Abstract
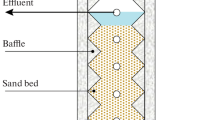
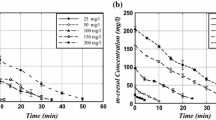
Biodegradation of phenol, o-cresol, and p-cresol was evaluated in continuous flow circulating packed bed bioreactors. Effect of loading rate on the removal rate of phenolic compounds was assessed by varying the influent concentration and flow rate. Regardless of the nature of phenolic compounds at a constant concentration, increase of loading rate caused the removal rate to pass through a maximum and then decline. Influent concentrations of 100 and 300 mg L−1 did not affect the removal rates of phenol and p-cresol, but higher rates were obtained at 500 mg L−1. With o-cresol, increase of influent concentration from 100 to 300 mg L−1 enhanced the removal rate but no further enhancement was observed at 500 mg L−1. The maximum removal rates for phenol, o-cresol, and p-cresol were 67.6–97.8, 38.7–73.8, and 77.2–107.2 mg L−1 h−1 at loading rates of 96.7–171.1, 61.6–163.9, and 87.4–183.9 mg L−1 h−1 (hydraulic residence time 2.9, 1.7–1.8, 1.2–2.8 h), respectively. Toxicity examination of untreated influents singled out phenol as the least toxic compound, while o-cresol and p-cresol showed similar toxicities. Treatment of influent containing 100 mg L−1 of phenolic compound led to the highest decrease in toxicity (> 93%) for all three compounds. With influent concentrations of 300 mg L−1 or higher, the decrease in toxicity of treated effluent was highest for phenol, followed by p-cresol, and then o-cresol. Finally, the observed patterns revealed that the toxicity of treated effluent was affected by the extent of biodegradation, as well as the toxic nature of phenolic compound under treatment.





Similar content being viewed by others
References
Ahamad, P. Y. A., Kunhi, A. A. M., & Divakar, S. (2001). New metabolic pathway for o-cresol degradation by Pseudomonas sp. CP4 as evidenced by 1H NMR spectroscopic studies. World Journal of Microbiology & Biotechnology, 17, 371–377. https://doi.org/10.1023/A:1016611702882.
Ailijiang, N., Chang, J., Liang, P., Li, P., Wu, Q., Zhang, X., & Huang, X. (2016). Electrical stimulation on biodegradation of phenol and responses of microbial communities in conductive carriers supported biofilms of the bioelectrochemical reactor. Bioresource Technology., 201, 1–7. https://doi.org/10.1016/j.biortech.2015.11.026.
Alkhalid, T., & El-Naas, M. H. (2012). Aerobic biodegradation of phenols: a comprehensive review. Critical Reviews in Environmental Science and Technology, 42, 1631–1690. https://doi.org/10.1080/10643389.2011.569872.
Aneez Ahamad, P. Y. A., & Kunhi, A. A. M. (2011). Enhanced degradation of phenol by Pseudomonas sp. CP4 entrapped in agar and calcium alginate beads in batch and continuous processes. Biodegradation, 22, 253–265. https://doi.org/10.1007/s10532-010-9392-6.
Balasubramanian, A., & Venkatesan, S. (2012). Removal of phenolic compounds from aqueous solutions by emulsion liquid membrane containing ionic liquid [BMIM]+ [PF 6]− in tributyl phosphate. Desalination, 289, 27–34. https://doi.org/10.1016/j.desal.2011.12.027.
Banerjee, A., & Ghoshal, A. K. (2010). Phenol degradation by Bacillus cereus: pathway and kinetic modeling. Bioresource Technology., 101, 5501–5507. https://doi.org/10.1016/j.biortech.2010.02.018.
Banerjee, A., & Ghoshal, A. K. (2016). Biodegradation of real petroleum wastewater by immobilized hyper phenol-tolerant strains of Bacillus cereus in a fluidized bed bioreactor. Biotechnology, 6, 137–141. https://doi.org/10.1007/s13205-016-0447-1.
