Abstract
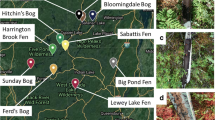
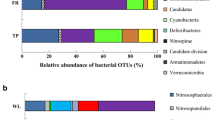
Peatlands serve as important stores of organic matter and regulators of nutrient and metal export to surface waters, yet relatively little is known regarding the impact of more than a century of metal, sulfur, and acid deposition on microbial activity in acidic, nutrient-poor peatlands that are common features around Sudbury, Ontario. In this study, eight peatlands were selected at varying distances from the Copper Cliff Smelter that was once the largest point source of sulfur dioxide and sampled for analysis of nutrient and metal content. Basal microbial respiration, relative response to substrate addition (four synthetic and four natural substrates) assessed as CO2 production rates and microbial biomass were assessed in surface (0–10 cm) peat samples. Bacterial and fungal communities within the peat samples were profiled using terminal restriction fragment length polymorphism analysis. Basal respiration (i.e., carbon mineralization in absence of substrate addition) was lowest and Cu and Ni concentrations and the degree of humification (assessed by the von Post scale) in surface peat samples were highest close to the smelter. Each peatland had a unique bacterial community when assessed using non-metric multidimensional scaling, whereas the fungal community was variable with no consistent patterns across the sites. Despite differences in microbial communities, substrate-induced respiration rates did not differ among peatlands as sites generally responded similarly to carbon substrate additions. Basal respiration rates were related to the humification status of the peat, which was potentially related to environmental degradation in the peatlands or surrounding terrestrial systems closer to the Sudbury smelters.




Similar content being viewed by others
References
Anand, M., Ma, K., Okonski, A., Levin, S., & McCreath, D. (2003). Characterising biocomplexity and soil microbial dynamics along a smelter-damaged landscape gradient. The Science of the Total Environment, 311, 247–259.
Avis, P. G., Dickie, I., & Mueller, G. M. (2006). A “dirty” business: testing the limitations of terminal restriction fragment length polymorphism (TRFLP) analysis of soil fungi. Molecular Ecology, 15(3), 873–882.
Azarbad, H., Niklińska, M., van Gestel, C. A. M., van Straalen, N. M., Röling, W. F. M., & Laskowski, R. (2013). Microbial community structure and functioning along metal pollution gradients. Environmental Toxicology and Chemistry, 32, 1992–2002.
Bååth, E. (1989). Effects of heavy metals in soil on microbial processes and populations (a review). Water, Air, and Soil Pollution, 47, 335–379.
Bååth, E., Díaz-Raviña, M., & Bakken, L. R. (2005). Microbial biomass, community structure and metal tolerance of a naturally Pb-enriched forest soil. Microbial Ecology, 50, 496–505.
Barrett, S. (2014). Factors controlling peat chemistry and vegetation composition in Sudbury peatlands after 30 years of emission reductions. MSc. Thesis, Trent University, Peterborough, Ontario, Canada.
Basiliko, N., & Yavitt, J. B. (2001). Influence of Ni, Co, Fe, and Na additions on methane production in Sphagnum-dominated Northern American peatlands. Biogeochemistry, 52, 133–153.
Brandt, K. K., Frandsen, R. J. N., Holm, P. E., & Nybroe, O. (2010). Development of pollution-induced community tolerance is linked to structural and functional resilience of a soil bacterial community following a five-year field exposure to copper. Soil Biology and Biochemistry, 42, 748–757.
Bräuer, S. L., Cadillo-Quiroz, H., Yashiro, E., Yavitt, J. B., & Zinder, S. H. (2006). Isolation of a novel acidiphilic methanogen from an acidic peat bog. Nature, 442, 192–194. doi:10.1038/nature04810.
Bridgham, S. D., & Richardson, C. J. (1992). Mechanisms controlling soil respiration (CO2 and CH4) in southern peatlands. Soil Biology and Biogeochemistry, 24(11), 1089–1099.
