AFNOR, Association Française Normalisation. (1980). Norme experimentale. T90–304. Essais deseaux Determination de I’inhibition de Scenesdesmus subspicatus par une substance. Paris.
Allen, N. S., Edge, M., Verran, J., Caballero, L., Abrusci, C., Stratton, J., Maltby, J., & Bygott, C. (2009). Photocatalytic surfaces: environmental benefits of nanotitania. The Open Materials Science Journal, 3, 6–27. https://doi.org/10.2174/1874088X00903010006.
Amiano, I., Olabarrieta, J., Vitorica, J., & Zorita, S. (2012). Acute toxicity of nanosized TiO2 to Daphnia magna under UVA irradiation. Environmental Toxicology and Chemistry, 31(1), 2564–2566. https://doi.org/10.1002/etc.1981.
Aschberger, K., Micheletti, C., Sokull-Klöttgen, B., & Christensen, F. M. (2011). Analysis of currently available data for characterising the risk of engineered nanomaterials to the environment and human health—lessons learned from four case studies. Environment International, 37, 1143–1156. https://doi.org/10.1016/j.envint.2011.02.005.
ASTM, American Society for Testing and Materials. (2001). Standard guide for conducting acute toxicity testing on test materials with fishes, macroinvertebrates, and amphibians. West Conshohocken, pp. E729-E796.
Ates, M., Daniels, J., Arslan, Z., & Farah, I. O. (2013). Effects of aqueous suspensions of titanium dioxide nanoparticles on Artemia salina: assessment of nanoparticle aggregation, accumulation, and toxicity. Environmental Monitoring and Assessment, 185, 3339–3348. https://doi.org/10.1007/s10661-012-2794-7.
Ayres, M., Ayres Júnior, M., Ayres, D. L., & Santos, A. A. (2007). BIOESTAT—Aplicações estatísticas nas áreas das Ciências Bio-Médicas (364 p). Belém: Mamirauá.
Baker, T. J., Tyler, C. R., & Galloway, T. S. (2013). Impacts of metal and metal oxide nanoparticles on marine organisms. Environmental Pollution, 186, 257–271. https://doi.org/10.1016/j.envpol.2013.11.014.
Barreto GJ, Silva VM (2015) Como interpretar os dados do Zetasizer Nano: análise de potencial zeta. Freely accessible via: <http://www.malvern.com/br/events>. Accessed on: 10 November, 2016.
Barreto, D. M., & Lombardi, A. T. (2016). Environmentally relevant concentrations of TiO2 nanoparticles affected cell viability and photosynthetic yield in the chlorophyceae Scenedesmus bijugus. Water, Air, & Soil Pollution, 227, 450. https://doi.org/10.1007/s11270-016-3139-x.
Baveye, P., & Laba, M. (2008). Aggregation and toxicology of titanium dioxide nanoparticles. Environmental Health Perspectives, 116, A152. https://doi.org/10.1289/ehp10915.
Brayner, R., Dahoumane, S. A., Yéprémian, C., Djediat, C., Meyer, M., Couté, A., & Fiévet, F. (2010). ZnO nanoparticles: synthesis, characterization, and ecotoxicological studies. Langmuir, 26(9), 6522–6528. https://doi.org/10.1021/la100293s.
Bundschuh, M., Seitz, F., Rosenfeldt, R. R., & Schulz, R. (2012). Titanium dioxide nanoparticles increase sensitivity in the next generation of the water flea Daphnia magna. PLoS ONE, 7(11), e48956. https://doi.org/10.1371/journal.pone.0048956.
Bundschuh, M., Vogt, R., Seitz, F., & Rosenfeldt, R. R. (2016). Do titanium dioxide nanoparticles induce food depletion for filter feeding organisms? A case study with Daphnia magna. Environmental Pollution, 214, 840–846. https://doi.org/10.1016/j.envpol.2016.04.092.
Campos, B., Rivetti, C., Rosenkranz, P., Navas, J. M., & Barata, C. (2013). Effects of nanoparticles of TiO2 on food depletion and life-history responses of Daphnia magna. Aquatic Toxicology, 130-131, 174–183. https://doi.org/10.1016/j.aquatox.2013.01.005.
Canesi, L., Ciacci, C., & Balbi, T. (2015). Interactive effects of nanoparticles with other contaminants in aquatic organisms: Friend or foe? Marine Environmental Research, 111, 128–134. https://doi.org/10.1016/j.marenvres.2015.03.010.