Bansal, V. K., Kumar, R., Prasad, R., & Prasad, S. (2008). Catalytic chemical and electrochemical wet oxidation of phenol using new copper (II) tetraazamacrocycle complexes under homogeneous conditions. Journal of Molecular Catalysis A: Chemical, 284(1), 69–76. https://doi.org/10.1016/j.molcata.2007.12.030.
Box, G. E., Hunter, J. S., & Hunter, W. G. (2005). Statistics for experimenters: design, innovation, and discovery. New York City, NY: Wiley-Interscience.
Dey, S., & Mukherjee, S. (2013). Performance study and kinetic modeling of hybrid bioreactor for treatment of bi-substrate mixture of phenol-m-cresol in wastewater: process optimization with response surface methodology. Journal of Environmental Sciences., 25(4), 698–709. https://doi.org/10.1016/S1001-0742(12)60096-5.
D'Souza, L., Sami, Y., Nemati, M., & Headley, J. (2014). Continuous co-biodegradation of linear and cyclic naphthenic acids in circulating packed-bed bioreactors. Environmental Progress & Sustainable Energy, 33(3), 835–843. https://doi.org/10.1002/ep.11856.
El-Naas, M. H., Alhaija, M. A., & Al-Zuhair, S. (2017). Evaluation of an activated carbon packed bed for the adsorption of phenols from petroleum refinery wastewater. Environmental Science and Pollution Research, 24(8), 7511–7520. https://doi.org/10.1007/s11356-017-8469-8.
Environment and Climate Change Canada (ECCC). (2018). www.canada.ca/en/environment-climate-change/services/canadian-environmental-protection-act-registry/substances-list/priority-list.html. Accessed 25 Feb 2018.
Farooqi, I. H., Basheer, F., & Hasnain Isa, M. (2007). Co-degradation of phenol and m-cresols by upflow anaerobic sludge blanket reactor. Water Science & Technology., 56(7), 73–79. https://doi.org/10.2166/wst.2007.677.
Gupta, V. K., Nayak, A., Agarwal, S., & Tyagi, I. (2014). Potential of activated carbon from waste rubber tire for the adsorption of phenolics: effect of pre-treatment conditions. Journal of Colloid and Interface Science, 417, 420–430. https://doi.org/10.1016/j.jcis.2013.11.067.
Haddadi, A., & Shavandi, M. (2013). Biodegradation of phenol in hypersaline conditions by Halomonas sp. strain PH2-2 isolated from saline soil. International Biodeterioration & Biodegradation, 85, 29–34. https://doi.org/10.1016/j.ibiod.2013.06.005.
Huang, J., Nemati, M., Hill, G., & Headley, J. (2012). Batch and continuous biodegradation of three model naphthenic acids in a circulating packed-bed bioreactor. Journal of Hazardous Materials, 201, 132–140. https://doi.org/10.1016/j.jhazmat.2011.11.052.
Jemaat, Z., Sua’rez-Ojeda, E. M., Pe’rez, J., & Carrera, J. (2014). Partial nitritation and o-cresol removal with aerobic granular biomass in a continuous airlift reactor. Water Research, 48, 354–362. https://doi.org/10.1016/j.watres.2013.09.048.
Kapałka, A., Fóti, G., & Comninellis, C. (2009). The importance of electrode material in environmental electrochemistry: formation and reactivity of free hydroxyl radicals on boron-doped diamond electrodes. Electrochimica Acta, 54(7), 2018–2023. https://doi.org/10.1016/j.electacta.2008.06.045.
Kotreshaa, D., & Vidyasagar, G. M. (2017). Phenol degradation in a packed bed reactor by immobilized cells of Pseudomonas aeruginosa MTCC 4997. Biocatalysis and Agricultural Biotechnology, 10, 386–389. https://doi.org/10.1016/j.bcab.2017.04.015.