Brūmelis, G., Lapina, L., Nikodemus, O., & Tabors, G. (2002). Use of the O horizon of forest soils in monitoring metal deposition in Latvia. Water, Air, and Soil Pollution, 135, 291–309.
Chodak, M., Gołębiewski, M., Morawska-Płoskonka, J., Kuduk, K., & Niklińska, M. (2013). Diversity of microorganisms from forest soils differently polluted with heavy metals. Applied Soil Ecology, 64, 7–14.
Cox, R. M., & Hutchinson, T. C. (1979). Metal co-tolerance in the grass Deschampsia caespitosa. Nature, 279, 231–233.
Cox, R. M., & Hutchinson, T. C. (1980). Multiple metal tolerances in the grass Deschampsia caespitosa from the Sudbury smelting area. New Phytologist, 84, 631–647.
Dudka, S., Ponce-Hernandez, R., & Hutchinson, T. C. (1995). Current level of total element concentrations in the surface layer of Sudbury’s soils. The Science of the Total Environment, 162(2–3), 161–171.
Edel-Hermann, V., Dreumont, C., Pérez-Piqueres, A., & Steinberg, C. (2004). Terminal restriction fragment length polymorphism analysis of ribosomal RNA genes to assess changes in fungal community structure in soils. FEMS Microbiology Ecology, 47, 397–404. doi:10.1016/S0168-6496(04)00002-9.
Environmental Conservation Task Force. (1981). Ecological land survey guidelines for environmental impact analysis. Ecological Land Classification Series No. 13. Federal Environmental Assessment and Review Process, Land Directorate. Ottawa, Ontario: Environment Canada and Federal Environmental Assessment Review Office.
Feinstein, L. M., Sul, W. J., & Blackwood, C. B. (2009). Assessment of bias associated with incomplete extraction of microbial DNA from soil. Applied and Environmental Microbiology, 75(16), 5428–5433.
Freedman, B., & Hutchinson, T. C. (1980a). Pollutant inputs from the atmosphere and accumulations in soils and vegetation near a nickel-copper smelter at Sudbury, Ontario, Canada. Canadian Journal of Botany, 58, 108–132.
Freedman, B., & Hutchinson, T. C. (1980b). Effects of smelter pollutants on forest leaf litter decomposition near a nickel-copper smelter at Sudbury, Ontario. Canadian Journal of Botany, 58, 1722–1736.
Gans, J., Wolinsky, M., & Dunbar, J. (2005). Reveal great bacterial diversity and high metal toxicity in soil. Science, 309, 1387–1390.
Gignac, L. D., & Beckett, P. J. (1986). The effect of smelting operations on peatlands near Sudbury, Ontario, Canada. Canadian Journal of Botany, 64, 1138–1147.
Gorham, E. (1991). Northern peatlands: role in the carbon cycle and probable responses to climatic warming. Ecological Applications, 1(2), 182–195.
Gunn, J., Keller, W., Negusanti, J., Rotvin, R., Beckett, P., & Winterhalder, K. (1995). Ecosystem recovery after emission reductions: Sudbury, Ontario. Water, Air, and Soil Pollution, 85, 1783–1788.
Juckers, M., & Watmough, S. A. (2014). Impacts of simulated drought on pore-water chemistry of peatlands. Environmental Pollution, 184, 73–80.
Kim, S.-Y., Lee, S.-H., Freeman, C., Fenner, N., & Kang, H. (2008). Comparative analysis of soil microbial communities and their responses to the short-term drought in bog, fen, and riparian wetlands. Soil Biology and Biochemistry, 40, 2874–2880.
Lauber, C. L., Zhou, N., Gordon, J. I., Knight, R., & Fierer, N. (2010). Effect of storage conditions on the assessment of bacterial community structure in soil and human-associated samples. FEMS Microbiology Letters, 307(1), 80–86.