Casali-Pereira, M. P., Daam, M. A., Resende, J. C., Vasconcelos, A. M., Espíndola, E. L. G., & Botta, C. M. R. (2015). Toxicity of Vertimec 18 EC (active ingredient abamectin) to the neotropical cladoceran Ceriodaphnia silvestrii. Chemosphere (Oxford), 139, 558–564. https://doi.org/10.1016/j.chemosphere.2015.08.006.
Chen, J., Li, H., Han, X., & Wei, X. (2015). Transmission and accumulation of nano-TiO2 in a 2-step food chain (Scenedesmus obliquus to Daphnia magna). Bulletin of Environmental Contamination and Toxicology, 95(25), 145–149. https://doi.org/10.1007/s00128-015-1580-y.
Cherchi, C., Miljkovic, M., Diem, M., & Gu, A. Z. (2015). nTiO2 induced changes in intracellular composition and nutrient stoichiometry in primary producer e cyanobacteria. Science of the Total Environment, 512-513, 345–352. https://doi.org/10.1016/j.scitotenv.2015.01.037.
Clément, L., Hurel, C., & Marmier, N. (2013). Toxicity of TiO2 nanoparticles to cladocerans, algae, rotifers and plants—effects of size and crystalline structure. Chemosphere, 90, 1083–1090. https://doi.org/10.1016/j.chemosphere.2012.09.013.
Cupi, N., Hartmann, N. B., & Baun, A. (2016). Influence of pH and media composition on suspension stability of silver, zinc oxide, and titanium dioxide nanoparticles and immobilization of Daphnia magna under guideline testing conditions. Ecotoxicology and Environmental Safety, 127, 144–152. https://doi.org/10.1016/j.ecoenv.2015.12.028.
Dabrunz, A., Duester, L., Prasse, C., Seitz, F., Rosenfeldt, R., Schilde, C., Schaumann, G. E., & Schulz, R. (2011). Biological surface coating and molting inhibition as mechanisms of TiO2 nanoparticle toxicity Daphnia magna. PLoS ONE, 6(5), e20112. https://doi.org/10.1371/journal.pone.0020112.
Dalai, S., Iswarya, V., Bhuvaneshwari, M., Pakrashi, S., Chandrasekaran, N., & Mukherjee, A. (2014). Different modes of TiO2 uptake by Ceriodaphnia dubia: relevance to toxicity and bioaccumulation. Aquatic Toxicology, 152, 139–146. https://doi.org/10.1016/j.aquatox.2014.04.002.
de Lucca, G.M. (2016). Ecotoxicological effects of titanium dioxide nanoparticles on the microalga Raphidocelis subcapitata and cladoceran Ceriodaphnia silvestrii by different exposure routes. Master's dissertation, Postgraduate Program in Ecology and Natural Resources, Federal University of São Carlos. 148 p.
Environmental Canada. (1992). Biological test methods: test of reproduction and survival using the cladoceran Ceriodaphnia dubia. Report EPS 1/RM/21. Otawa: Environmental Canada Conservation and Protection 72 p.
Fan, W., Liu, L., Peng, R., & Wang, W. X. (2016). High bioconcentration of titanium dioxide nanoparticles in Daphnia magna determined by kinetic approach. Science of the Total Environment, 569-570, 1224–1231. https://doi.org/10.1016/j.scitotenv.2016.06.197.
Fouqueray, M., Dufils, B., Vollat, B., Chaurand, P., Botta, C., Abacci, K., Labilli, J., Rose, J., & Garric, J. (2012). Effects of aged TiO2 nanomaterial from sunscreen on Daphnia magna exposed by dietary route. Environmental Pollution, 163, 55–61. https://doi.org/10.1016/j.envpol.2011.11.035.
Gliwicz, Z. M., & Boavida, M. J. (1996). Clutch size and body size at first reproduction in Daphnia pulcaria at different levels of food and predation. Journal of Plankton Research, 18(6), 863–880. https://doi.org/10.1093/plankt/18.6.863.
Green, J. (1956). Growth, size and reproduction in Daphnia (Crustacea: Cladocera). Journal of Zoology, 126, 173–204. https://doi.org/10.1111/j.1096-3642.1956.tb00432.x.
Griffitt, R. J., Luo, J., Gao, J., Bonzongo, J. C., & Barber, D. S. (2008). Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environmental Toxicology and Chemistry, 27(9), 1972–1978. https://doi.org/10.1897/08-002.1.
Hall, S., Bradley, T., Moore, J. T., & Kuykindall, L. (2009). Acute and chronic toxicity of nano-scale TiO2 particles to freshwater fish, cladocerans, and green algae, and effects of organic and inorganic substrate on TiO2 toxicity. Nanotoxicology, 3, 91–97. https://doi.org/10.1080/17435390902788078.