Kurzbaum, E., Raizner, Y., Cohen, O., Suckeveriene, R. Y., Kulikov, A., Hakimi, A., Kruh, L. I., Armon, R., Farber, Y., & Menashe, O. (2017). Encapsulated Pseudomonas putida for phenol biodegradation: use of a structural membrane for construction of a well-organized confined particle. Water Research, 121, 37–45. https://doi.org/10.1016/j.watres.2017.04.079.
Liu, J., Xie, J., Ren, Z., & Zhang, W. (2013). Solvent extraction of phenol with cumene from wastewater. Desalination and Water Treatment, 51(19–21), 3826–3831. https://doi.org/10.1080/19443994.2013.796993.
Marrot, B., Barrios-Martinez, A., Moulin, P., & Roche, N. (2006). Biodegradation of high phenol concentration by activated sludge in an immersed membrane bioreactor. Biochemical Engineering Journal, 30(2), 174–183.
Medel, A., Bustos, E., Esquivel, K., Godínez, L. A., & Meas, Y. (2012). Electrochemical incineration of phenolic compounds from the hydrocarbon industry using boron-doped diamond electrodes. International Journal of Photoenergy, 681875, 6 pages. https://doi.org/10.1155/2012/681875.
Moreno, L., Nemati, M., & Predicala, B. (2018). Biodegradation of phenol in batch and continuous flow microbial fuel cells with rod and granular graphite electrodes. Environmental Technology, 39(2), 144–156. https://doi.org/10.1080/09593330.2017.1296895.
Olmez-Hanci, T., & Arslan-Alaton, I. (2013). Comparison of sulfate and hydroxyl radical based advanced oxidation of phenol. Chemical Engineering Journal, 224, 10–16. https://doi.org/10.1016/j.cej.2012.11.007.
Pérez-González, D., Gómez, J., & Beristain-Cardoso, R. (2012). Biological removal of p-cresol, phenol, p-hydroxybenzoate and ammonium using a nitrifying continuous-flow reactor. Bioresource Technology., 120, 194–198. https://doi.org/10.1016/j.biortech.2012.06.052.
Praveen, P., & Loh, K. C. (2016). Osmotic membrane bioreactor for phenol biodegradation under continuous operation. Journal of Hazardous Materials, 305, 115–122. https://doi.org/10.1016/j.jhazmat.2015.11.034.
Praveen, P., Nguyen, D. T. T., & Loh, K. C. (2015). Biodegradation of phenol from saline wastewater using forward osmotic hollow fiber membrane bioreactor coupled chemostat. Biochemical Engineering Journal., 94, 125–133. https://doi.org/10.1016/j.bej.2014.11.014.
Qiao, J. Q., Yuan, N., Tang, C. J., Yang, J., Zhou, J., Lian, H. Z., & Dong, L. (2012). Determination of catalytic oxidation products of phenol by RP-HPLC. Research on Chemical Intermediates, 38(2), 549–558. https://doi.org/10.1007/s11164-011-0370-3.
Ramos, C., Suárez-Ojeda, M. E., & Carrera, J. (2016). Long-term performance and stability of a continuous granular airlift reactor treating a high-strength wastewater containing a mixture of aromatic compounds. Journal of Hazardous Materials., 303, 154–161. https://doi.org/10.1016/j.jhazmat.2015.10.031.
Riess, R., Nemati, M., & Hill, G. (2005). Improved mass transfer and biodegradation rates of naphthalene particles using a novel bead mill bioreactor. Journal of Chemical Technology and Biotechnology., 80, 662–668. https://doi.org/10.1002/jctb.1247.
Sabarunisha Begum, S., & Radha, K. V. (2013). Biodegradation kinetic studies on phenol in internal draft tube (inverse fluidized bed) biofilm reactor using Pseudomonas fluorescens: performance evaluation of biofilm and biomass characteristics. Bioremediation Journal, 17(4), 264–277. https://doi.org/10.1080/10889868.2013.827622.