Levesque, M. P., & Mathur, S. P. (1979). A comparison of various means of measuring the degree of decomposition of virgin peat materials in the context of their relative biodegradability. Canadian Journal of Soil Science, 59(4), 397–400.
Lin, X., Green, S., Tfaily, M. M., Prakash, O., Konstantinidis, K. T., Corbett, J. E., Chanton, J. P., Cooper, W. T., & Kostka, J. E. (2012). Microbial community structure and activity linked to contrasting biogeochemical gradients in bog and fen environments of the Glacial Lake Agassiz Peatland. Applied and Environmental Microbiology, 78, 7023–7031.
Liu, Y., Zhou, T., Crowley, D., Li, L., Liu, D., Zheng, J., Yu, X., Pan, G., Hussain, Q., Zhang, X., & Zheng, J. (2012). Decline in topsoil microbial quotient, fungal abundance and C utilization efficiency of rice paddies under heavy metal pollution across South China. PLoS ONE, 7(6), e38858.
Lueders, T., & Friedrich, M. W. (2003). Evaluation of PCR amplification bias by terminal restriction fragment length polymorphism analysis of small-subunit rRNA and mcrA genes by using defined template mixtures of methanogenic pure cultures and soil DNA extracts. Applied and Environmental Microbiology, 69, 320–326.
Lukow, T., Dunfield, P. F., & Liesak, W. (2000). Use of the T-RFLP technique to assess spatial and temporal changes in the bacterial community structure within an agricultural soil planted with transgenic and non-transgenic potato plants. FEMS Microbiology Ecology, 32(3), 241–247.
McCall, J., Gunn, J., & Struik, H. (1995). Photo interpretive study of recovery of damaged lands near the metal smelters of Sudbury, Canada. Water, Air, and Soil Pollution, 85, 847–852.
Mertens, J., Springael, D., Troyer, I. D., Cheyns, K., Wattiau, P., & Smolders, E. (2006). Long-term exposure to elevated zinc concentrations induced structural changes and zinc tolerance of the nitrifying community in soil. Environmental Microbiology, 8(12), 2170–2178.
Niklińska, M., Chodak, M., & Laskowski, R. (2005). Characterization of the forest humus microbial community in a heavy metal polluted area. Soil Biology and Biochemistry, 37, 2185–2194.
Ocio, J. A., & Brookes, P. C. (1990). An evaluation of methods for measuring the microbial biomass in soils following recent additions of wheat straw and the characterization of the biomass that develops. Soil Biology and Biochemistry, 22(5), 685–694.
Oksanen, J. (2011). Multivariate analysis of ecological communities in R: vegan tutorial. R package version, 2–0, 1–43.
Peltoniemi, K., Fritze, H., & Laiho, R. (2009). Response of fungal and actinobacterial communities to water-level drawdown in boreal peatland sites. Soil Biology and Biochemistry, 41, 1902–1914.
Pester, M., Knorr, K. H., Friedrich, M. W., Wagner, M., & Loy, A. (2012). Sulfate-reducing microorganisms in wetlands—fameless actors in carbon cycling and climate change. Frontiers in Microbiology, 3, 1–19.
Preston, M. D., Smemo, K. A., McLaughlin, J. W., & Basiliko, N. (2012). Peatland microbial communities and decomposition processes in the James Bay Lowlands, Canada. Frontiers in Microbiology, 3, 1–15.
Rousk, J., Brookes, P. C., & Bååth, E. (2009). Contrasting soil pH effects on fungal and bacterial growth suggest functional redundandcy in carbon mineralization. Applied and Environmental Microbiology, 75, 1589–1596.
Rousk, J., Bååth, E., Brookes, P. C., Lauber, C. L., Lozupone, C., Caporaso, J. G., Knight, R., & Fierer, N. (2010). Soil bacterial and fungal communities across a pH gradient in an arable soil. The ISME Journal, 4, 1340–1351.