Handy, R. D., Alen, H. E., Drevnick, P. E., Gorsuch, J. W., Green, A. S., Haldorsen, A. K. L., Hook, S. E., Mount, D. R., & Stubblefield, W. A. (2005). Toxic effects of dietborne metals: Laboratory studies. In J. S. Meyer, W. J. Adams, K. V. Brix, S. N. Luoma, D. R. Mount, W. A. Stubblefield, & C. M. Wood (Eds.), Toxicity of dietborne metals to aquatic organisms. Soc. Environ. Toxicol. Chem (Vol. 291, pp. 59–98). Pensacola: SETAC press.
Hartmann, N. B., Kammer, F. V., Hofmann, T., Baalousha, M., Ottofuelling, S., & Baun, A. (2010). Algal testing of titanium dioxide nanoparticles-testing considerations, inhibitory effects and modification of cadmium biovailability. Toxicology, 269, 190–197. https://doi.org/10.1016/j.tox.2009.08.008.
Iswarya, V., Bhuvaneshware, M., Chandrasekaran, N., & Mukheryee, A. (2016). Individual and binary toxicity of anatase and rutile nanoparticles towards Ceriodaphnia dubia. Aquatic Toxicology, 178, 209–221. https://doi.org/10.1016/j.aquatox.2016.08.007.
Jacobasch, C., Völker, C., Giebner, S., Völker, J., Alsenz, H., Potouridis, T., Heidenreich, H., Kayser, G., Oehlmann, J., & Oetken, M. (2014). Long-term effects of nanoscaled titanium dioxide on the cladoceran Daphnia magna over six generations. Environmental Pollution, 186, 180–186. https://doi.org/10.1016/j.envpol.2013.12.008.
Jaconeti PCM (2005) Validação de ensaios ecotoxicológicos com organismos autóctones Daphnia laevis e Ceriodaphnia silvestrii. Dissertation. Ipen: Autarquia associada a Universidade de São Paulo.
Ji, J., Long, Z., & Lin, D. (2011). Toxicity of oxide nanoparticles to the green algae Chlorella sp. Chemical Engineering Journal, 170, 525–530 https://doi.org/10.1016/j.cej.2010.11.026.
Ju-Nam, Y., Lead, J.R. (2008). Manufactured nanoparticles: An overview of their chemistry, interactions and potential environmental implications. Science of the Total Environment 400, 396–414
Kim, K. T., Klaine, S. J., Cho, J., Kim, S. H., & Kim, S. D. (2010). Oxidative stress responses of Daphnia magna exposed to TiO2 nanoparticles according to size fraction. Science of the Total Environment, 408, 2268–2272. https://doi.org/10.1016/j.scitotenv.2010.01.041.
Kim, K. T., Klaine, S. J., & Kim, S. D. (2014). Acute and chronic response of Daphnia magna exposed to TiO2 nanoparticles in agitation system. Bulletin of Environmental Contamination and Toxicology, 93(4), 456–460. https://doi.org/10.1007/s00128-014-1295-5.
Klaine, S. J., Alvarez, P. J. J., Batley, G. E., Fernandes, T. F., Handy, R. D., Lyon, D. Y., Mahendra, S., MCLaughlin, M. J., & Lead, J. R. (2008). Nanomaterials in the environmental: Behavior, fate, biovailability and effects. Environmental Toxicology and Chemistry, 27(9), 1825–1851. https://doi.org/10.1002/etc.4147.
Klerks, P. L., & Weis, J. L. (1987). Genetic adaptation to heavy metals in aquatic organisms: a review. Environmental Pollution, 45(3), 173–205. https://doi.org/10.1016/0269-7491(87)90057-1.
Krug, H. F., & Wick, P. (2011). Nanotoxicology: An interdisciplinary challenge. Angewandte Chemie (International Ed. in English), 50(6), 1260–1278. https://doi.org/10.1002/anie.201001037.
Lee, W. M., & An, Y. I. (2013). Effects of zinc oxide and titanium dioxide nanoparticles on green algae under visible, UV-A, and UV-B irradiations: no evidence of enhanced algal toxicity under UV pre-irradiation. Chemospere, 91, 536–544. https://doi.org/10.1016/j.chemosphere.2012.12.033.
Li, M., Czymmek, K. J., & Huanga, C. P. (2011). Responses of Ceriodaphnia dubia to TiO2 and Al2O3 nanoparticles: A dynamic nano-toxicity assessment of energy budget distribution. Journal of Hazardous Materials, 187(1-3), 502–508. https://doi.org/10.1016/j.jhazmat.2011.01.061.