Sahariaha, B. P., Anandkumar, J., & Chakraborty, S. (2017). Stability of continuous and fed batch sequential anaerobic–anoxic–aerobic moving bed bioreactor systems at phenol shock load application. Environmental technology. Published online. https://doi.org/10.1080/09593330.2017.1343388.
Saravanan, P., Pakshirajan, K., & Saha, P. (2008). Kinetics of phenol and m-cresol biodegradation by an indigenous mixed microbial culture isolated from a sewage treatment plant. Journal of Environmental Sciences., 20, 1508–1513. https://doi.org/10.1016/S1001-0742(08)62557-7.
Saravanan, P., Pakshirajan, K., & Saha, P. (2009). Treatment of phenolics containing synthetic wastewater in an internal loop airlift bioreactor (ILALR) using indigenous mixed strain of Pseudomonas sp. under continuous mode of operation. Bioresource Technology., 100, 4111–4116. https://doi.org/10.1016/j.biortech.2009.03.053.
Saravanan, P., Pakshirajan, K., & Saha, P. (2011). Studies on growth kinetics of predominantly Pseudomonas sp. in internal loop airlift bioreactor using phenol and m-cresol. Korean Journal of Chemical Engineering., 28(7), 1550–1555. https://doi.org/10.1007/s11814-010-0531-x.
Silva, C. D., Gómez, J., Houbron, E., Cuervo-López, F. M., & Texier, A.-C. (2009). p-Cresol biotransformation by a nitrifying consortium. Chemosphere, 75, 1387–1391. https://doi.org/10.1016/j.chemosphere.2009.02.059.
Surkatti, R., & El-Naas, M. H. (2014). Biological treatment of wastewater contaminated with p-cresol using Pseudomonas putida immobilized in polyvinyl alcohol (PVA) gel. Journal of Water Process Engineering., 1, 84–90. https://doi.org/10.1016/j.jwpe.2014.03.008.
Toussaint, M. W., Shedd, T. R., van der Schalie, W. H., & Leather, G. R. (1995). A comparison of standard acute toxicity tests with rapid-screening toxicity tests. Environmental Toxicology and Chemistry, 14(5), 907–915. https://doi.org/10.1002/etc.5620140524.
US Environmental Protection Agency (US EPA). www.epa.gov/sites/production/files/2015-09/documents/priority-pollutant-list-epa.pdf Accessed 25 Feb 2018.
Verschueren, K. (2001). Handbook of environmental data on organic chemicals: Vol. 1 (Ed. 4). New York City, NY: John Wiley and Sons.
Wang, S., & Loh, K. (1999). Modeling the role of metabolic intermediates in kinetics of phenol biodegradation. Enzyme and Microbial Technology., 25, 177–184. https://doi.org/10.1016/S0141-0229(99)00060-5.
Xu, C., Han, H., Jia, S., & Zhao, Q. (2016). Influence of phenol on ammonia removal in an intermittent aeration bioreactor treating biologically pretreated coal gasification wastewater. Journal of Environmental Sciences., 43, 99–105. https://doi.org/10.1016/j.jes.2015.08.013.
Yaacob, N. S., Mohamad, R., Ahmad, S. A., Abdullah, H., Ibrahim, A. L., & Bin Ariff, A. (2016). The influence of different modes of bioreactor operation on the efficiency of phenol degradation by Rhodococcus UKMP-5M. Rendiconti Lincei Scienze Fisiche e Naturali., 27, 749–760. https://doi.org/10.1007/s12210-016-0567-x.
Acknowledgements
Provision of a scholarship from the China Scholarship Council to Yi Zhou is gratefully acknowledged.
Funding
This study was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) (Discovery Grants #261376-2013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, Y., Nemati, M. Treatment of Waters Contaminated by Phenol and Cresols in Circulating Packed Bed Bioreactors—Biodegradation and Toxicity Evaluations. Water Air Soil Pollut 229, 288 (2018). https://doi.org/10.1007/s11270-018-3949-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-018-3949-0