Rydin, H. & Jeglum, J. (2006). The biology of peatlands. London, UK: Oxford University Press.
Sandaa, R., Torsvik, V., Enger, Ø., Daae, F. L., Castberg, T., & Hahn, D. (1999). Analysis of bacterial communities in heavy metal-contaminated soils at different levels of resolution. FEMS Microbiology Ecology, 30(3), 237–251.
Schipper, L. A., & Lee, W. G. (2004). Microbial biomass, respiration and diversity in ultramafic soils of West Dome, New Zealand. Plant and Soil, 262, 151–158.
Schütte, U. M. E., Abdo, Z., Bent, S. J., Shyu, C., Williams, C. J., Pierson, J. D., & Forney, L. J. (2008). Advances in the use of terminal restriction fragment length polymorphism (T-RFLP) analysis of 16S rRNA genes to characterize microbial communities. Applied Microbiology and Biotechnology, 80, 365–380.
Soil Classification Working Group (1998). The Canadian System of Soil Classification. Ottawa, ON, Agriculture and Agri-Food Canada 187 Publication 1646
Szkokan-Emilson, E. J., Kielstra, B., Watmough, S. A., & Gunn, J. M. (2014a). Drought-induced release of metals from peatlands in watersheds recovering from historical metal and sulphur deposition. Biogeochemistry, 116, 131–145.
Szkokan-Emilson, E. J., Watmough, S. A., & Gunn, J. M. (2014b). Wetlands as long-term sources of metals to receiving waters in mining-impacted landscapes. Environmental Pollution, 192, 91–103.
Stefanowicz, A. M., Niklińska, M., & Laskowski, R. (2008). Metals affect soil bacterial and fungal functional diversity differently. Environmental Toxicology and Chemistry / SETAC, 27, 591–8.
Strickland, M. S., Osburn, E., Lauber, C., Fierer, N., & Bradford, M. A. (2009). Litter quality is in the eye of the beholder: initial decomposition rates as a function of inoculum characteristics. Functional Ecology, 23(3), 627–636.
Taylor, G. J., & Crowder, A. A. (1983). Accumulation of atmospherically deposited metals in wetland soils of Sudbury, Ontario. Water, Air, and Soil Pollution, 19, 29–42.
Ukonmaanaho, L., Nieminen, T. M., Rausch, N., & Shotyk, W. (2004). Heavy metal and arsenic profiles in ombrogenous peat cores from four differently loaded areas in Finland. Water, Air, and Soil Pollution, 159, 277–294.
Vance, E. D., Brookes, P. C., & Jenkinson, D. S. (1987). An extraction method for measuring soil microbial biomass C. Soil biology and Biogeochemistry, 19(6), 70–707.
Von Post, L. (1937). The geographic survey of Irish bogs. The Irish Naturalist’s Journal, 6, 210–227.
Whitby, L. M., Stokes, P. M., Hutchinson, T. C., & Myslki, G. (1976). Ecological consequence of acidic and heavy-metal discharges from the Sudbury smelters. Canadian Mineralogist, 14, 47–57.
Winsborough, C., & Basiliko, N. (2010). Fungal and bacterial activity in Northern Peatlands. Geomicrobiology Journal, 27, 315–320.
Winterhalder, K. (1996). Environmental degradation and rehabilitation of the landscape around Sudbury, a major mining and smelting area. Environmental Reviews, 4(3), 185–224.
Acknowledgements
We thank the National Sciences and Engineering Research Council of Canada (NSERC), Vale INCO, Xstrata and the City of Greater Sudbury for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luke, S., Preston, M.D., Basiliko, N. et al. Microbial Communities, Biomass, and Carbon Mineralization in Acidic, Nutrient-Poor Peatlands Impacted by Metal and Acid Deposition. Water Air Soil Pollut 226, 19 (2015). https://doi.org/10.1007/s11270-014-2265-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-014-2265-6