Lin MY (2008) Interactions between titanium dioxide nanoparticles and algal cells at moderate particle concentration. Dissertation, University of Delaware.
Lombardi, A. T., Vieira, A. A. H., & Sartori, L. A. (2002). Mucilaginous capsule adsorption and intracellular uptake of copper by Kirchneriella aperta (Chlorococcales). Journal of Phycology, 38(2), 322–337. https://doi.org/10.1046/j.1529-8817.2002.00126.x.
Lovern, S. B., & Klaper, R. (2006). Daphnia magna mortality when exposed to titanium dioxide and fullerene (c60) nanoparticles. Environmental Toxicology and Chemistry, 25, 1132–1137. https://doi.org/10.1897/05-278R.1.
Lucca GM (2016) Efeitos ecotoxicológicos das nanopartículas de dióxido de titânio sobre a alga Pseudokirchneriella subcapitata e sobre o cladócero Ceriodaphnia silvestrii por diferentes vias de exposição. Dissertation, Universidade Federal de São Carlos.
Machado KS, Casali-Pereira MP, Oliveira DM, Isique WD, Leite MA, Minillo A (2009) Bioensaios de toxidade aguda com fármacos: diclofenaco de sódio e paracetamol. In: VI congresso de meio ambiente da AUGM, São Carlos. Anais de eventos da UFSCar. 5.
Mansano, A. S., Moreira, R. A., Dornfield, H. C., Diniz, L. G. R., Vieira, E. M., Daam, M. A., Rocha, O., & Seleghim, M. H. R. (2016). Acute and chronic toxicity of diuron and carbofuran to the neotropical cladoceran Ceriodaphnia silvestrii. Environmental Science and Pollution Research. https://doi.org/10.1007/s11356-016-8274-9.
Moreira, R. A., Mansano, A. S., Silva, L. C., & Rocha, O. (2014). A comparative study of the acute toxicity of the herbicide atrazine to cladocerans Daphnia magna, Ceriodaphnia silvestrii and Macrothrix flabelligera. Acta Limnologica Brasiliensia, 26(1), 1–8. https://doi.org/10.1590/S2179-975X2014000100002.
Nowack, B., & Bucheli, T. D. (2007). Occurrence, behavior and effects of nanoparticles in the environment. Environmental Pollution, 150(1), 5–22. https://doi.org/10.1016/j.envpol.2007.06.006.
OECD, Organization for Economic Cooperation and Development. (2008). Guidelines for testing of chemicals. Daphnia magna reproduction test. Paris: OECD 211.
Paschoalino, M. P., Marcone, G. P. S., & Jardim, W. F. (2010). Os nanomateriais e a questão ambiental. Quimica Nova, 33(2), 421–430. https://doi.org/10.1590/S0100-40422010000200033.
Petosa, A., Jaisi, D., Quevedo, I. R., Elimelech, M., & Tufenkji, N. (2010). Aggregation and deposition of engineered nanomaterials in aquatic environments: role of physicochemical interactions. Environmental Science & Technology, 44(17), 6532–6549. https://doi.org/10.1021/es100598h.
Rosa GAB (2008) Estudo dos efeitos do fármaco propranolol para Ceriodaphnia silvestrii (Cladocera, crustácea) com ênfase em efeitos nas populações. Dissertation. Universidade de São Paulo.
Sadiq, M., Dalai, S., Chandrasekaran, N., & Mukherjee, A. (2011). Ecotoxicity study of titania (TiO2) NPs on two microalgae species: Scenedesmus sp. and Chlorella sp. Ecotoxicology and Environmental Safety, 74(5), 118–1187. https://doi.org/10.1016/j.ecoenv.2011.03.006.
Schaumann, G. E., Phillippe, A., Bundschuh, M., Metreveli, G., Klitzke, S., Rakcheev, D., Grün, A., Kumahor, S., Kühn, M., Baumann, T., Lang, F., Manz, W., Schulz, R., & Vogel, H. J. (2015). Understanding the fate and biological effects of Agand TiO2-nanoparticles in the environment: the quest for advanced analytics and interdisciplinary concepts. The Science of the Total Environment, 535(1), 3–19. https://doi.org/10.1016/j.scitotenv.2014.10.035.
Salieri, B., Righi, S., Pasteris, A., & Olsen, S. I. (2015). Freshwater ecotoxicity characterization factor for metal oxide nanoparticles: a case study on titanium dioxide nanoparticle. The Science of the Total Environment, 505, 494–502. https://doi.org/10.1016/j.scitotenv.2014.09.107.
Smith, C. J., Shaw, B. J., & Handy, R. D. (2007). Toxicity of single walled carbon nanotubes to rainbow trout, (Oncorhynchus mykiss): respiratory toxicity, organ pathologies, and other physiological effects. Aquatic Toxicology, 82(2), 94–109. https://doi.org/10.1016/j.aquatox.2007.02.003.
Takenaka, R. A., Oliveira, M. J. D., & Rocha, O. (2007). Toxicidade de extratos de florações de cianobactérias de reservatórios do rio Tietê, SP, aos dafinídeos Ceriodaphnia dubia e Ceriodaphnia silvestrii (Cladocera, Crustacea). Journal of the Brazilian Society of Ecotoxicology, 2(2), 147–156. https://doi.org/10.5132/jbse.2007.02.007.
Tan, C., & Wang, W. X. (2017). Influences of TiO2 nanoparticles on dietary metal uptake in Daphnia magna. Environmental Pollution, 231(1), 311–318. https://doi.org/10.1016/j.envpol.2017.08.024.
USEPA, US Environmental Protection Agency. (2002). Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. Washington, DC: USEPA.
Wiench, K., Wohlleben, W., Hisgen, V., Radke, K., Salinas, E., Zok, S., & Landsiedel, R. (2009). Acute and chronic effects of nano- and non-nano-scale TiO2 and ZnO particles on mobility and reproduction of the freshwater invertebrate Daphnia magna. Chemosphere, 76(10), 1356–1365. https://doi.org/10.1016/j.chemosphere.2009.06.025.
Yu, H., Pan, J., Bai, Y., Zong, X., Li, X., & Wang, L. (2013). Hydrothermal synthesis of a crystalline rutile TiO2 nanorod based network for efficient dye-sensitized solar cells. Chemistry - A European Journal, 19(40), 13569–13574. https://doi.org/10.1002/chem.201300999.
Zhu, X., Zhu, L., Chen, Y., & Tian, S. (2009). Acute toxicities of manufactured nanomaterial suspensions to Daphnia magna. Journal of Nanoparticle Research, 11(1), 67–75. https://doi.org/10.1007/s11051-008-9426-8.
Zhu, X., Wang, J., Zhang, X., Chang, Y., & Chen, Y. (2010a). Trophic transfer of TiO2 nanoparticles from Daphnia to Zebrafish in a simplified freshwater food chain. Chemosphere, 79(9), 928–933. https://doi.org/10.1016/j.chemosphere.2010.03.022.
Zhu, X., Chang, Y., & Chen, Y. (2010b). Toxicity and bioaccumulation of TiO2 nanoparticle aggregates in Daphnia magna. Chemosphere, 78(3), 209–215. https://doi.org/10.1016/j.chemosphere.2009.11.013.
Acknowledgements
The first author would like to thank the “Coordenação de Aperfeiçoamento de Pessoal do Ensino Superior” (CAPES) for the scholarship. The authors would also like to thank the “Conselho Nacional de Desenvolvimento Científico e Tecnológico” (CNPq - process number 305229/2016-8) and the “Fundação de Amparo à Pesquisa do Estado de São Paulo” (FAPESP processes number 2014/14139-3 and 2016/00753-7) for the financial support. The authors thank Mrs. Jane Godwin Coury for the English language revision.
Funding
This study was funded by “Coordenação de Aperfeiçoamento de Pessoal do Ensino Superior” (CAPES) (scholarship), “Conselho Nacional de Desenvolvimento Científico e Tecnológico” (CNPq - process number 305229/2016-8), and the “Fundação de Amparo à Pesquisa do Estado de São Paulo” (FAPESP processes number 2014/14139-3 and 2016/00753-7) (financial support).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Highlights
• Toxicity of TiO2 NPs was determined to a Neotropical cladoceran.
• Cultures of the microalgae Raphidocelis subcapitata were contaminated with TiO2 NPs.
• Chronic effects of TiO2 NPs were evaluated by dietary route.
• The route of exposure induced high mortality of tropical cladocerans exposed to low concentrations of TiO2 NPS.
• From the concentration of 0.01 mg L−1 TiO2 NPs, there was a decrease in the survival of females of Ceriodaphnia silvestrii.
Rights and permissions
About this article
Cite this article
de Lucca, G.M., Freitas, E.C. & da Graça Gama Melão, M. Effects of TiO2 Nanoparticles on the Neotropical Cladoceran Ceriodaphnia silvestrii by Waterborne and Dietary Routes. Water Air Soil Pollut 229, 307 (2018). https://doi.org/10.1007/s11270-018-3964-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-018-3964-1